Exhibit 99.3

IM Cannabis Corp.
Management’s Discussion and Analysis
For the Three and Nine Months Ended September 30, 2022
November 14, 2022
Management’s Discussion and Analysis
TABLE OF CONTENTS
| 5 |
|
| 7 |
|
| 7 |
|
| 10 |
|
| 12 |
|
| 14 |
|
| 16 |
|
| 21 |
|
| 51 |
2
Management’s Discussion and Analysis
INTRODUCTION
IM Cannabis Corp. (“IM Cannabis” or the “Company”) is a British Columbia company whose business formed on October
11, 2019 as the result of a reverse takeover with IMC Holdings Ltd. (the “Reverse Takeover Transaction”), pursuant to which the Company changed its name from “Navasota Resources Inc.” to “IM Cannabis Corp.”
and changed its business from mining to the international medical cannabis industry. The Company’s common shares (the “Common Shares”) trade under the ticker symbol “IMCC” on both the NASDAQ Capital Market
(“NASDAQ”) and the Canadian Securities Exchange (“CSE”) as of March 1, 2021 and November 5, 2019, respectively. The Reverse Takeover Transaction is more fully
described under “Review of Financial Performance – Share Capital – Financial Background”.
This Management’s Discussion and Analysis (“MD&A”) reports on the consolidated financial condition and operating results of IM Cannabis for the three
and nine months ended September 30, 2022. Throughout this MD&A, unless otherwise specified, references to “we”, “us”, “our” or similar terms, as well as the “Company” and “IM Cannabis” refer to IM Cannabis Corp., together with its
subsidiaries, on a consolidated basis, and the “Group” refers to the Company, its subsidiaries, Focus Medical Herbs Ltd. and High Way Shinua Ltd.
This MD&A should be read in conjunction with the interim condensed consolidated financial statements of the Company and the notes thereto for the three and nine months ended September 30,
2022 (the “Interim Financial Statements”), and with the Company’s audited annual consolidated financial statements and the notes thereto for the years ended December 31, 2021 and 2020 (the “Annual Financial Statements”). References herein to “Q3 2022” and “Q3 2021” refer to the threeFdis months ended September 30, 2022 and September 30, 2021, respectively, and references to “2021” refer to the
year ended December 31, 2021.
The Interim Financial Statements have been prepared by management in accordance with the International Financial Reporting Standards (“IFRS”) as issued by
the International Accounting Standards Board (“IASB”). IFRS requires management to make certain judgments, estimates and assumptions that affect the reported amount of assets and liabilities at the date of
the Interim Financial Statements and the amount of revenue and expenses incurred during the reporting period. The results of operations for the periods reflected herein are not necessarily indicative of results that may be expected for future
periods. The Interim Financial Statements include the accounts of the Group, which includes, among others, the following entities:
|
Legal Entity
|
Jurisdiction
|
Relationship with the Company
|
|
IMC Holdings Ltd. (“IMC Holdings”)
|
Israel
|
Wholly-owned subsidiary
|
|
I.M.C. Pharma Ltd.
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
Focus Medical Herbs Ltd. (“Focus”)
|
Israel
|
Private company over which IMC Holdings exercises “de facto control” under IFRS 10 Consolidated Financial Statements (“IFRS 10”)
|
|
R.A. Yarok Pharm Ltd. (“Pharm Yarok”)
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
Rosen High Way Ltd. (“Rosen High Way”)
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
High Way Shinua Ltd. (“HW Shinua”)
|
Israel
|
Private company over which IMC Holdings exercises “de facto” control under IFRS 10
|
|
Revoly Trading and Marketing Ltd. dba Vironna Pharm (“Vironna”)
|
Israel
|
Subsidiary of IMC Holdings
|
|
Oranim Plus Pharm Ltd. (“Oranim Plus”)
|
Israel
|
Subsidiary of IMC Holdings
|
|
Trichome Financial Corp. (“Trichome”)
|
Canada
|
Wholly-owned subsidiary
|
|
Trichome JWC Acquisition Corp. (“TJAC”)
|
Canada
|
Wholly-owned subsidiary of Trichome
|
|
MYM Nutraceuticals Inc. (“MYM”)
|
Canada
|
Wholly-owned subsidiary of Trichome
|
|
Highland Grow Inc. (“Highland”)
|
Canada
|
Wholly-owned subsidiary of MYM International Brands Inc., a wholly-owned subsidiary of MYM
|
|
Adjupharm GmbH (“Adjupharm”)
|
Germany
|
Subsidiary of IMC Holdings
|
3
Management’s Discussion and Analysis
All intercompany balances and transactions were eliminated on consolidation. All dollar figures in this MD&A are expressed in thousands of Canadian Dollars ($), except per share data and unless otherwise noted.
All references to “NIS” are to New Israeli Shekels. All references to “€” or to “Euros” are to Euros. All references to “US$” or to “U.S. Dollars” are to United States Dollars. The Company’s shares, options, units and warrants are not expressed
in thousands. Prices are not expressed in thousands.
NON-IFRS FINANCIAL MEASURES
Certain non-IFRS financial measures are referenced in this MD&A that do not have any standardized meaning under IFRS, including “Gross Margin”, “EBITDA” and “Adjusted EBITDA”. The Company
believes that these non-IFRS financial measures and operational performance measures, in addition to conventional measures prepared in accordance with IFRS, enable readers to evaluate the Company’s operating results, underlying performance and
prospects in a similar manner to the Company’s management. For a reconciliation of these non-IFRS financial measures to the most comparable IFRS financial measures, as applicable, see the “Metrics and Non-IFRS
Financial Measures” section of the MD&A.
NOTE REGARDING THE COMPANY’S ACCOUNTING PRACTICES
The Company complies with IFRS 10 to consolidate the financial results of Focus, a holder of an Israeli Medical Cannabis Agency (the “IMCA”) license which allows it to import and supply cannabis products, on the basis of which IMC Holdings exercises “de facto control”. For a full
explanation of the Company’s application of IFRS 10, see “Legal and Regulatory – Restructuring” and “Legal and Regulatory – Risk Factors”.
For the period ended September 30, 2022, the Company analyzed the terms of the definitive agreement with HW Shinua in accordance with IFRS 10 and concluded that it is required to consolidate the
financial results of HW Shinua as of the date of signing the definitive agreement therewith. The definitive agreement provides the Company with the power to unilaterally make all decisions regarding the financial and operating policies of HW
Shinua and the right to obtain all related economic benefits. HW Shinua is to be acquired by the Group pursuant to a July 28, 2021 definitive agreement in which IMC Holdings acquired all of the issued and
outstanding shares of each of (i) Pharm Yarok; and (ii) Rosen High Way; and will acquire HW Shinua, an applicant for a medical cannabis transportation license, upon receipt of requisite approvals from the IMCA.
4
Management’s Discussion and Analysis
OVERVIEW – CURRENT OPERATIONS IN ISRAEL, GERMANY AND CANADA
IM Cannabis is an international cannabis company providing premium cannabis products to medical patients in Israel and Germany, and adult-use recreational consumers
in Canada, the world’s three largest federally legal cannabis markets. The Company leverages a transnational ecosystem powered by a unique data-driven approach and a globally sourced product supply chain. With an unwavering commitment
to responsible growth and compliance with the strictest regulatory environments, the Company strives to amplify its commercial and brand power to become a global high-quality cannabis player.
In Israel, the Company imports, distributes and sells cannabis to local medical patients by operating medical cannabis retail pharmacies, online platforms,
distribution centres and logistical hubs operating through IMC Holdings’ subsidiaries and Focus, leveraging proprietary data and patient insights.
In Germany, the IM Cannabis ecosystem operates through Adjupharm, importing and distributing cannabis to pharmacies for patients, and acting as the Company’s entry point for
potential Europe-wide distribution in the future.
The Company operates in Canada through Trichome and its subsidiaries TJAC and MYM, where it cultivates, processes, packages and sells premium and ultra-premium cannabis at its own facilities under the WAGNERS and Highland Grow brands for the adult-use market in Canada, and exports premium and ultra-premium medical cannabis to
Israel. The Company commenced exiting the operations in Canada, and considers these operations as held for sale pursuant to IFRS.
On November 7, 2022, the Company announced that it is pivoting its focus and resources to achieve sustainable and profitable growth in its highest value markets, Israel and Germany, while also
commencing its exit from the Canadian cannabis market as part of the Company’s ongoing restructuring plan in Canada through which it is taking a disciplined approach to spending and implementing cost efficiencies (the “Canadian Restructuring”). For more information, see “Strategy in Detail – Geographies and Markets – Canada” and “Corporate Events and Highlights –
Subsequent Events” sections below.
OUR GOAL – DRIVE PROFITABLE REVENUE GROWTH
Our primary goal is to sustainably increase revenue in each of our core markets, concentrating efforts on the highest value markets in Israel and Germany to accelerate our path to profitability
and long-term shareholder value while actively managing costs and margins.
HOW WE PLAN TO ACHIEVE OUR GOAL – CORE STRATEGIES
Our strategy of sustainable and profitable growth consists of:
| • |
Continue building on the increasing demand and positive momentum in Israel, supported by strategic alliances with Canadian suppliers and a highly skilled sourcing team, to cement its leadership
position in Israel.
|
| • |
Develop and execute a long-term growth plan in Germany, based on the strong sourcing infrastructure in Israel which is powered by advanced product knowledge and regulatory expertise establishing, in the Company’s view, a competitive
advantage ahead of proposals for the legalization of recreational cannabis in Germany.
|
| • |
Properly position brands with respect to target-market, price, potency and quality, such as our IMC brand in Israel and Germany and the WAGNERS brand in Canada and Israel. The IMC brand’s legacy strain Roma® was
voted as the Favourite Strain of the Month in August 2022 by ‘Cannabis’, one of Israel’s leading cannabis blogs for Israeli medical cannabis patients.1
|
| • |
Strong focus on efficiencies and synergies as a global organization with domestic expertise in Israel and Germany.
|
| • |
High-quality, reliable supply to our customers and patients, leading to recurring sales.
|
| • |
Ongoing introduction of new SKUs to keep consumers and patients engaged.
|
1 https://cannbis.co.il/blog/%D7%96%D7%9F-%D7%94%D7%97%D7%95%D7%93%D7%A9-%D7%A8%D7%95%D7%9E%D7%90-%D7%A7%D7%A0%D7%90%D7%91%D7%99%D7%A1-%D7%A8%D7%A4%D7%95%D7%90%D7%99-%D7%90%D7%95%D7%92%D7%95%D7%A1%D7%98-22
5
Management’s Discussion and Analysis
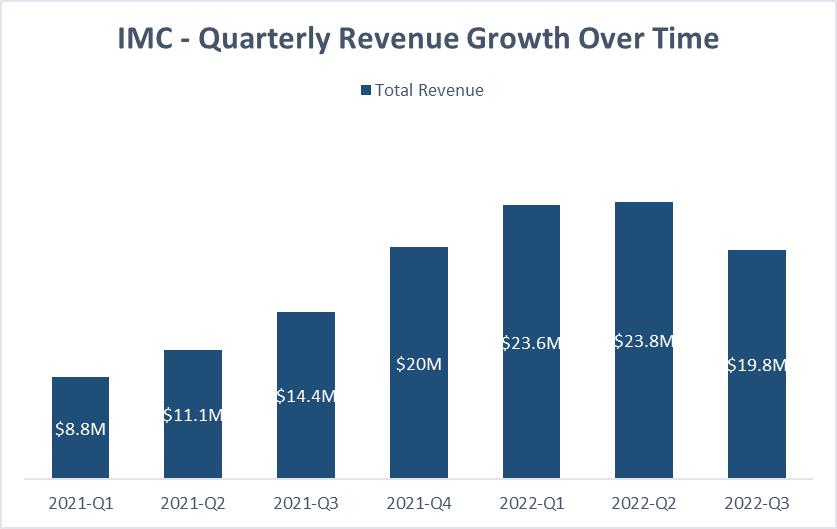
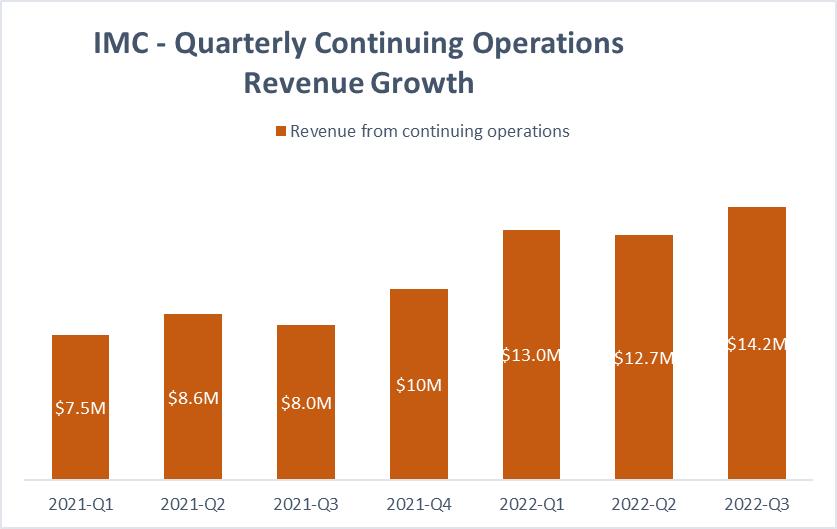
RESULTS – REVENUE GROWTH IN Q3 2022
6
Management’s Discussion and Analysis
The Company operates in the Israeli and German medical cannabis markets and the Canadian adult-use recreational market. Effective November 7, 2022, the Company commenced the process of exiting
the Canadian cannabis market to focus its resources on reinforcing and further pursuing growth opportunities in Israel, Germany and Europe, implementing a leaner organization strategy with the primary focus on achieving profitability in 2023.
Israel
In Israel, we continue to expand IMC brand recognition and supply the growing Israeli medical cannabis market with our branded products. The Company offers medical cannabis patients a rich
variety of high-end medical cannabis products through strategic alliances with Canadian suppliers supported by a highly skilled sourcing team. In addition to the benefits of the Group’s long-term presence in Israel, we believe that with our
strong sourcing infrastructure in Israel, and advanced product knowledge, regulatory expertise and strong commercial partnerships, the Company is well-positioned to address the ongoing needs and preferences of medical cannabis patients in Israel.
The Company entered additional segments of the medical cannabis value chain in Israel, namely the distribution and retail segments. The Company, through IMC Holdings, acquired three licensed
pharmacies in 2022, each selling medical cannabis products to patients: (i) Oranim Plus, Israel’s largest pharmacy in Jerusalem and third largest in Israel, (ii) Vironna, a leading pharmacy in the Arab sector, and (iii) Pharm Yarok, the largest
pharmacy in the Sharon plain area and the biggest call centre in the country (Oranim Plus, Vironna, and Pharm Yarok collectively, the “Israeli Pharmacies”).
The acquisitions of the Israeli Pharmacies position IM Cannabis as a large distributor of medical cannabis in Israel. We are strategically focused on establishing and reinforcing a direct
connection with medical cannabis patients, providing direct access to IM Cannabis products, obtaining and leveraging market data and gaining a deeper understanding of consumer preferences. The acquisition of the Israeli Pharmacies allows the
Company to increase purchasing power with third-party product suppliers, offers potential synergies with our established call centre and online operations, achieves higher margins on direct to patient sales and creates the opportunity for
up-sales across a growing range of products.
In addition, the Company acquired Rosen High Way, a trading house, increasing the Company’s market share in Israel and leading to the expansion of its sales channels, distribution, delivery and
storage capacity, and strengthened the Group’s ability to directly reach medical cannabis patients and service dozens of pharmacies across the country.
The Company has also acquired home-delivery services and an online retail footprint, operating under the name “Panaxia-to-the-Home”, which includes a
customer service centre and an Israeli medical cannabis distribution license, from Panaxia Pharmaceutical Industries Israel Ltd. and Panaxia Logistics Ltd., part of the Panaxia Labs Israel, Ltd. group of companies (the “Panaxia Transaction”). The Panaxia Transaction includes a further option (the “Panaxia Option”) to acquire a pharmacy for no additional consideration.
The closing of the Panaxia Option was initially expected to occur in Q3 2022, however the parties to the Panaxia Transaction are considering a new arrangement and are renegotiating the terms. Accordingly, the payment by the Company of the fifth
and final installment of the share consideration under the Panaxia Transaction will take place at a new date that is yet to be determined by the parties.
7
Management’s Discussion and Analysis
Germany
In Europe, the Company operates in Germany through Adjupharm, its German subsidiary and EU-GMP certified medical cannabis producer and distributor. We continue to lay our foundation in Germany,
which is currently the European market with the largest number of medical cannabis patients.2 Leveraging our global supply chain, IM Cannabis continues to focus on growing its business in
Germany to be well-positioned through brand recognition in preparation for future regulatory reforms.
Similar to Israel, the Company’s focus in Germany is to import premium and ultra-premium indoor-grown dried cannabis from its supply partners, which we believe will satisfy the rapid growth in
demand for high-THC premium and ultra-premium cannabis across a variety of strains and qualities.
In the Company’s view, the strong sourcing infrastructure in Israel, powered by advanced product knowledge and regulatory expertise, will establish a competitive advantage in Germany ahead of
proposals for the legalization of recreational cannabis. This is based on the premise that the German and Israeli markets share a number of common attributes such as robust commercial infrastructure, highly developed digital capabilities,
favourable demographics and customer preferences.
While the Company does not currently distribute products in other European countries, the Company intends to leverage the foundation established by Adjupharm, its state-of-the-art, approximately
8,000 square foot warehouse space and EU-GMP production facility in Germany (the “Logistics Centre”), and its network of distribution partners to expand into other jurisdictions across the continent. The
Company expects that the Logistics Centre will allow the Company to execute all aspects of its supply chain, including the repackaging of bulk cannabis and distribution capabilities.
Adjupharm received a revised EU-GMP license in May 2022 that permits it to engage in additional production, cannabis testing and release activities. It allows Adjupharm to repackage bulk
cannabis, to perform stability studies and offer such services to third parties.
Canada
Following the completion of the Company’s acquisition of Trichome on March 18, 2021 (the “Trichome Transaction”) and MYM on July 9, 2021 (the “MYM Transaction”), the Company’s global cannabis platform expanded to include operations in the adult-use recreational cannabis market in Canada to supplement its established medical cannabis operations in
Israel and Germany.
Through its wholly-owned subsidiary, TJAC, Trichome operates as a licensed producer of cannabis products in the Canadian cannabis market and sells adult-use recreational cannabis products under
the WAGNERS brand, which is widely available at cannabis retailers throughout Canada.
2 The European Cannabis Report – Edition 7 https://prohibitionpartners.com/2022/03/31/launching-today-the-european-cannabis-report-7th-edition/
8
Management’s Discussion and Analysis
MYM operates through its wholly-owned subsidiary, Highland, a licensed producer. Highland produces cannabis products for the adult-use recreational cannabis market under the Highland Grow brand,
which is widely available at cannabis retailers throughout Canada.
The WAGNERS brand, acquired in the Trichome Transaction, targets the premium cannabis market segment (ranging from $7.50-$9.99 per gram at the consumer level), while the Highland Grow brand,
acquired in the MYM Transaction, targets the ultra-premium market segment (at a price range starting at $12.99 per gram at the consumer level). The Trichome Transaction and the MYM Transaction together form the Company’s Canadian cultivation and
recreational sales operations.
In Q2 2022, the Company commenced the Canadian Restructuring aimed at saving approximately $1,000 quarterly in cash expenses. On August 5, 2022, as part of the Canadian Restructuring, the Company
entered into an agreement to sell all of the issued and outstanding shares of SublimeCulture Inc. (“Sublime”), halted cultivation at the Highland facility in Antigonish, Nova Scotia (“Highland Facility”), which continues to be used for packaging and storage, and conducted workforce reduction throughout its Canadian operations.
All Canadian cultivation, genetics, and logistics have been centralized in the Company’s facilities in Kitchener, Ontario which has sufficient cultivation capacity to support the streamlining
initiative.
On November 7, 2022, in connection with the Company’s efforts to achieve operational efficiencies, the Company announced that it is pivoting its focus and resources on growth in its highest value
markets in Israel and Germany while also commencing its exit from the Canadian cannabis market as part of the Canadian Restructuring. With this move, the Company aims for a leaner organization with a primary focus on achieving profitability in
2023.
Trichome, and certain of its wholly-owned subsidiaries, including TJAC, MYM, Trichome Retail Corp., MYM International Brands Inc., and Highland (collectively, the “Trichome Group”), filed and obtained creditor protection under the Canadian Companies’ Creditors Arrangement Act (“CCAA”) pursuant to an order of the
Ontario Superior Court of Justice (Commercial List) (the “Court”) on November 7, 2022. The CCAA proceedings are solely in respect of the Trichome Group. As such, the Company’s assets or subsidiaries in
Israel and Germany are not parties to the CCAA proceedings.
The Trichome Group is expected to continue to operate its business for the time being however, it is expected that there will be changes in the Canadian operations consistent with those of a
company operating through CCAA proceedings, including employee and contract terminations. The Trichome Group has said that it intends to use the CCAA proceedings to implement a sale and investment solicitation process for the sale of its assets
or restructuring of its business.
In connection with the CCAA proceedings, TJAC, as borrower (the "Borrower"), the remaining Trichome Group, as guarantors (together with the Borrower, the "Credit Parties"), and Courtland Credit Lending Corporation (the “DIP Lender”), entered into a debtor-in-possession (“DIP”)
facility agreement dated November 6, 2022 (the "DIP Agreement"). Pursuant to the DIP Agreement, the DIP Lender has agreed to provide a super-priority interim revolving credit facility (subject to certain
mandatory repayment provisions) to the Borrower (the "DIP Facility"). In accordance with the DIP Agreement, the DIP Facility is to be used during the CCAA proceedings by the Borrower to fund its working
capital needs. The DIP Facility is subject to customary covenants, conditions precedent, and representations and warranties made by the Credit Parties to the DIP Lender. The current DIP Lender's charge approved by Court is up to the maximum
amount of $1,825 reflecting the anticipated borrowings under the DIP Facility up to November 17, 2022.
For more information, see “Corporate Events and Highlights – Subsequent Events”.
9
Management’s Discussion and Analysis
The IMC brand is well-known in the Israeli medical cannabis market. Building on its long-term success in Israel, the Company launched the IMC brand in Germany in 2020.
The Company’s strategy is to leverage its international relationships and apply a multi-country strategy using established relationships from its global platform to import premium and
ultra-premium cannabis and high-end brands to the Israeli medical cannabis market with plans to import into Germany in the future as well. The Company believes that the import and sale of cannabis brands from leading cannabis suppliers and supply
partners into the Israeli and German markets, in addition to the sale of WAGNERS products, can satisfy the increasing demand of both Israeli and German patients for indoor grown high-THC premium cannabis.
Israeli Medical Cannabis Business
|
The Company currently sells four different product lines under the IMC brand as well as the WAGNERS brand in Israel, which was successfully launched in Israel
earlier this year.
|
 |
|
The IMC brand has established its reputation in Israel for quality and consistency over the past 10 years and more recently with new high-end, ultra-premium strains that have made it to
the top-sellers list in pharmacies across the country.
The Group maintains a portfolio of strains sold under the IMC umbrella from which popular medical cannabis dried flowers and full-spectrum cannabis extracts are produced.
In 2021, IMC was rebranded with a refreshed logo, packaging, design language and tone with a bold new design to better position itself in the competitive Israeli medical cannabis
market, and introduced a variety of new products for medical cannabis patients.
The IMC brand offers four different product lines, leading with the Craft Collection which offers the highest quality Canadian craft cannabis flower and has
established IMC as the leader of the ultra-premium segment in Israel, selling at the highest price points and margins.
|
|
|
The Craft Collection – The IMC brand’s ultra-premium product line with indoor-grown, hang-dried and hand-trimmed high-THC cannabis flowers. The
Craft Collection includes exotic and unique cannabis strains such as Cherry Crasher, Peanut Butter MAC and Watermelon Zkittlez.
|
 |
|
The Top-Shelf Collection – The newest addition to IMC’s brand portfolio, launched in September 2022 as IMC’s premium
product line, offers indoor-grown, high-THC cannabis flowers with strains such as Lemon Rocket and Diesel Drift. Inspired by the 1970’s cannabis culture in America, the Top-Shelf Collection targets the growing segment of medical
patients who are cannabis culture enthusiasts.
|
 |
10
Management’s Discussion and Analysis
The Signature Collection – The IMC brand’s high-quality product line with greenhouse-grown or indoor grown, high-THC cannabis flowers. The Signature
Collection currently includes well known cannabis dried flowers such as Roma®, Tel Aviv and London as well as the new strains launched in Q3 2022, Strawnana and Sydney.
The Full Spectrum Extracts – The IMC brand’s full spectrum, strain-specific cannabis extracts, including high-THC Roma® oil, balanced Paris oil and Super
CBD oil and the new Roma® T15 oil and Tel Aviv oil, which launched in Q3.
For more information, see “Strategy in Detail – Brands – New Product Offerings” section of the MD&A.
Canadian Adult-Use Recreational Cannabis Business
In Canada, the Company currently has a product portfolio in the premium dried flower, pre-roll, infused premium pre-roll and hash categories under the premium WAGNERS brand and ultra-premium
Highland Grow brand. The WAGNERS brand was acquired through the Trichome Transaction and launched by TJAC in mid-2021, while the Highland Grow brand was acquired through the MYM Transaction.
The WAGNERS brand offers consistent, premium cannabis at an approachable price point for consumers. The Highland Grow brand offers cannabis consumers an ultra-premium product, curated to their
tastes.
WAGNERS and Highland Grow products are primarily sold in 3.5 gram flower and 3 x 0.5 gram flower pre-roll formats. Other flower formats are available in certain provinces, such as 7 or 14 gram
units. Hash is typically sold in 1, 2 and 4 gram formats.
Key WAGNERS flower strains include Cherry Jam, Pink Bubba, Blue Lime Pie, Golden Ghost OG, Tiki Rain and Trpy Zlrp.

The Highland Grow brand portfolio includes five active flower strains: Diamond Breath, The Leviathan, Frostbite, Space Jager as well as White Lightning.

11
Management’s Discussion and Analysis
German Medical Cannabis Business
In Germany, the Company sells IMC-branded dried flower products. The medical cannabis products sold in the German market are branded generically as IMC to increase recognition of the Company’s
brand in establishing a foothold with German healthcare professionals. The Company’s IMC-branded cannabis products were launched in Germany with one high-THC flower strain in 2020. In Q4 2021, Adjupharm launched another high-THC flower strain and
two full spectrum extracts. In Q1 2022 Adjupharm launched a third strain, a high-CBD flower, to offer a more complete portfolio to German physicians and patients. In Q2 2022 the Company’s IMC Hindu Kush strain was the top selling T20 in the
market, strengthening Adjupharm’s position as one of the top 10 cannabis companies in Germany. September 2022 was Adjupharm’s strongest sales month to date.
In July 2021, Adjupharm was recognized by the German Brand Institute with the “German Brand Award 2021”, recognizing its excellence in brand strategy and creation, communication, and integrated
marketing. The Group’s competitive advantage in Germany lies in its track record, experience and brand reputation in Israel and proprietary data supporting the potential effectiveness of medical cannabis for the treatment of a variety of
conditions.

Israel
Over the last decade, Focus Medical was the primary cultivator of medical cannabis products sold under the IMC brand in the Israeli market. Until July 2022, Focus Medical held an IMCA license to
cultivate medical cannabis at its cultivation facility (the “Focus Facility”). In Q2 2022, the Company closed the Focus Facility to concentrate on leveraging its skilled sourcing team and strategic
alliances with Canadian suppliers as well as the import of medical cannabis from its Canadian Facilities. In July 2022, Focus Medical received an IMCA license which allows it to import cannabis products and supply medical cannabis to patients
through licensed pharmacies. To supplement growing demand, the Company plans to continue its relationships with third-party cultivation facilities in Israel for the propagation and cultivation of the Company’s existing proprietary genetics and
for the development of new products.
12
Management’s Discussion and Analysis
Pursuant to the applicable Israeli cannabis regulations, following the cultivation or import of medical cannabis, medical cannabis products are then packaged by contracted licensed producers of
medical cannabis. The packaged medical cannabis products are then sold by the Group under the Company’s brands to local Israeli pharmacies directly or through contracted distributors.
Canada
In Canada, our primary customers are provincially-owned cannabis wholesalers who in turn sell to private and public retail locations where the consumer ultimately purchases cannabis products.
The following table describes the Canadian facilities (the “Canadian Facilities”):
|
Facility
|
Location
|
Description
|
|
Manitou Facility
(“Manitou Facility”) |
Ontario
|
Flagship 32,050 square metre facility, with approximately 4,340 square metre of cultivation space, located in Kitchener, Ontario.
This facility is in the process of being closed as part of the Canadian Restructuring.
|
|
Trillium Facility
(“Trillium Facility”) |
Ontario
|
Approximately 1,400 square metre processing and cultivation facility, located in Kitchener, Ontario.
|
|
Highland Facility
|
Nova Scotia
|
Approximately 530 square metre cultivation and storage facility. Although cannabis cultivation has been halted at this facility, the facility continues to be utilized for packaging and
storage, located in Antigonish, Nova Scotia.
|
The Canadian Facilities are authorized to cultivate and process cannabis pursuant to their Health Canada-issued licenses (the “TJAC Licenses” and the “MYM License”, respectively), however, only the Trillium Facility and the Highland Facility hold licenses to sell cannabis on a non-B2B basis. In September 2022, TJAC, received authorization for the distribution and sale its high-quality cannabis products in the Quebec provincial adult-use market for a three-year term through the Société Québécoise du Cannabis, the governing body which oversees the sale
of recreational cannabis in the Province of Quebec.
In connection with the Canadian Restructuring, upon further review by the Trichome Group, it was determined that the Manitou Facility would wind down its operations. This is expected to occur
throughout Q4 2022.
13
Management’s Discussion and Analysis
Germany
The Company continues to expand its presence in the German market by forging partnerships with pharmacies and distributors across the country and developing Adjupharm and the Logistics Centre as
the Company’s European hub. Adjupharm sources its supply of medical cannabis for the German market from various EU-GMP certified European and Canadian suppliers. The Logistics Centre upgraded Adjupharm’s production technology and increased its
storage capacity to accommodate its anticipated growth.
Adjupharm currently holds wholesale, narcotics handling, manufacturing, procurement, storage, distribution, and import/export licenses granted to it by the
applicable German regulatory authorities (the “Adjupharm Licenses”).
Between our various geographies, the strategy for new products varies given that each market is at a different stage of development with respect to regulatory regimes, patient and customer
preferences and adoption rates.
Israel
The Company is strategically focused on importing premium and ultra-premium cannabis from Canada to supplement cannabis supplied by third-party Israeli cultivators. Canadian indoor-grown cannabis
is usually sold at a higher price point due to Israeli consumer preference for premium and ultra premium cannabis.

As part of the Top Shelf Collection, the Company launched the Diesel Drift and Lemon Rocket strains, with an additional strain, Tropicanna Gold, which is expected to be introduced in Q4 2022.

14
Management’s Discussion and Analysis
In Q3 2022, the Company expanded its Signature Collection with the introduction of two new varieties, Strawnana, an indoor-grown flower, and Sydney, the Company’s first high-CBD cannabis strain,
Sydney.

As part of its recent rebranding the Company expanded its Roma® product portfolio in Q3 2022 to include pre-rolls and an oils range,
offering the widest range of different product SKUs for a single strain in the Israeli market. This delivers a variety of formats of IMC’s most successful and well-known strain to Israeli medical cannabis patients. IMC’s Roma® strain is a
high-THC medical cannabis flower that offers a therapeutic continuum and is known for its strength and longevity of effect. In August 2022, 100 kilograms of Roma® were sold in Israel.
The WAGNERS brand launched in Israel in Q1 2022, with premium indoor-grown cannabis from the Canadian Facilities. The WAGNERS brand was the first international premium, indoor-grown brand
introduced to the Israel cannabis market, at a competitive price point.

15
Management’s Discussion and Analysis
KEY HIGHLIGHTS FOR THE QUARTER ENDED SEPTEMBER 30, 2022
In Q3 2022, the Company continued to integrate the strategic acquisitions completed in Q1 2022. Effective November 7, 2022, the Company began focusing its efforts and resources on growth in the
Israeli and German cannabis markets with a goal of reaching profitability in 2023, and commenced exiting the Canadian cannabis market. The Company’s key highlights and events for the three months ended September 30, 2022 include:
Biome Grow Inc. Default
On April 4, 2022, the Company issued a Notice of Event of Default and Acceleration (the “Notice of Default”) to Biome Grow Inc. (the “Guarantor”) and its subsidiary, Cultivator Catalyst Corp. (together with the Guarantor, the “Obligors”), for a total outstanding principal plus accrued and unpaid
interest of approximately $2,680 (the “Biome Loan”). The Company issued the Notice of Default after several failed attempts to engage the Obligors regarding an
extension and repayment of the Biome Loan.
On April 20, 2022, the Company issued a demand letter to the Obligors seeking immediate payment, along with a Notice to Enforce Security pursuant to section 244 of the Bankruptcy and Insolvency Act (Canada). On May 3, 2022, MYM filed an application with the Superior Court of Justice in Ontario (the “Superior Court”) to appoint a receiver to take control
of the Obligors’ assets, including the security, to effect repayment of the Biome Loan.
The Biome Loan and related security agreements were entered into in July 2020, approximately one year prior to the Company’s acquisition of MYM. As part of the Biome Loan, the Obligors agreed to
repay all outstanding principal and accrued and unpaid interest no later than January 31, 2022. The amount of the Biome Loan and interest payable is secured by assets held in escrow by the Obligors pursuant to a general security agreement (the “Collateral”).
On May 12, 2022, the Company applied to and received from the Superior Court an interim order to, among other things, freeze the assets of the Obligors including
the assets, which comprise MYM’s Collateral for the Biome Loan. MYM has applied to the Superior Court, which granted MYM’s request for the receivership of the assets of the Obligors and has scheduled an
in-person hearing for the receivership application on September 12, 2022.
In September 2022, MYM and the Obligors reached an agreement and signed a term sheet for the settlement of the receivership application and amendment to the Biome Loan (the “Biome Term Sheet”). The Biome Term Sheet was signed on September 9, 2022, prior to the September 12, 2022 in-person receivership application hearing with the Superior Court. The Superior Court approved the
adjournment of the receivership application, pending the implementation of the settlement outlined in the Biome Term Sheet, pursuant to which, the Biome Loan will continue to bear interest at a rate of 8% per annum on the principal balance of the
Biome Loan, compounding every four months on the aggregate balance of the outstanding principal balance plus all accrued and unpaid interest (the “Indebtedness”). The Biome Loan matures December 9, 2023
unless extended through mutual agreement by both parties.
Based on the Biome Term Sheet, the Obligors are required to make a payment to MYM on December 31, 2022. The value of the payment on December 31, 2022 will depend on the volume weighted average
price (the “VWAP”) of the Company’s common shares during the final ten trading days of November 2022. The repayment will be 5% or 10% of the total Indebtedness, depending on the VWAP over that period of
time.
16
Management’s Discussion and Analysis
On October 4, 2022, a loan amendment agreement (“Biome Settlement Agreement”) was executed in line with the terms noted in the Biome Term Sheet.
As a result of the Biome Settlement Agreement, the Biome Loan was considered extinguished under IFRS 9 Financial Instruments and a gain of $239 was
recognized. The Biome Loan is now recognized as a fair value through profit and loss financial instrument, instead of amortized cost. In addition to the gain on extinguishment, the Company recognized for
the nine-month period ended September 30, 2022, an expected credit loss of $1,803 and a fair value gain of $64.
NASDAQ Compliance Notice
On July 13, 2022, the Company received written notification from Nasdaq (the “Notification Letter”) that the closing bid price of the
Common Shares had fallen below US$1.00 per share over a period of 30 consecutive business days, with the result that the Company was not in compliance with the minimum bid price requirement (the “Minimum Share
Price Listing Standard”). The Notification Letter provided that the Company had 180 calendar days following receipt of such notice to regain compliance with the Minimum Share Price Listing Standard. The Company’s deadline for regaining
compliance with the Minimum Share Price Listing Standard is January 9, 2023 (the “Cure Deadline”). To regain compliance, the Common Shares must have a closing bid
price of at least US$1.00 for a minimum of ten consecutive business days. In the event that the Company does not regain compliance by the Cure Deadline, the Company may be eligible for additional time to regain compliance with the Minimum Share
Price Listing Standard. To qualify, the Company will be required to meet the continued listing requirement for market value of publicly held shares and all other initial listing standards for Nasdaq, with the exception of the Minimum Share Price
Listing Standard, and will need to provide written notice of its intention to cure the deficiency during the second compliance period, by effecting a reverse stock split, if necessary. If the Company meets these requirements, Nasdaq will inform
the Company that it has been granted an additional 180 calendar days. However, if it appears to Nasdaq that the Company will not be able to cure the deficiency, or if the Company is otherwise not eligible, Nasdaq will provide notice to the
Company that its Common Shares will be subject to delisting.
On October 20, 2022, the Company obtained shareholder approval at its annual and special meeting of shareholders (the “Meeting”) to
consolidate (or reverse split) the issued and outstanding Common Shares into a lesser number of issued and outstanding Common Shares (the “Consolidation”) on the
basis of one (1) post-Consolidation Common Share for a number of pre-Consolidation Common Shares within a range of between six (6) and ten (10) pre-Consolidation Common Shares, as and when determined by the board of directors (the “Board”) in its sole discretion Following such Consolidation (or reverse split), the Common Shares will continue to trade on the Nasdaq under the symbol “IMCC”. The Consolidation is expected to increase the
market price of our Common Shares in order to comply with the Minimum Share Price Listing Standard.
17
Management’s Discussion and Analysis
The Company’s common shares are also listed on the CSE and the Notification Letter does not affect the Company’s compliance status with such listing.
Canadian Restructuring
On August 5, 2022, the Company commenced the Canadian Restructuring as part of its disciplined approach to spending and implementing cost efficiencies. The Company entered into an agreement to
sell all of the issued and outstanding shares of Sublime on an “as is, where is” basis to a group of purchasers that included current and former members of the Sublime management team for aggregate proceeds of approximately $100 less working
capital adjustments, for a final net purchase price of $89 (the “Sublime Transaction”). The Sublime Transaction included the sale of Sublime’s lease obligation of the approximately
930 square metre cultivation and storage facility and Sublime’s related operations.
The Sublime Transaction constituted a “related party transaction” within the meaning of Multilateral Instrument 61-101 – Protection of Minority Security Holders
in Special Transactions (“MI 61-101”). Pursuant to Sections 5.5(a) and 5.7(1)(a) of MI 61-101, the transaction is exempt from the formal valuation and minority shareholder approval requirements of
such instrument. For more information, see “Transactions with Related Parties”.
Closing of First Tranche of Private Placement
On August 19, 2022, the Company announced a private placement for aggregate gross proceeds of up to US$5,000 (the “Private Placement”) led by the Company’s
management and executive team.
On August 24, 2022, the Company announced that it closed the first tranche of the Private Placement, consisting of 4,887,496 Common Shares at a price of US$0.50 per Common Share for aggregate
proceeds of approximately US$2,444 (the “First Tranche”). Certain insiders of the Company, including Oren Shuster, Chief Executive Officer (“CEO”) and Director and
Shai Shemesh, Chief Financial Officer (“CFO”), among others, subscribed for an aggregate of 1,563,496 Common Shares in the first tranche of the Private Placement for aggregate proceeds of US$782. For more
information on the Private Placement see “Corporate Highlights and Events – Subsequent Events”.
Changes to the Board
On September 13, 2022, the Company announced that Einat Zakariya and Moti Marcus were appointed to the Board. Einat Zakariya and Moti Marcus replaced Vivian Bercovici and Haleli Barath, who
resigned to pursue other opportunities.
Einat Zakariya is the current CEO and partner of LIV collection, a brand subsidiary of Ewave Holdings Ltd., and CEO and Partner of Ewave Nadlan International Investments Ltd. Ms. Zakariya has
proven expertise in the real-estate industry and brings vast experience in CEO roles as well as strategic consulting, marketing, advertising, and sales. She previously sat on the boards of several major organizations.
Moti Marcus is the current CEO of Packer Quality Materials, one of the largest companies in Israel for the sale and processing of special and unique metals. Mr. Marcus has a strong track record
in CFO roles, management, and mergers and acquisitions. He has served on the boards of several institutions and is a member of the Israel Ministry of Finance “Team of Select Directors.”
18
Management’s Discussion and Analysis
The Company and SNDL Inc. Export to Israel
On September 15, 2022, the Company and SNDL Inc. (“SNDL”) announced that SNDL completed its initial international export of approximately 167 kilograms of
premium dried flower from Canada to Israel as part of its total commitment with the Company. SNDL and the Company have agreed to the aggregate export of 1,000 kilograms of high-quality dried flower products for processing and distribution in the
Israeli medical cannabis market.
SUBSEQUENT EVENTS
Closing of Second Tranche of Private Placement
On October 5, 2022, the Company announced that it closed the second tranche of the Private Placement, consisting of 1,112,504 Common Shares at a price of US$0.50 per Common Share for aggregate
proceeds of approximately US$556 (the “Second Tranche”) and increasing the total amount raised from the Private Placement to US$3,000. Marc Lustig, Executive Chairman and Director of the Company, subscribed
for 1,112,504 Common Shares in the second tranche of the Private Placement for aggregate proceeds of approximately US$556.
The Company used the proceeds from the Private Placement for general working capital purposes.
The Private Placement was considered a “related party transaction” within the meaning of MI 61-101 as a result of the participation by certain insiders of the Company. The Company relied on
Sections 5.5(a) and 5.7(1)(a) of MI 61-101 for exemptions from the requirements to obtain a formal valuation and minority shareholder approval, respectively, because the fair market value of the aggregate insider participation in the Private
Placement was below 25% of the Company’s market capitalization for purposes of MI 61-101.
Launch of BLK MKT Brand in Israeli Medical Cannabis Market
On October 12, 2022, the Company and Avant Brands Inc. (“Avant”) announced the signing of an international trademark licensing agreement (the “Licensing Agreement”) granting the Company the exclusive right to launch the BLKMKT brand in the Israeli medical cannabis market. Under the terms of the Licensing Agreement, a subsidiary of Avant will license
the Company’s premium- cannabis flagship BLKMKT brand to an Israeli subsidiary of the Company for use on the Company’s medical cannabis product packaging. All such packaging will contain cannabis cultivated exclusively by Avant, and sold to the
Company’s affiliates. The integration of unique and exclusive varieties of the high-quality BLKMKT brand into the Company’s current premium product portfolio will serve to bolster the cooperative and synergistic partnership forged between the
Avant and the Company over the past two years. The Licensing Agreement signals IMC’s commitment to implementing a premium strategy and acts as another step to establish the Company’s leadership of the ultra-premium segment in Israel.
Annual General and Special Meeting
On October 20, 2022, the Company held the Meeting at which time all matters put to shareholders were approved including, but not limited to, the election of directors to the Board, the
appointment of Kost Forer Gabbay & Kasierer, a member of Ernst & Young Global as auditor of the Company, the adoption of new modernized articles of the Company, and the consolidation of all of the issued and outstanding Common Shares of
the Company at a ratio of between six (6) and ten (10) pre-consolidation Common Shares for every one (1) post-consolidation Common Share, as and when determined by the Board.
Trichome Group Commences CCAA Proceedings
On November 7, 2022, the Trichome Group filed for and obtained, from the Court an initial order (the “Initial Order”) pursuant to the CCAA. Pursuant to the
Initial Order, the Trichome Group obtained a broad stay of all proceedings (the “Stay”) against the members of the Trichome Group, and their assets, businesses and directors and officers that is effective
until November 17, 2022. The Stay may be extended subject to further orders of the Court.
19
Management’s Discussion and Analysis
The CCAA proceedings are solely in respect of the Trichome Group. As such, the Company’s assets and subsidiaries in Israel and Germany are not parties to the CCAA proceedings.
The CCAA proceedings will afford the Trichome Group the stability and flexibility required to restructure its business, including through a sale and investment solicitation process to be approved
by the Court.
The Trichome Group is expected to continue to operate its business for the time being however, it is expected that there will be changes in the Canadian operations consistent with those of a
company operating through CCAA proceedings, including employee and contract terminations. The Trichome Group has said that it intends to use the CCAA proceedings to implement a sale and investment solicitation process for the sale of its assets
or restructuring of its business.
In connection with the CCAA proceedings, the Credit Parties and the DIP Lender, entered into the DIP Agreement. Pursuant to the DIP Agreement, the DIP Lender has agreed to provide a
super-priority interim revolving credit facility to the Borrower. In accordance with the DIP Agreement, the DIP Facility is to be used during the CCAA proceedings by the Borrower to fund its working capital needs. The DIP Facility is subject to
customary covenants, conditions precedent, and representations and warranties made by the Credit Parties to the DIP Lender. The current DIP Lender's charge approved by Court is up to the maximum amount of $1,825, reflecting the anticipated
borrowings under the DIP Facility up to November 17, 2022.
2022 Share Consolidation
On November 14, 2022, the Board had approved the Consolidation on the basis of one (1) post-Consolidation Common Shares for ten (10) pre-Consolidation Common Shares, to be effected on or about
November 17, 2022.
20
Management’s Discussion and Analysis
FINANCIAL HIGHLIGHTS
Below is the analysis of the changes that occurred for the three and nine months ended September 30, 2022 with further commentary provided below.
|
For the nine months
ended September 30
|
For the three months
ended September 30
|
For the Year ended December 31 | ||||||||||||||||||
|
2022
|
2021
|
2022
|
2021
|
2021
|
||||||||||||||||
|
Net Revenues*
|
$
|
39,874
|
$
|
24,141
|
$
|
14,170
|
$
|
8,040
|
$
|
34,053
|
||||||||||
|
Gross profit before fair value impacts in cost of sales*
|
$
|
8,500
|
$
|
7,516
|
$
|
2,819
|
$
|
2,033
|
$
|
8,594
|
||||||||||
|
Gross margin before fair value impacts in cost of sales (%)*
|
21
|
%
|
31
|
%
|
20
|
%
|
25
|
%
|
25
|
%
|
||||||||||
|
Operating Loss*
|
$
|
(20,082
|
)
|
$
|
(14,294
|
)
|
$
|
(5,526
|
)
|
$
|
(8,124
|
)
|
$
|
(23,036
|
)
|
|||||
|
Profit (loss)*
|
$
|
(15,271
|
)
|
$
|
7,697
|
$
|
(4,532
|
)
|
$
|
830
|
$
|
(665
|
)
|
|||||||
|
Loss per share attributable to equity holders of the Company – Basic (in CAD)*
|
$
|
(0.19
|
)
|
$
|
0.19
|
$
|
(0.06
|
) |
$
|
0.03
|
$
|
0.00
|
||||||||
|
Loss per share attributable to equity holders of the Company - Diluted (in CAD)*
|
$
|
(0.26
|
)
|
$
|
(0.24
|
) |
$
|
(0.06
|
) |
$
|
(0.09
|
) |
$
|
(0.36
|
)
|
|||||
|
For the nine months
ended September 30
|
For the three months
ended September 30
|
For the Year ended December 31
|
||||||||||||||||||
|
2022
|
2021
|
2022
|
2021
|
2021
|
||||||||||||||||
|
Average net selling price of dried flower (per Gram)*
|
$
|
8.13
|
$
|
5.91
|
$
|
9.08
|
$
|
6.61
|
$
|
6.18
|
||||||||||
|
Quantity harvested and trimmed (in Kilograms)*2
|
-
|
988
|
-
|
451
|
1,935
|
|||||||||||||||
|
Quantity of dried flower sold (in Kilograms)*
|
4,460
|
3,058
|
1,453
|
1,175
|
4,278
|
|||||||||||||||
* From continuing operations
Notes:
| 1. |
Cannabis selling prices in the Canadian market are characterized by lower selling prices than dried flowers in the Israeli and German market.
|
| 2. |
Including other cannabis products such as Concentrates, Kief, Hash and Pre-rolls.
|
| 3. |
Harvested flowers, after trimming and ready for manufacturing.
|
21
Management’s Discussion and Analysis
The Overview of Financial Performance includes reference to “Gross Margin”, which is a non-IFRS financial measure that the Company defines as the difference between revenue and cost of revenues
divided by revenue (expressed as a percentage), prior to the effect of a fair value adjustment for inventory and biological assets. For more information on non-IFRS financial measures, see the “Non-IFRS Financial
Measures” and “Metrics and Non-IFRS Financial Measures” sections of the MD&A.
OPERATIONAL RESULTS
In each of the markets in which the Company operates, the Company must navigate evolving customer and patient trends in order for it to continue to be competitive with other suppliers of medical
cannabis products.
The Company believes that there are a number of key factors creating tailwinds to facilitate further industry growth. In Israel, the number of licensed medical patients continues to increase and
currently stands at 119,656 as of September 2022. This figure is expected to continue growing in the coming years and may further benefit from regulatory change liberalizing the cannabis market in Israel. Moreover, the acquisitions of the Israeli
Pharmacies positions IM Cannabis as a large distributor of medical cannabis in Israel. As the Israeli cannabis market has become increasingly competitive, the ability to import premium cannabis from Canada is a key determinant of the Company’s
success in Israel.
The German medical cannabis market has been slower to develop due to the difficulty in medical patients accessing prescriptions and insurance reimbursements. The Company has, however, seen an
increase in the number of patients paying out-of-pocket for medical cannabis products in Germany, which the Company believes is supportive of its business plan as it relies less on the need for patient’s insurance coverage for re-imbursement.
The newly elected coalition government in Germany has endorsed the legalization of adult-use cannabis. While no specific legislation has yet been tabled and any implementation is expected to take
time, the Company believes that Germany has the potential to be the second largest federally legal, adult-use market in the world.
The Company’s outlook in Germany is further supported by its focus on the cultivation and distribution of premium and ultra-premium cannabis products exclusively, which the Company believes to be
in the greatest demand in all of its markets. In comparison to other markets, the Company faces less competition in Germany and therefore is less likely to face significant price competition.
The Company is focusing its resources on reinforcing and further pursuing growth opportunities in Israel, Germany and Europe, implementing a leaner organization strategy with the primary focus on
achieving profitability in 2023.
As the Company refocus its strategy by discontinuing its operation in the Canadian market, it enables the Company to have also lower debt levels that were reduced by approximately 57% from $11.9
million to $5.1 million as of September 30th. Current liabilities are reduced by approximately 50% from $51.7 million to $25.6 million.
22
Management’s Discussion and Analysis
REVENUES AND GROSS MARGINS
REVENUES
The revenues of the Group are primarily generated from sales of medical cannabis products to customers in Israel and Germany as well as adult-use recreational cannabis products to customers in
Canada. The three reportable geographical segments in which the Company operates are Israel, Canada and Germany.
For the nine months ended September 30:
|
Israel
|
Canada – discontinued operations
|
Germany
|
Adjustments
|
Total
|
||||||||||||||||||||||||||||||||||||
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
|||||||||||||||||||||||||||||||
|
Revenues
|
$
|
37,364
|
$
|
16,959
|
$
|
27,272
|
$
|
10,131
|
$
|
2,510
|
$
|
7,182
|
$
|
-
|
$
|
-
|
$
|
67,146
|
$
|
34,272
|
||||||||||||||||||||
|
Inter-segment revenues
|
$
|
-
|
$
|
-
|
$
|
2,481
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
(2,481
|
)
|
$
|
-
|
$
|
-
|
$
|
-
|
|||||||||||||||||||
|
Total revenues
|
$
|
37,364
|
$
|
16,959
|
$
|
29,753
|
$
|
10,131
|
$
|
2,510
|
$
|
7,182
|
$
|
(2,481
|
)
|
$
|
-
|
$
|
67,146
|
$
|
34,272
|
|||||||||||||||||||
|
Segment income (loss)
|
$
|
(13,325
|
)
|
$
|
(6,228
|
)
|
$
|
(138,785
|
)
|
$
|
(12,373
|
)
|
$
|
(2,708
|
)
|
$
|
(2,404
|
)
|
$
|
-
|
$
|
-
|
$
|
(154,818
|
)
|
$
|
(21,005
|
)
|
||||||||||||
|
Unallocated corporate expenses
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
(4,049
|
)
|
$
|
(5,662
|
)
|
$
|
(4,049
|
)
|
$
|
(5,662
|
)
|
||||||||||||||||
|
Total operating (loss) income
|
$
|
(13,325
|
)
|
$
|
(6,228
|
)
|
$
|
(138,785
|
)
|
$
|
(12,373
|
)
|
$
|
(2,708
|
)
|
$
|
(2,404
|
)
|
$
|
(4,049
|
)
|
$
|
(5,662
|
)
|
$
|
(158,867
|
)
|
$
|
(26,667
|
)
|
||||||||||
|
Depreciation & amortization
|
$
|
1,790
|
$
|
1,037
|
$
|
3,926
|
$
|
2,501
|
$
|
152
|
$
|
66
|
$
|
-
|
$
|
-
|
$
|
5,868
|
$
|
3,604
|
||||||||||||||||||||
For the three months ended September 30:
|
Israel
|
Canada – discontinued operations
|
Germany
|
Adjustments
|
Total
|
||||||||||||||||||||||||||||||||||||
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
2022
|
2021
|
|||||||||||||||||||||||||||||||
|
Revenues
|
$
|
13,158
|
$
|
7,152
|
$
|
5,586
|
$
|
6,353
|
$
|
1,012
|
$
|
888
|
$
|
-
|
$
|
-
|
$
|
19,756
|
$
|
14,393
|
||||||||||||||||||||
|
Total revenues
|
$
|
13,158
|
$
|
7,152
|
$
|
5,586
|
$
|
6,353
|
$
|
1,012
|
$
|
888
|
$
|
-
|
$
|
-
|
$
|
19,756
|
$
|
14,393
|
||||||||||||||||||||
|
Segment income (loss)
|
$
|
(3,110
|
)
|
$
|
(5,707
|
)
|
$
|
(123,062
|
)
|
$
|
(6,121
|
)
|
$
|
(699
|
)
|
$
|
(1,278
|
)
|
$
|
-
|
$
|
-
|
$
|
(126,871
|
)
|
$
|
(13,106
|
)
|
||||||||||||
|
Unallocated corporate expenses
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
(1,717
|
)
|
$
|
(1,139
|
)
|
$
|
(1,717
|
)
|
$
|
(1,139
|
)
|
||||||||||||||||
|
Total operating (loss) income
|
$
|
(3,110
|
)
|
$
|
(5,707
|
)
|
$
|
(123,062
|
)
|
$
|
(6,121
|
)
|
$
|
(699
|
)
|
$
|
(1,278
|
)
|
$
|
(1,717
|
)
|
$
|
(1,139
|
)
|
$
|
(128,588
|
)
|
$
|
(14,245
|
)
|
||||||||||
|
Depreciation & amortization
|
$
|
474
|
$
|
490
|
$
|
1,182
|
$
|
1,445
|
$
|
152
|
$
|
26
|
$
|
-
|
$
|
-
|
$
|
1,808
|
$
|
1,961
|
||||||||||||||||||||
23
Management’s Discussion and Analysis
The consolidated revenues of the Group from continuing operations for the nine months ended September 30, 2022 were attributed to the sale of medical cannabis products in Israel and Germany.
| ● |
Revenues from continuing operations for the nine months ended September 30, 2022 and 2021 were $39,874 and $24,141, respectively, representing an increase of $15,733 or 65%. Revenues For the three
months ended September 30, 2022 and 2021 were $14,170 and $8,040, respectively, representing an increase of $6,130 or 76%. The increase in revenues is primarily attributed to the increase in the quantity of
medical cannabis products sold, as well as from the higher average selling price per gram the Company realized from its portfolio of premium branded cannabis products in Israel. Additional increases were derived from the Company’s
organic growth and related synergies in the areas where it operates.
|
| ● |
Revenues from the Israeli operation were attributed to the sale of medical cannabis through the Company’s agreement with Focus Medical and the consolidation of revenues from the Israeli Pharmacies.
|
| ● |
In Germany, Company revenues were attributed to the sale of medical cannabis through Adjupharm.
|
| ● |
Revenues from the discontinued Canadian operation include revenues from the sale of adult-use recreational cannabis in Canada through the acquisitions of TJAC and MYM.
|
| ● |
Total dried flower sold for the nine months ended September 30, 2022 was 4,460kg at an average selling price of $8.13 per gram compared to 3,058kg for the same period in 2021 at an average selling price of
$5.91 per gram, mainly attributable to the higher average selling price per gram the Company recognized through the acquisition of the Israeli Pharmacies. Total dried flower sold for the three months ended September 30, 2022 was
1,453kg at an average selling price of $9.08 per gram compared to 1,175kg for the three months ended September 30, 2021 at an average selling price of $6.61 per gram.
|
COST OF REVENUES
Cost of revenues is comprised of cultivation costs, purchase of materials and finished goods, utilities, salary expenses and import costs, including the purchase of raw materials, production,
product testing, shipping and sales related costs. At harvest, the biological assets are transferred to inventory at their fair value which becomes the deemed cost for the inventory. Inventory is later expensed to the cost of sales when sold.
Direct production costs are expensed through the cost of sales.
The fair value of biological assets is categorized within Level 3 of the fair value hierarchy. The inputs and assumptions used in determining the fair value of biological assets include:
| 1. |
Selling price per gram - calculated as the weighted average historical selling price for all strains of cannabis sold by the Group, which is expected to approximate future selling prices.
|
| 2. |
Post-harvest costs - calculated as the cost per gram of harvested cannabis to complete the sale of cannabis plants post-harvest, consisting of the cost of direct and indirect materials, depreciation and labor as well as labelling and
packaging costs.
|
| 3. |
Attrition rate - represents the weighted average percentage of biological assets which are expected to fail to mature into cannabis plants that can be harvested.
|
24
Management’s Discussion and Analysis
| 4. |
Average yield per plant - represents the expected number of grams of finished cannabis inventory which are expected to be obtained from each harvested cannabis plant.
|
| 5. |
Stage of growth - represents the weighted average number of weeks out of the average weeks growing cycle that biological assets have reached as of the measurement date. The growing cycle is
approximately 12 weeks.
|
The following table quantifies each significant unobservable input, and also provides the impact that a 10% increase/decrease in each input would have on the fair value of biological assets grown
by the Company:
|
10% change as of
|
||||||||||||||||
|
September 30, 2022
|
December 31, 2021
|
September 30, 2022
|
December 31, 2021
|
|||||||||||||
|
In CAD
|
In Thousands of CAD
|
|||||||||||||||
|
Average selling price per gram of dried cannabis
|
$
|
3.51
|
$
|
3.64
|
128
|
$
|
296
|
|||||||||
|
Average post-harvest costs per gram of dried cannabis
|
$
|
0.75
|
$
|
1.16
|
$
|
9
|
$
|
140
|
||||||||
|
Attrition rate
|
31
|
%
|
27
|
%
|
96
|
100
|
||||||||||
|
Average yield per plant (in grams)
|
40
|
47
|
91
|
228
|
||||||||||||
|
Average stage of growth
|
45
|
%
|
47
|
%
|
96
|
212
|
||||||||||
Note:
| 1. |
The cost of revenues from continuing operations for the nine months ended September 30, 2022 and 2021 were $31,374 and $16,625, respectively, representing an increase of $14,749 or 89%. Cost of revenues for the three months ended
September 30, 2022 and 2021 were $11,351 and $6,007, respectively, representing an increase of $5,344 or 89%.
|
GROSS PROFIT
The Company’s formula for calculating gross profit includes:
| ● |
production costs (current period costs that are directly attributable to the cannabis growing and harvesting process);
|
| ● |
materials and finished goods purchase costs;
|
| ● |
a fair value adjustment on sale of inventory (the change in fair value associated with biological assets that were transferred to inventory upon harvest); and
|
| ● |
a fair value adjustment on growth of biological assets (the estimated fair value less cost to sell of biological assets as at the reporting date).
|
Gross profit also includes the net change in fair value of biological assets, inventory expensed and production costs. Biological assets consist of cannabis plants at various after-harvest stages
which are recorded at fair value less costs to sell after harvest.
25
Management’s Discussion and Analysis
Gross profit from continuing operations for the nine months ended September 30, 2022 and 2021 was $6,559 and $5,354, respectively, representing an increase of $1,205 or 23%. For the three months
ended September 30, 2022 and 2021 gross profit (loss) was $1,953 and $(75), respectively, representing an increase of $2,028 or 2,704%.
Gross profit included gains (losses) from unrealized changes in fair value of biological assets and realized fair value adjustments on inventory sold of $(1,941) and $(2,162) for the nine months
ended September 30, 2022 and 2021, respectively. Losses from unrealized changes in fair value of biological assets and realized fair value adjustments on inventory sold for the three months ended September 30, 2022 and 2021 were $(866) and
$(2,108), respectively. Fair value adjustments were impacted primarily due to lower valuation to unrealized biological assets during the nine months ended September 30, 2022.
In the nine months ended September 30, 2022, the impact of global inflation on the Company resulted in higher than usual operating costs, and in particular higher costs of raw materials, shipping
and transport services and the cost of hiring skilled labor to ensure the Company remains on track with scheduled manufacturing and regulatory milestones. There is no assurance that inflation will not continue to have similar impacts on the
Company’s operations in Q3 and Q4 of 2022.
EXPENSES
GENERAL AND ADMINISTRATIVE
General and administrative expenses from continuing operations for the nine months ended September 30, 2022 and 2021 were $11,670 and $11,848, respectively, representing a decrease of $178 or 2%.
For the three months ended September 30, 2022 and 2021, general and administrative expenses were $4,315 and $3,653, respectively, representing an increase of $662 or 18%.
The increase in the general and administrative expense is mainly attributable to the growing corporate activities in Israel following the Company’s acquisitions in 2021. The expenses derived
mainly from professional services, legal fees and other consulting services. The general and administrative expenses are comprised mainly from salaries to employees in the amount of $3,191, professional fees in the amount of $3,979, depreciation
and amortization in the amount of $644 and insurance costs in the amount of $1,640.
On April 6, 2022, Focus Medical announced its decision, from March 30, 2022, to close the Focus Facility in Israel and therefore the Company recorded restructuring expenses related to impairment
of property, plant and equipment, biological assets and right of use asset and liabilities, in the total amount of $4,383.
In June 2022, the Company commenced the Canadian Restructuring, as part of its disciplined approach to spending and implementing cost efficiencies, aimed at saving approximately $1,000 in
quarterly cash expenses. As a result of the Canadian Restructuring, the Company recognized a restructuring expense of $121, related inventory write-offs of $192, impairment of tangible assets of $2,885, impairment of intangible asset of $1,581
and gain from selling the operation in the amount of $273. For more information, see “Strategy in Detail – Geographies and New Markets – Canada”.
26
Management’s Discussion and Analysis
SELLING AND MARKETING
Selling and marketing expenses from continuing operations for the nine months ended September 30, 2022 and 2021 were $8,379 and $3,845, respectively, representing an increase of $4,534 or 118%.
For the three months ended September 30, 2022, selling and marketing expenses were $2,797, compared to $1,709 for the three months ended September 30, 2021, representing an increase of $1,088 or 64%. The increase in the selling and marketing
expenses was due mainly to the Company’s increased marketing efforts in Israel, brand launch in Germany, and increased distribution expenses relating to the growth in sales and consolidation of selling and marketing expenses of entities acquired
in 2021. The increase in cost is also partially attributed to the rising costs of distribution, shipping and transport of the Company’s products.
SHARE-BASED COMPENSATION
Share-based compensation expense from continuing operations for the nine months ended September 30, 2022 and 2021 was $2,209 and $3,955, respectively, representing a decrease $1,746 or 44%. For
the three months ended September 30, 2022 and 2021, share-based compensation expense was $367 and $2,687, respectively, representing a decrease of $2,320 or 86%. The decrease for the nine months ended September 30, 2022 was mainly due to the
cancellation of incentive stock options (“Options”) held by employees who no longer worked for the Company.
FINANCING
Financing income (expense), net, from continuing operations for the nine months ended September 30, 2022 and 2021 was $3,782 and $22,197, respectively, representing a decrease of $18,415 or 83%.
For the three months ended September 30, 2022 and 2021, financing income (expense), net was $1,198 and $8,620, respectively, representing a decrease of $7,422 or 86%. The change for the period was mainly due to the updated valuation of the
Company’s warrants. This change includes the non-cash financial expense related to the financial instruments, affected by the Company’s decreased share price, in the amount of $5,892 and $(21,169), respectively.
NET INCOME/LOSS
Net income (loss) from continuing operations for the nine months ended September 30, 2022 and 2021 was $(15,271) and $7,697, respectively, representing a net loss increase of $22,968 or 298%. For
the three months ended September 30, 2022 and 2021, Net income (loss) was $(4,532) and $830 respectively, representing a net loss increase of $5,362 or 646%. The net loss increase related to factors impacting net income from operations described
above, and financing income driven by revaluation of warrants and other financial instruments in the amount of $5,892 which were recorded against liability on the grant day and were re-evaluated at September 30, 2022 through profit or loss.
NET INCOME (LOSS) PER SHARE BASIC AND DILUTED
Basic loss per share is calculated by dividing the net profit attributable to holders of Common Shares by the weighted average number of Common Shares outstanding during the period. Diluted
profit per Common Share is calculated by adjusting the earnings and number of Common Shares for the effects of dilutive warrants and other potentially dilutive securities. The weighted average number of Common Shares used as the denominator in
calculating diluted profit per Common Share excludes unissued Common Shares related to Options as they are antidilutive. Basic Income (Loss) per Common Share from continuing operations for the nine months ended September 30, 2022 and 2021 were
$(0.19) and $0.19 per Common Share, respectively. For the three months ended September 30, 2022 and 2021 were $(0.06) and $0.03, respectively.
27
Management’s Discussion and Analysis
Diluted Income (Loss) per Common Share from continuing operations for the nine months ended September 30, 2022 and 2021 were $(0.26) and $(0.24) per Common Share, respectively. Diluted Income
(Loss) per Common Share for the three months ended September 30, 2022 and 2021 were $(0.06) and $(0.09), respectively.
TOTAL ASSETS
Total assets as at September 30, 2022 were $129,066, compared to $287,388 as at December 31, 2021, representing a decrease of $158,322 or 55%. This decrease was primarily due to the goodwill
impairment of Trichome in the amount of $107,854, the closure of the Focus Facility and the Sublime Transaction leading to a depreciation of right-of-use assets and property plant and equipment in the amount of approximately $7,950. The
additional decrease is attributed to the revaluation of other receivables in the amount of approximately of $4,191 and also by the translation of items denominated in NIS in the Company’s balance sheet.
INTANGIBLE ASSETS
On March 18, 2021, the Trichome Transaction was completed whereby the Company acquired all of the issued and outstanding securities of Trichome for a total Common Share consideration valued at
approximately $99,028. Upon completion of the Trichome Transaction, the businesses of IM Cannabis and Trichome have been combined.
| ● |
Through the Trichome Transaction, the Company recognized goodwill of approximately $67,269 and intangible assets, primarily attributed to the cultivation license, worth approximately $6,458 (based on a
preliminary purchase price allocation). The goodwill arising on acquisition is attributed to the expected benefits from the synergies of the combination of the activities of the Company and Trichome, as well as value attributed to the
assembled workforce, which is included in goodwill. The goodwill recognized is not expected to be deductible for income tax purposes. The Canadian Restructuring and commencement of an exit from the Canadian market, which was announced on
November 7, 2022, resulted in indicators of impairment under IAS 36. These indicators of impairment led to an impairment analysis, in which it was concluded that a write-down was required. In Q3 2022, an impairment loss of $67,171 was
recorded for the goodwill initially recognized through the Trichome Transaction.
|
| ● |
The Company recognized the fair value of the assets acquired and liabilities assumed in the business combination according to a provisional measurement. The purchase consideration and the fair value of the
acquired assets and liabilities may be adjusted within 12 months from the acquisition date. At the date of final measurement, adjustments are generally made by restating comparative information previously determined provisionally. As of
the date of the Interim Financial Statements, a final valuation for the fair value of the identifiable assets acquired and liabilities assumed by an external valuation specialist had been obtained.
|
28
Management’s Discussion and Analysis
On July 9, 2021, the Company completed the MYM Transaction. As a result, the Company recognized goodwill of approximately $39,932 and intangible assets consisting of brand name and customer
relationships worth approximately $17,200 (based on a preliminary purchase price allocation study). The goodwill arising on acquisition is attributed to the expected benefits from the synergies of the combination of the activities of the Company
and MYM, as well as value attributed to the assembled workforce, which is included in goodwill. The goodwill recognized is not expected to be deductible for income tax purposes. As part of the closure of the Sublime Transaction the Company
recorded an impairment loss for the intangible assets in the amount of $1,581.
| ● |
The Company recognized the fair value of the assets acquired and liabilities assumed in the business combination according to a provisional measurement. The purchase consideration and the fair value of the
acquired assets and liabilities may be adjusted within 12 months from the acquisition date. At the date of final measurement, adjustments are generally made by restating comparative information previously determined provisionally. As of
the date of the Interim Financial Statements, a final valuation for the fair value of the identifiable assets acquired and liabilities assumed by an external valuation specialist had yet been obtained.
|
Furthermore, similar to the impairment loss recorded in Q3 2022 on the goodwill acquired via the Trichome Transaction, the Company also recorded an impairment loss of $40,592 on
the goodwill generated through the MYM Transaction. This too was a result of the Canadian Restructuring and expected exit of the Canadian market.
INVESTMENT IN XINTEZA
On December 26, 2019, IMC Holdings entered into a share purchase agreement with Xinteza API Ltd. (“Xinteza”), a company with a unique
biosynthesis technology, whereby the Company acquired, on an as-converted and fully diluted basis, 25.37% of Xinteza’s outstanding share capital, for consideration of US$1,700 (approximately $2,165 as of December 31, 2021) paid in several
installments (the “Xinteza SPA”). As of September 30, 2022, the Company has paid all outstanding installments pertaining to the Xinteza SPA and currently holds 23.35% of the outstanding share capital of
Xinteza on an as-converted and fully diluted basis. On February 24, 2022, IMC Holdings entered into a simple agreement for future equity with Xinteza, under which IMC Holdings paid US$100 (approximately $125), in exchange for right to certain
shares of Xinteza.
TOTAL LIABILITIES
Total liabilities as of September 30, 2022 were $73,094, compared to $82,443 at December 31, 2021, representing an decrease of $9,349 or 11%. The decrease was mainly due to a decrease of $3,446
in purchase consideration payable, $1,710 in accrued expenses and $5,906 in warrants, offset by an increase of $2,395 in bank loans and financial facilities and $5,820 in trade payables.
ASSETS HELD FOR SALE
Total assets held for sale as of September 30, 2022 were $60,618, compared to $nil at December 31, 2021, representing an increase of $60,618 or 100%. Assets held for sale derived from the
discontinued operations of the Trichome Group and represent all assets of the Trichome Group.
29
Management’s Discussion and Analysis
LIABILITY ASSOCIATED WITH ASSETS HELD FOR SALE
Total liability associated with assets held for sale as of September 30, 2022 were $43,613, compared to $nil at December 31, 2021, representing an increase of $43,613 or 100%. Liabilities
associated with assets held for sale derived from the discontinued operations of the Trichome Group and represent all liabilities of the Trichome Group.
LIQUIDITY AND CAPITAL RESOURCES
For the nine months ended September 30, 2022, the Company recorded revenues of $39,874. In addition, Company collected $335 in proceeds from the exercises of Options.
The Company can face liquidity fluctuations from time to time, resulting from delays in sales and slow inventory movements.
On May 14, 2021, the Company’s subsidiary, TJAC, entered into a revolving credit facility (the “Revolver”) for $5,000 with a private Canadian creditor (the
“Creditor”). The Revolver has an initial term of 12 months that can be extended upon the mutual agreement of both parties. Per annum interest is equal to the greater of (i) 9.75% and, (ii) the Toronto
Dominion Bank prime rate, plus 7.30%. The Revolver has a standby fee of 2.40% per annum, which is charged against the unused portion. Advanced amounts are secured against the assets of TJAC and Trichome, with Trichome providing a guarantee for
the Revolver. To maintain the Revolver, TJAC must abide by certain financial covenants, such as the maintenance of a tangible net worth greater than $5,000 and a debt service coverage ratio of 2:1. On
September 23, 2021, TJAC increased the limit on the Revolver from $5,000 to $7,500 and added Highland’s assets to the Revolver borrowing base. The increase will be used to finance TJAC and MYM’s receivables in order to manage the timing of cash
flows. On October 18, 2021, TJAC and MYM increased the limit on the Revolver to $10,000. The increase will be used to finance TJAC and MYM’s receivables in order to manage the timing of cash flows. On March 29, 2022, the limit on the Revolver
increased from $10,000 to $15,000 and was renewed for an additional 12 months. TJAC is in breach of certain terms of the Revolver, some of which have not been waived by the Creditor. On November 6, 2022, TJAC entered into an additional agreement
with the Creditor, the DIP Agreement, pursuant to which, the Creditor has agreed to provide a DIP Facility to TJAC. For more information regarding the DIP Agreement, see “Corporate
Events and Highlights – Subsequent Events”.
In January 2022, Focus Medical entered into a revolving credit facility with Bank Mizrahi (the “Mizrahi Facility”), which is guaranteed by certain Focus
assets. Advances from the Mizrahi Facility will be used for working capital needs. The Mizrahi Facility has a total commitment of up to NIS 15,000 (approximately $6,000) and has a one-year term for on-going needs and 6-month term for imports and
purchases needs. The Mizrahi Facility is renewable upon mutual agreement by the parties. The borrowing base available for draw at any time throughout the Mizrahi Facility and is subject to several covenants to be measured on a quarterly basis.
The Mizrahi Facility bears interest of Israeli prime interest plus 1.5% (approximately 4.25% as of July 2022) per annum.
On August 24, 2022, the Company announced that it closed the First Tranche of the Private Placement, consisting of 4,887,496 Common Shares at a price of US$0.50 per Common Share for aggregate
proceeds of approximately US$2,444.
30
Management’s Discussion and Analysis
On October 5, 2022, the Company announced that it closed the Second Tranche of the Private Placement, consisting of 1,112,504 Common Shares at a price of US$0.50 per Common Share for aggregate
proceeds of approximately US$556 and increasing the total amount raised from the Private Placement to US$3,000.
As of September 30, 2022, the Group's cash and cash equivalents totaled $3,223 and the Group's working capital deficit from continuing operations (current assets less current liabilities)
amounted to ($10,323). In the nine months ended September 30, 2022, the Group had an operating loss from continuing operation of ($20,082) and negative cash flows from continuing operating activities of ($9,361).
The Group’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outlays for operating expenses and capital expenditures,
including cost saving plans and restructuring actions taken in 2022. The Company’s board of directors approved a cost saving plan, implemented in whole or in part, to allow the Company to continue its operations and meet its cash obligations. The
cost saving plan consists of cost reduction due to efficiencies and synergies, which include mainly the following steps: discontinuing operation of loss-making activities, reduction in payroll and headcount, reduction in compensation paid to key
management personnel, operational efficiencies and reduced capital expenditures. The Company had experienced lack of timely execution of its cost saving plan, mainly in respect of the Trichome Group.
These conditions raise substantial doubt about the Company’s ability to continue as a going concern. The interim condensed consolidated financial statements do not include any adjustments
relating to the recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
As at September 30, 2022, the Group’s financial liabilities consisted of accounts payable which have contractual maturity dates within one year. The Group manages its liquidity risk by reviewing
its capital requirements on an ongoing basis. Based on the Group’s working capital position at September 30, 2022, management considers liquidity risk to be moderate.
As at September 30, 2022, the Group has identified the following liquidity risks related to financial liabilities (undiscounted):
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
> 10 years
|
|||||||||||||
|
Contractual Obligations
|
$
|
21,825
|
$
|
11,026
|
$
|
13,464
|
-
|
|||||||||
The maturity profile of the Company’s other financial liabilities (trade payables, other account payable and accrued expenses, and warrants) as of September 30, 2022 are less than one year.
|
Payments Due by Period
|
||||||||||||||||||||
|
Contractual Obligations
|
Total
|
Less than one year
|
1 to 3 years
|
4 to 5 years
|
After 5 years
|
|||||||||||||||
|
Debt
|
$
|
19,025
|
$
|
18,655
|
$
|
370
|
$
|
-
|
$
|
-
|
||||||||||
|
Finance Lease Obligations
|
$
|
27,185
|
$
|
3,065
|
$
|
5,619
|
$
|
5,037
|
$
|
13,464
|
||||||||||
|
Operating Leases
|
$
|
41
|
$
|
41
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||
|
Other Obligations
|
$
|
64
|
$
|
64
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||
|
Total Contractual Obligations
|
$
|
46,315
|
$
|
21,825
|
$
|
5,989
|
$
|
5,037
|
$
|
13,464
|
||||||||||
31
Management’s Discussion and Analysis
The Interim Financial Statements have been prepared on the basis of accounting principles applicable to a going concern, which assumes that the Company will continue in operation for the
foreseeable future and will be able to realize its assets and discharge its liabilities in the normal course of operations. The Interim Financial Statements do not include any adjustments to the amounts and classification of assets and
liabilities that would be necessary should the Company be unable to continue as a going concern. Such adjustments could be material.
SHARE CAPITAL
The Company’s authorized share capital consists of an unlimited number of Common Shares without par value, 75,695,325 of which were issued and outstanding as at the date hereof.
The Common Shares confer upon their holders the right to participate in the general meeting with each Common Share carrying the right to one vote on all matters. The Common Shares also allow
holders to receive dividends if and when declared and to participate in the distribution of surplus assets in the case of liquidation of the Company.
OTHER SECURITIES
As of September 30, 2022, the Company also has the following outstanding securities which are convertible into, or exercisable or exchangeable for, voting or equity securities of the Company:
4,631,631 Options, 550,000 restricted share units and 384,917 2019 Broker Compensation Options (as defined below), 3,043,478 2021 Offered Warrants (as defined below).
FINANCIAL BACKGROUND
On October 11, 2019, the Company completed the Reverse Takeover Transaction, effected by way of a “triangular merger” between the Company, IMC Holdings and a wholly-owned subsidiary of the
Company pursuant to Israeli statutory law.
In connection with the Reverse Takeover Transaction, the Company completed a private placement offering of 19,460,527 subscription receipts (each a “Subscription
Receipt”) on a pre-2021 Share Consolidation basis (as defined below) of a wholly-owned subsidiary of the Company at a price of $1.05 per Subscription Receipt for aggregate gross proceeds of $20,433. Upon completion of the Reverse
Takeover Transaction, each Subscription Receipt was exchanged for one unit comprised of one (1) common share and one-half of one (1/2) warrant (each whole warrant, a “2019 Listed Warrant”). Each 2019 Listed
Warrant was exercisable for one Common Share at an exercise price of $1.30 until October 11, 2021. A total of 9,730,258 2019 Listed Warrants were issued and listed for trading on the CSE under the ticker “IMCC.WT”. The 2019 Listed Warrants
expired on October 11, 2021.
32
Management’s Discussion and Analysis
The Company also issued to the agent who acted on its behalf in connection with the Reverse Takeover Transaction, a total of 1,199,326 2019 Broker Compensation Options (the “2019 Broker Compensation Options”). Following the 2021 Share Consolidation, the 2019 Broker Compensation Options were adjusted to require four 2019 Broker Compensation Options to be exercised for one underlying
unit at an adjusted exercise price of $4.20, with each unit exercisable into one Common Share and one-half of one Common Share purchase warrant (the “2019 Unlisted Warrants”). Following the 2021 Share
Consolidation, the 2019 Unlisted Warrants were adjusted to require four 2019 Unlisted Warrants to be exercised for one Common Share at an adjusted exercise price of $5.20. The 2019 Broker Compensation Options and the 2019 Unlisted Warrants have
now expired.
On February 12, 2021, IM Cannabis consolidated all of its issued and outstanding Common Shares on the basis of one (1) post-consolidation Common Share for each four (4) pre-consolidation Common
Shares (the “2021 Share Consolidation”) to meet the Minimum Share Price Listing Standard.
On May 7, 2021, the Company completed an offering (the “2021 Offering”) for a total of 6,086,956 Common Shares and 3,043,478 Common Share purchase warrants
(the “2021 Offered Warrants”). Each 2021 Offered Warrant is exercisable for one (1) Common Share at an exercise price of US$7.20 for a term of 5 years from the date of closing of the 2021 Offering.
The Company also issued a total of 182,609 broker compensation options (the “2021 Broker Compensation Options”) to the agents who acted on its behalf in
connection with the 2021 Offering. Each 2021 Broker Compensation Option is exercisable for one (1) Common Share at an exercise price of US$6.61, at any time following November 5, 2021 until November 5, 2024. There are 182,609 2021 Broker
Compensation Options outstanding.
For the nine months ended September 30, 2022 and 2021, the Company recognized a revaluation gain (loss) of $nil and $(74), respectively. For the three months ended September 30, 2022 and 2021,
the Company recognized a revaluation gain (loss) of $nil and $3,154 in the consolidated statement of profit or loss and other comprehensive income, in which the unrealized gain is included in finance income (expense).
As of September 30, 2022, and 2021, there were 3,043,478 and 3,043,478 2021 Offered Warrants outstanding, respectively, re-measured by the Company, using the Black-Scholes pricing model, in the
amount of $116 and $6,451, respectively. For the three and nine months ended September 30, 2022 and 2021, the Company recognized the revaluation gain (loss) in the consolidated statement of profit or loss and other comprehensive income, in which
the unrealized gain is included in finance income (expense).
33
Management’s Discussion and Analysis
OPERATING, FINANCING AND INVESTING ACTIVITIES
The following table highlights the Company’s cash flow activities for the three and nine months ended September 30, 2022 and 2021 and year ended December 31, 2021:
|
For the nine months
ended September 30,
|
For the three months
ended September 30,
|
For the year ended
December 31,
|
||||||||||||||||||
|
Net cash provided by (used in):
|
2022
|
2021
|
2022
|
2021
|
2021
|
|||||||||||||||
|
Operating activities
|
$
|
(10,123
|
)
|
$
|
(39,134
|
)
|
$
|
807
|
$
|
(16,123
|
)
|
$
|
(34,372
|
)
|
||||||
|
Investing activities
|
$
|
(826
|
)
|
$
|
(1,930
|
)
|
$
|
(187
|
)
|
$
|
(6,001
|
)
|
$
|
(9,012
|
)
|
|||||
|
Financing activities
|
$
|
2,908
|
$
|
45,937
|
$
|
(4,213
|
)
|
$
|
2,416
|
$
|
48,731
|
|||||||||
|
Effect of foreign exchange
|
$
|
(1,879
|
)
|
$
|
3,358
|
$
|
1,715
|
$
|
2,774
|
$
|
(329
|
)
|
||||||||
|
Increase (Decrease) in cash
|
$
|
(9,920
|
)
|
$
|
8,231
|
$
|
(1,878
|
)
|
$
|
(16,934
|
)
|
$
|
5,018
|
|||||||
Operating activities used cash of $10,123 and $39,134 for the nine months ended September 30, 2022 and 2021, respectively. For the three months ended September 30, 2022 and 2021, operating
activities used cash of $(807) and $16,123, respectively. This variance is primarily due to increase in the business activities of the Company including corporate expenses for salaries, professional fees and marketing expenses in Israel, Germany
and Canada. In the nine months ended September 30, 2022, cash was predominantly used to expand the Company’s Israeli operations and support its Canadian operation.
Investing activities used cash of $826 and $1,930 for the nine months ended September 30, 2022 and 2021, respectively. For the three months ended September 30, 2022 and 2021, investing activities
used cash of $187 and $6,001, respectively. Cash was used to enhance production through the purchase of equipment mainly for Highland and TJAC in the amount of $1,076 and was offset by repayment of loan receivable in the amount of $350.
Financing activities used cash of $2,908 and $45,937 for the nine months ended September 30, 2022 and 2021, respectively. For the three months ended September 30, 2022 and 2021, financing
activities used cash of $(4,213) and $2,416, respectively. Most of the cash was derived from receipt of loans in the amount of $2,510 as well as from the first tranche of the financing in the amount of USD$2,444 during the nine months ended
September 30, 2022, offset by payment of lease in the amount of $2,337.
34
Management’s Discussion and Analysis
SELECTED ANNUAL INFORMATION – CONTINUED AND DISCONTINUED OPERATIONS
|
For the year ended
|
December 31, 2021
|
December 31, 2020
|
||||||
|
Revenues
|
$
|
54,300
|
$
|
15,890
|
||||
|
Net Loss
|
$
|
(18,518
|
)
|
$
|
(28,734
|
)
|
||
|
Basic net income (Loss) per share:
|
$
|
(0.31
|
)
|
$
|
(0.74
|
)
|
||
|
Diluted net income (Loss) per share:
|
$
|
(0.66
|
)
|
$
|
(0.74
|
)
|
||
|
Total assets
|
$
|
287,388
|
$
|
38,116
|
||||
|
Total non-current liabilities
|
$
|
31,216
|
$
|
19,237
|
||||
SUMMARY OF QUARTERLY RESULTS
|
For the three months ended
|
September 30, 2022
|
June 30, 2022
|
March 31, 2022
|
December 31, 2021
|
||||||||||||
|
Revenues
|
$
|
14,170
|
$
|
12,703
|
$
|
13,001
|
$
|
9,912
|
||||||||
|
Net income (Loss)
|
$
|
(4,532
|
)
|
$
|
(3,736
|
)
|
$
|
(7,081
|
)
|
$
|
(8,363
|
)
|
||||
|
Basic net income (Loss) per share:
|
$
|
(0.06
|
)
|
$
|
(0.27
|
)
|
$
|
(0.14
|
)
|
$
|
(0.19
|
)
|
||||
|
Diluted net loss per share:
|
$
|
(0.06
|
)
|
$
|
(0.30
|
)
|
$
|
(0.17
|
)
|
$
|
(0.19
|
)
|
||||
|
For the three months ended
|
September 30, 2021
|
June 30, 2021
|
March 31, 2021
|
December 31, 2020
|
||||||||||||
|
Revenues
|
$
|
8,040
|
$
|
8,591
|
$
|
7,511
|
$
|
4,900
|
||||||||
|
Net income (Loss)
|
$
|
828
|
$
|
1,332
|
$
|
5,536
|
$
|
(19,976
|
)
|
|||||||
|
Basic net income (Loss) per share:
|
$
|
(0.06
|
)
|
$
|
(0.10
|
)
|
$
|
0.11
|
$
|
(0.5
|
)
|
|||||
|
Diluted net income (Loss) per share:
|
$
|
(0.06
|
)
|
$
|
(0.23
|
)
|
$
|
(0.06
|
)
|
$
|
(0.5
|
)
|
||||
The Company has consistently increased revenues on a quarterly basis as a result of the Group’s acquisition strategy and its organic growth. While revenues increased, net income (loss) was
effected by the Company’s rapid growth which included acquisitions fees, integration costs, costs related to the Company’s NASDAQ listing in Q1 2021 and fees related to the 2021 Offering.
INTERIM MD&A – QUARTERLY HIGHLIGHTS
The Company is focused in selling premium and ultra-premium cannabis products based on its end-costumers’ preferences. The products sales are based on the ability of the Company to import and
purchase high quality indoor grown cannabis products from its third- party suppliers and partners; the Company’s efforts to have continuous supply of premium cannabis products is also dependent on controls and regulations of the authorities as
well as other business and operational factors in the origin countries and local activity. The Company has engaged with several suppliers to purchase its premium and ultra premium products and continues to enhance organically its sales in the
Israeli and German market.
35
Management’s Discussion and Analysis
METRICS AND NON-IFRS FINANCIAL MEASURES
This MD&A makes reference to certain non-IFRS financial measures including “Gross Margin”, “EBITDA”, and “Adjusted EBITDA”. These measures are not recognized measures under IFRS and do not
have a standardized meaning prescribed by IFRS and are therefore unlikely to be comparable to similar measures presented by other companies. Rather, these measures are provided as additional information to complement those IFRS measures by
providing further understanding of our results of operations from management’s perspective. Accordingly, these measures should neither be considered in isolation nor as a substitute for analysis of our financial information reported under IFRS.
Management defines Gross Margin as the difference between revenue and cost of goods sold divided by revenue (expressed as a percentage), prior to the effect of a fair value adjustment for
inventory and biological assets. Management defines EBITDA as income earned or lost from operations, as reported, before interest, tax, depreciation and amortization. Adjusted EBITDA is defined as EBITDA, adjusted by removing other non-recurring
or non-cash items, including the unrealized change in fair value of biological assets, realized fair value adjustments on inventory sold in the period, share-based compensation expenses, and revaluation adjustments of financial assets and
liabilities measured on a fair value basis. Management believes that Adjusted EBITDA is a useful financial metric to assess its operating performance on a cash adjusted basis before the impact of non-recurring or non-cash items.
The non-IFRS financial measures can provide investors with supplemental measures of our operating performance and thus highlight trends in our core business that may not otherwise be apparent
when relying solely on IFRS measures. We also believe that securities analysts, investors and other interested parties frequently use non-IFRS financial measures in the evaluation of issuers. Our management also uses these non-IFRS financial
measures in order to facilitate operating performance comparisons from period to period, to prepare annual operating budgets and forecasts and to determine components of management compensation. As required by Canadian securities laws, we
reconcile these non-IFRS financial measures to the most comparable IFRS measures.
GROSS MARGIN
|
Nine months ended
|
September 30, 2022
|
September 30, 2021
|
||||||
|
Net Revenue
|
$
|
39,874
|
$
|
24,141
|
||||
|
Cost of sales
|
$
|
31,374
|
$
|
16,625
|
||||
|
Gross profit before FV adjustments
|
$
|
8,500
|
$
|
7,516
|
||||
|
Gross margin before FV adjustments
|
21
|
%
|
31
|
%
|
||||
36
Management’s Discussion and Analysis
EBITDA AND ADJUSTED EBITDA FROM CONTINUING OPERATIONS
|
For the nine months
ended September 30,
|
For the three months
ended September 30,
|
For the Year ended
December 31,
|
||||||||||||||||||
|
2022
|
2021
|
2022
|
2021
|
2021
|
||||||||||||||||
|
Operating Loss
|
$
|
(20,082
|
)
|
$
|
(14,294
|
)
|
$
|
(5,526
|
)
|
$
|
(8,124
|
)
|
$
|
(34,053
|
)
|
|||||
|
Depreciation & Amortization
|
$
|
1,942
|
$
|
1,103
|
$
|
626
|
$
|
516
|
$
|
2,125
|
||||||||||
|
EBITDA
|
$
|
(18,140
|
)
|
$
|
(13,191
|
)
|
$
|
(4,900
|
)
|
$
|
(7,608
|
)
|
$
|
(31,928
|
)
|
|||||
|
IFRS Biological assets fair value adjustments, net
|
$
|
1,941
|
$
|
2,086
|
$
|
867
|
$
|
2,032
|
$
|
2,263
|
||||||||||
|
Share-based payments
|
$
|
2,209
|
$
|
3,955
|
$
|
367
|
$
|
2,687
|
$
|
5,442
|
||||||||||
|
Costs related to the NASDAQ listing
|
$
|
-
|
$
|
1,261
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||
|
Restructuring cost
|
$
|
4,383
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||
|
Other non-recurring costs
|
$
|
-
|
$
|
570
|
$
|
-
|
$
|
570
|
$
|
-
|
||||||||||
|
Adjusted EBITDA (Non-IFRS)
|
$
|
(9,607
|
)
|
$
|
(5,319
|
)
|
$
|
(3,666
|
)
|
$
|
(2,319
|
)
|
$
|
(24,223
|
)
|
|||||
The Company’s Adjusted EBITDA for the nine months ended September 30, 2022 decreased primarily due to the lower gross margins as well as an increase in the general and administrative costs mainly
attributable to the growing corporate activities in Israel, and Germany, professional services derived from legal fees and other consulting services, salaries to employees and increased insurance costs upon listing on NASDAQ.
CONTINGENT LIABILITIES AND COMMITMENTS
RENTAL LIABILITIES
The table below summarizes the maturity profile of the Group’s lease liabilities based on contractual undiscounted payments (including interest payments):
September 30, 2022:
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
>10 years
|
|||||||||||||
|
Lease liabilities
|
$
|
3,065
|
$
|
10,656
|
$
|
13,464
|
-
|
|||||||||
September 30, 2021:
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
>10 years
|
|||||||||||||
|
Lease liabilities
|
$
|
3,068
|
$
|
11,844
|
$
|
15,379
|
-
|
|||||||||
The maturity profile of the Company’s other financial liabilities with liquidity risk (trade payables, other account payable and accrued expenses) as of September 30, 2022 and 2021, are less than
one year.
37
Management’s Discussion and Analysis
LITIGATION AND REGULATORY PROCEEDINGS
CLASS ACTION T.Z. 35676-08-19 TEL AVIV - JAFFA DISTRICT COURTS
On August 19, 2019, a cannabis consumer (the “Applicant”) filed a motion
for approval of a class action to Tel Aviv - Jaffa District Court (the “Motion”) against 17 companies (the “Parties”) operating in the field of medical cannabis in Israel, including Focus. The Applicant’s argument is that the Parties did not accurately mark the concentration of active ingredients
in their products. The personal suit sum for each class member stands at NIS 15,585 and the total amount of the class action suit is estimated at NIS 685,740,000. On June 2, 2020, the Parties submitted their response to the Motion. The Parties
argue in their response that the threshold conditions for approval of a class action were not met, since there is no reasonable possibility that the causes of action in the Motion will be decided in favor of the class group. On July 3, 2020 the
Applicant submitted his response to the Parties’ response. On July 5, 2020 the Applicant was absent from the hearing. As a result, on July 23, 2020 the Parties filed an application for a ruling of expenses which received a response from the
Applicant on August 12, 2020, asking to decline this request. On September 29, 2020 the court ruled that the Applicant would pay the Parties’ expenses amount of NIS 750. On July 14, 2021 a prehearing was held. The court recommended the parties
negotiate independently to avoid litigation, and if negotiations fail, then to begin mediation proceedings. The parties agreed to follow the court’s recommendations. On November 3, 2021 the court ruled the Parties will file an update regarding
the mediation procedure in 30 days. The parties conducted unsuccessful negotiations. On March 14, 2022, the Applicant filed a request to amend the Motion (the “Applicant’s Request for
Amendment”) and the judge disqualified herself from hearing the case. As a result, the case was redirected. On June 21, 2022 the Parties filed a response to the Applicant’s Request for Amendment. On
September 12, 2022, the court ruled on the Applicant’s Request for Amendment and accepted the Applicant’s request to clarify its claims regarding product labeling, while rejecting the Applicant’s other
requests. In addition, ruled that the Applicant will file an amended Motion (the “Amended Motion”) by November 24, 2022, and the Parties response will be submitted until January 24, 2023. A preliminary hearing is scheduled for March 23, 2023.
Due to the current preliminary state of the litigation process and based on the opinion of legal counsel to Focus, the Company’s management believes that it is not reasonably possible to assess
the outcome of the proceeding. Therefore, no provision has been recorded in respect thereof.
SUPREME COURT OF JUSTICE 2335/19 AND 8249/2
On March 2019 a petition was filed to the Supreme Court of Israel by the Medical Cannabis Association against the Israeli Ministry of Health (“MOH”)
regarding the new regulatory framework of the cannabis market following the government decision 1587 (the (the “Reform” and “Petition”, respectively). Subsequently,
additional 10 respondents joined the Petition concerning the Reform.
The Reform discusses several issues related to the regulating of the cannabis market. One of the main issues for which the Petition was filed was related to cannabis
prices. Until September 2019, under the previous regime, patients licensed for consumption of medical cannabis products by the IMCA received all of their medical cannabis products authorized under their respective licenses at a fixed
monthly price, regardless of each patient’s authorized amount. Following the Reform, licenses to patients were no longer entitling them to such fixed monthly price. In the Petition, the following were requested from
the court:
| ● |
that the MOH immediately suspend the implementation of the new regulation that harms, disproportionally, the medical cannabis patients;
|
38
Management’s Discussion and Analysis
| ● |
that the implementation of the new regulation, as is, would cause violation of constitutional rights of the medical cannabis patients; and
|
| ● |
that the MOH amends the flaws of the new regulation, prior to becoming effective, and to establish new regulations regarding labeling and use of pesticides.
|
The decision provided for an interim injunction, extending the validity of patient licenses until the earlier of March 31, 2020 or 10 days after the date the MOH reaches a
conclusion regarding the price control of medical cannabis products.
According to the decision, Focus Medical was attached to the proceedings as a respondent. Accordingly, Focus Medical filed its response to the petition on November
12, 2019. On March 8, 2020, the court decided to extend the validity of the interim injunction, so that the medical cannabis use licenses, which were extended under the decision, would continue to be valid until May 15, 2020, or 10 days after
the price committee’s decision on the matter before it, whichever comes first, subject to another court decision. The court also decided that if a further extension of the period of the interim injunction is granted beyond May 15, 2020, to the
extent required, it would be subject to medical surveillance by the attending physician, the details of which were to be included in the patient’s existing use license.
In light of several applications by the respondent represented by the state attorney’s office, for extension to file updated notice to the court, the interim injunction was
extended on July 30, 2020, until and subject to other decision of the court.
On October 29, 2020, the respondents represented by the state attorney’s office filed an update notice stating that the appeals committee unanimously decided against imposing
price controls on medical cannabis products and that the prices committee would hold a follow-up hearing in four months. The respondents also requested to update the court again in two months.
On November 25, 2020, the petitioner submitted their response to the respondents’ update.
On March 25, 2021, the respondents represented by the State Attorney’s Office filed an updating notice stating that the Prices Committee had come to a decision against imposing
price controls on medical cannabis products. However, the Prices Committee announced that it will issue an RFI to the corporations engaged in the medical cannabis market and assess the market every six months. Following the aforementioned, the
respondents represented by the State Attorney’s Office believe that the appeal should be rejected and the interim injunction should be canceled. On April 13, 2021, three of the respondents filed a response to the court, requesting to reject the
appeal and to cancel the interim injunction.
On April 25, 2021, the petitioner filed a response to the update notice from March 25, 2021, objecting to the position of the respondents represented by the State
Attorney’s Office, requesting the court to resolve as requested in the petition and grant the requested remedies to the petitioner. On July 6, 2021, the petitioner filed an urgent request to the court, to issue orders to the respondents
represented by the State Attorney’s Office, to request information from corporations engaged in the medical cannabis market in order to continue the examination of the market, according to the Prices Committee’s announcement mentioned above,
and requested the court reschedule the September 19, 2021 hearing date to an earlier date. The petitioner’s request was rejected by the court on July 7, 2021, and on September 19, 2021, a hearing was held. On November 16, 2021 the court ruled
the motion will delete, and the interim injunction will be cancelled in 10 days. Following a request submitted by the petitioner, on November 15, 2021 the court determined the interim injunction will extend until March 1, 2022. Additional
requests submitted for an extension of the interim order were denied. Following the aforementioned, the petition was denied.
39
Management’s Discussion and Analysis
On December 1, 2021 the medical cannabis association filed a motion to supreme court of justice of Israel for further hearing regarding the court ruling on 2335/19 as detailed
above. The petitioner also submitted a request for an exemption from the obligation to pay a fee or provide deposit a deposit. On February 9, 2022 the petitioner submitted an urgent request for a ruling by the court as well as a request to extend
the validity of the interim injunction, for at least three additional months. On February 24, 2022 the court overruled both the request for a further hearing in the petition, as well as the request to extend the validity of the interim
injunction. The motion was denied. The implication of the court's decision is that the changes in the regulation as mentioned in the Reform will continue to apply and the new pricing of the cannabis products will be in accordance with free market
principles.
PLANNING AND CONSTRUCTION 66813-06-21 BEER SHEVA MAGISTRATE COURT
On July 11, 2021 the Company was informed that on June 30, 2021, a claim was filed to Beer Sheva Magistrate Court, by the municipal committee presiding over planning and
construction in southern Israel against Focus, Focus’ directors and officers, including Oren Shuster and Rafael Gabay, and certain landowners, claiming for inadequate permitting for construction relating to the Focus Facility (the “Construction Proceedings”).
On December 6, 2021 the defendants filed a motion request for dismissal the indictment on the ground of defense of justice. The municipal committee filed its response and after
that the defendants filed a response to the municipal committee’s response. As of the date of this letter no decision has yet been made on the application.
A hearing was initially set to December 1, 2021 but postponed to June 13, 2022 and further postponed to September 29, 2022 and further postponed to January 8, 2023,
in order to allow the parties to negotiate towards a resolution. Further to court’s decision to postpone the initial hearing to January 8, 2023, the court stated that if the parties do not reach an
agreement by December 18, 2022, a decision will be made on the motion request for dismissal filed by Focus. These days the parties are in the final stages of closing a compromise outline, the outline is subject the state attorney office and the
court approvals.
At this stage, based on the opinion of Focus’ legal counsel, Company management cannot assess the chances of the claim advancing or the potential outcome of the Construction
Proceedings. Therefore, no provision has been recorded in respect thereof.
COVID-19 TEST KITS CLAIM, DISTRICT COURT OF STUTTGART
On November 19, 2021, Adjupharm filed a statement of claim (the “Claim”) to the District Court of Stuttgart (the “Stuttgart Court”) against Stroakmont & Atton Trading GmbH (“Stroakmont & Atton”), its shareholders and managing directors regarding a debt owed by Stroakmont & Atton to
Adjupharm in an amount of approximately EUR 947,563 for COVID-19 test kits purchased by Stroakmont & Atton from Adjupharm in May 2021. The Claim was accepted on December 2, 2021. In January 2022, Stroakmont & Atton filed its statement of
defence to the Stuttgart Court in which they essentially stated two main arguments for their defense:
| 1. |
that the contractual partner of the Company is not the defendant, Stroakmont & Atton is not the real purchaser rather a company named Uniclaro GmbH.
|
40
Management’s Discussion and Analysis
| 2. |
that the Company allegedly placed an order with Uniclaro GmbH for a total of 4.3 million Clongene COVID-19 tests, of which Uniclaro GmbH claims to have a payment claim against the Company for a partial delivery
of 380,400 Clongene COVID-19 tests in the total amount of EUR 941,897.20. Uniclaro GmbH has assigned this alleged claim against the Company to Stroakmont & Atton Trading GmbH, and Stroakmont & Atton Trading GmbH has precautionary
declared a set-off against the Company’s claim.
|
On March 22, 2022 Adjupharm filed a response to Stroakmont & Atton’s statement of defence and rejected both allegations with a variety of legal arguments and facts and also offered evidence
to the contrary in the form of testimony from the witnesses in question.
The burden of proof for both allegations lies with the opponents and they offered evidences to the court in the form of testimony from certain witnesses. If the opponents succeed in proving both
allegations to the court, the chances of winning the lawsuit will be considerably reduced. However, it will not be easy for the opponents to present evidence of these allegations.
On May 27, 2022, the conciliation hearing and main hearing were held. The Stuttgart Court ruled that the Company shall submit another writ by August 29, 2022. The Stuttgart Court also scheduled a
pronouncement date for September 7, 2022, when the Stuttgart Court will enter a judgement or hold an evidentiary hearing with witnesses. Following the pronouncement date on September 7, 2022 an evidentiary hearing with witnesses is scheduled for
January 11, 2023.
At this stage, the Company management cannot assess the chances of the claim advancing or the potential outcome of this these proceedings.
INITIATION OF PROCEEDINGS FOR LOAN REPAYMENT
On April 4, 2022, MYM issued a Notice of Default and on April 20, 2022, issued a Notice of Intent to Enforce Security pursuant to section 22 of the Bankruptcy and Insolvency Act (Canada) for the outstanding Biome Loan in the amount of $2.680, including accrued and unpaid
interest, owing by the Obligors. MYM has applied to the Superior Court to appoint a receiver to take control of the Obligors’ assets, including MYM’s security that is held in escrow, to effect repayment of the Biome Loan.
On May 12, 2022 the Company applied to and received from the Superior Court an interim order to, among other things, freeze the assets of the Obligors
including the assets which comprise MYM’s Collateral for the Biome Loan. MYM has applied to the Superior Court, which granted MYM’s request for the receivership of the assets of the Obligors and has scheduled
an in-person hearing for the receivership application on September 12, 2022.
In September 2022, MYM and the Obligors reached an agreement and signed the Biome Term Sheet on September 9, 2022, prior to the September 12, 2022 in-person receivership application hearing with
the Superior Court. The Superior Court approved the adjournment of the receivership application, pending the implementation of the settlement outlined in the Biome Term Sheet, pursuant to which, the Biome Loan will continue to bear interest at a
rate of 8% per annum on the principal balance of the Biome Loan, compounding every four months on the aggregate balance of the outstanding “Indebtedness”. The Biome Loan matures December 9, 2023 unless extended through mutual agreement by both
parties.
41
Management’s Discussion and Analysis
Based on the Biome Term Sheet, the Obligors are required to make a payment to MYM on December 31, 2022. The value of the payment on December 31, 2022 will depend on the VWAP of the Company’s
common shares during the final ten trading days of November 2022. The repayment will be 5% or 10% of the total Indebtedness, depending on the VWAP over that period of time.
On October 4, 2022, the Biome Settlement Agreement was executed in line with the terms noted in the Biome Term Sheet.
PROCEEDINGS UNDER CCAA
On November 7, 2022, the Trichome Group filed for and obtained creditor protection under CCAA from the Court, pursuant to the Initial Order.
Pursuant to the Initial Order, KSV Advisory was appointed as monitor and a Stay was obtained against the members of the Trichome Group, and their assets, businesses and directors and officers
that is effective until November 17, 2022. The Stay may be extended subject to further orders of the Court.
Operations in Canada are expected to continue through Trichome Group until a sale transaction or one or more other restructuring transactions is completed.
In connection with the CCAA proceedings, the Credit Parties and the DIP Lender, entered into the DIP Agreement. Pursuant to the DIP Agreement, the DIP Lender has agreed to provide a
super-priority interim revolving credit facility to the Borrower. In accordance with the DIP Agreement, the DIP Facility is to be used during the CCAA proceedings by the Borrower to fund its working capital needs. The DIP Facility is subject to
customary covenants, conditions precedent, and representations and warranties made by the Credit Parties to the DIP Lender. The current DIP Lender's charge approved by Court is up to the maximum amount of $1,825, reflecting the anticipated
borrowings under the DIP Facility up to November 17, 2022.
The CCAA proceedings will afford the Trichome Group the stability and flexibility required to restructure its business, including through a sale and investment solicitation process to be approved
by the Court. Accordingly, the Company’s operations in Canada are expected to continue through the Trichome Group until a sale transaction or one or more restructuring transactions are completed. Court materials filed in connection with
Trichome's CCAA proceedings can be found at https://www.ksvadvisory.com/insolvency-cases/case/trichome.
OFF-BALANCE SHEET ARRANGEMENTS
IM Cannabis had no off-balance sheet arrangements as at September 30, 2022.
TRANSACTIONS WITH RELATED PARTIES
Trichome, through a management service agreement, provided investment management services to the Trichome Financial Cannabis Private Credit LP (the “Fund”)
during the year ended December 31, 2021. The Fund has not engaged in any activity in 2022.
42
Management’s Discussion and Analysis
Under the Focus Agreement (as defined below), IMC Holdings retains an option with Messrs. Shuster and Gabay to re-acquire the sold interest in Focus Medical at its sole discretion and in
accordance with Israeli cannabis regulations. See “Legal and Regulatory – Restructuring” section of the MD&A.
The Company is a party to Indemnification Agreement with certain directors and officers of the Company and Trichome to cover certain tax liabilities, interest and penalties arising from the
Trichome Transaction. See “Risk Factors - Tax Remittance” section of the MD&A.
On August 5, 2022, the Company sold the wholly owned subsidiary of TJAC, Sublime, to a group of purchasers that included current and former members of the Sublime management team for aggregate
proceeds of $100 less working capital adjustments, for a final net purchase price of $89. The transaction constituted a “related party transaction” within the meaning of MI 61-101, however pursuant to Sections 5.5(a) and 5.7(1)(a) of MI 61-101,
the transaction is exempt from the formal valuation and minority shareholder approval requirements of such instrument.
On August 24, 2022, the Company announced that it closed the First Tranche and on October 5, 2022 announced that it closed the Second Tranche. Insiders of the Company, led by Oren Shuster, CEO
and Director, and Shai Shemesh, CFO, subscribed for 1,563,496 Common Shares for aggregate proceeds of US$781,748 in the First Tranche, and Marc Lustig, Executive Chairman and Director of the Company, subscribed for
1,112,504 Common Shares for aggregate proceeds of US$556,252 in the Second Tranche (collectively, the “Insiders”). As a result of the
participation by Insiders, the Private Placement is considered a “related party transaction” pursuant to MI 61-101. The Company relied on Sections 5.5(a) and 5.7(1)(a) of MI 61-101 for exemptions from the requirements to obtain a formal valuation
and minority shareholder approval, respectively, because the fair market value of the Insiders’ participation in the Private Placement was below 25% of the Company’s market capitalization for purposes of MI 61-101.
Other than the aforesaid transactions noted above, the Company had no other transactions with related parties outside of the Group except those pertaining to transactions with key management
personnel and shareholders in the ordinary course of their employment or directorship. Transactions with related parties for the sale of Focus Medical due to the restructuring process were adjusted in the Company’s consolidated financial
statements following the application of IFRS 10. See the “Legal and Regulatory – Restructuring” section of the MD&A.
PROPOSED TRANSACTIONS
There are no proposed transactions as at the date of this MD&A that have not been disclosed.
43
Management’s Discussion and Analysis
CRITICAL ACCOUNTING ESTIMATES
In the process of applying the significant accounting policies, the Group has made the following judgments which have the most significant effect on the amounts recognized in the financial
statements:
JUDGMENTS
Determining the fair value of share-based payment transactions
The fair value of share-based payment transactions is determined upon initial recognition by an acceptable option pricing model. The inputs to the model include share price, exercise price and
assumptions regarding expected volatility, expected life of the share options and expected dividend yield.
Discount rate for a lease liability
When the Company is unable to readily determine the discount rate implicit in a lease in order to measure the lease liability, the Company uses an incremental borrowing rate. That rate represents
the rate of interest that the Company would have to pay to borrow over a similar term and with similar security, the funds necessary to obtain an asset of similar value to the right-of-use asset in a similar economic environment. When there are
no financing transactions that can serve as a basis, the Company determines the incremental borrowing rate based on its credit risk, the lease term and other economic variables deriving from the lease contract’s conditions and restrictions. The
rates at which the Company can borrow will also vary based on the jurisdiction of the leased property, whether it be Israel, Germany, or Canada. In certain situations, the Company is assisted by an external valuation expert in determining the
incremental borrowing rate.
ESTIMATES AND ASSUMPTIONS
The preparation of the financial statements requires management to make estimates and assumptions that have an effect on the application of the accounting policies and on the reported amounts of
assets, liabilities, revenues and expenses. Changes in accounting estimates are reported in the period of the change in estimate.
The key assumptions made in the financial statements concerning uncertainties at the reporting date and the critical estimates computed by the Group that may result in a material adjustment to
the carrying amounts of assets and liabilities within the next financial year are discussed below.
ASSESSMENT OF GOING CONCERN
The use of the going concern basis of preparation of the financial statements. At each reporting period, management assesses the basis of preparation of the financial statements. The going
concern basis of presentation assumes that the Company will continue its operations for the foreseeable future and be able to realize its assets and discharge its liabilities and commitments in the normal course of business.
The Group’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outlays for operating expenses and capital expenditures,
including cost saving plans and restructuring actions taken in 2022. The Company’s board of directors approved a cost saving plan, implemented in whole or in part, to allow the Company to continue its operations and meet its cash obligations. The
cost saving plan consists of cost reduction due to efficiencies and synergies, which include mainly the following steps: discontinuing operation of loss-making activities, reduction in payroll and headcount, reduction in compensation paid to key
management personnel, operational efficiencies and reduced capital expenditures. The Company had experienced lack of timely execution of its cost saving plan, mainly in respect of the Trichome Group.
44
Management’s Discussion and Analysis
These conditions raise substantial doubt about the Company’s ability to continue as a going concern. The interim condensed consolidated financial statements do not include any adjustments
relating to the recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
BIOLOGICAL ASSETS AND INVENTORY
In calculating the value of the biological assets and inventory, management is required to make several estimates, including estimating the stage of growth of the cannabis up to the point of
harvest, harvesting costs, selling costs, average or expected selling prices and list prices, expected yields for the cannabis plants, and oil conversion factors. The valuation of work-in-process and finished goods also requires the estimate of
conversion costs incurred, which become part of the carrying amount for the inventory. The Company must also determine if the cost of any inventory exceeds its net realizable value, such as cases where prices have decreased, or inventory has
spoiled or has otherwise been damaged. See Notes 3 and 4 of the Interim Financial Statements for further information.
BUSINESS COMBINATIONS
In determining the fair value of all identifiable assets acquired and liabilities assumed, the most significant estimates generally relate to contingent consideration and intangible assets.
Management exercises judgment in estimating the probability and timing of when earn-outs are expected to be achieved, which is used as the basis for estimating fair value. Identified intangible assets are fair valued using appropriate valuation
techniques which are generally based on a forecast of the total expected future net cash flows of the acquiree. Valuations are highly dependent on the inputs used and assumptions made by management regarding the future performance of these assets
and any changes in the discount rate applied.
IMPAIRMENT OF PROPERTY, PLANT AND EQUIPMENT AND FINITE LIFE INTANGIBLE ASSETS
The Company assesses impairment of property, plant and equipment and finite life intangible assets when an impairment indicator arises (e.g., change in use or discontinued use, obsolescence or
physical damage). When the asset does not generate cash inflows that are largely independent of those from other assets or group of assets, the asset is tested at the cash generating unit (“CGU”) level. In
assessing impairment, the Company compares the carrying amount of the asset or CGU to the recoverable amount, which is determined as the higher of the asset or CGU’s fair value less costs of disposal and its value-in-use. Value-in-use is assessed
based on the estimated future cash flows, discounted to their present value using a pre-tax discount rate that reflects applicable market and economic conditions, the time value of money and the risks specific to the asset. An impairment loss is
recognized whenever the carrying amount of the asset or CGU exceeds its recoverable amount and is recorded in the consolidated statements of comprehensive loss.
IMPAIRMENT OF INTANGIBLE ASSETS WITH INDEFINITE LIFE AND GOODWILL
Goodwill and intangible assets with an indefinite life or not yet available for use are tested for impairment annually, and whenever events or circumstances that make it more likely than not that
an impairment may have occurred, such as a significant adverse change in the business climate or a decision to sell or dispose all or a portion of a reporting unit. Finite life intangible assets are tested whenever there is an indication of
impairment. Goodwill and indefinite life intangible assets are tested for impairment by comparing the carrying value of each CGU containing the assets to its recoverable amount. Goodwill is allocated to CGUs or groups of CGU’s for impairment
testing based on the level at which it is monitored by management, and not at a level higher than an operating segment. Goodwill is allocated to those CGUs or groups of CGUs expected to benefit from the business combination from which the
goodwill arose, which requires the use of judgment. An impairment loss is recognized for the amount by which the CGU’s carrying amount exceeds it recoverable amount. The recoverable amounts of the CGUs’ assets have been determined based on either
fair value less costs of disposal or value-in-use method. There is a material degree of uncertainty with respect to the estimates of the recoverable amounts of the CGU, given the necessity of making key economic assumptions about the future.
Impairment losses recognized in respect of a CGU are first allocated to the carrying value of goodwill and any excess is allocated to the carrying value of assets in the CGU. Any impairment is recorded in profit and loss in the period in which
the impairment is identified. A reversal of an asset impairment loss is allocated to the assets of the CGU on a pro rata basis. In allocating a reversal of an impairment loss, the carrying amount of an asset shall not be increased above the lower
of its recoverable amount and the carrying amount that would have been determined had no impairment loss been recognized for the asset in the prior period. Impairment losses on goodwill are not subsequently reversed.
45
Management’s Discussion and Analysis
LEGAL CLAIMS
In estimating the likelihood of legal claims filed against certain entities of the Group, the Company’s management rely on the opinions of the respective legal counsel of each relevant entity of
the Group. These estimations are based on each legal counsel’s best professional judgment, taking into account the stage of proceedings and legal precedents in respect of the different issues. Since the outcome of the claims may be determined in
courts, the results could differ from these estimations.
DEFERRED TAX ASSETS
Deferred tax assets are recognized for unused carry-forward tax losses and deductible temporary differences to the extent that it is probable that taxable profit will be available against which
the losses can be utilized. Significant management judgment is required to determine the amount of deferred tax assets that can be recognized, based upon the timing and level of future taxable profits, its source and the tax planning strategy.
VALUATION OF LOANS RECEIVABLE
For loans receivable measured at amortized cost or at Fair Value Through Profit or Loss (“FVTPL”) under IFRS 9 Financial
Instruments (“IFRS 9”), judgment is used by the Company in determining the fair value of the loan at inception of the lending arrangement and at each reporting period. The fair value of the loan
at any given point in time is calculated based on the present value of estimated future loan payments, discounted using an interest rate that would be charged by another market participant for a financing arrangement with similar characteristics.
Judgment is used by the Company in determining what the interest rate would be for sourcing a similar financing arrangement in the market. This can lead to material fair value gains or losses on loans held at FVTPL.
46
Management’s Discussion and Analysis
DERECOGNITION AND MODIFICATION OF LOANS RECEIVABLE
The Company uses its judgment in determining whether the change in the terms of the lending arrangement qualifies as a derecognition of the loan or a modification of the loan under IFRS 9.
Depending on the Company’s judgment, the manner in which the loan is treated, be it a modification or a settlement, can result in materially different results in interest revenue or other income. If there is a modification in a lending
arrangement subsequent to initial recognition, the Company also reassesses the need to modify the expected credit loss associated with the loan.
SHARE-BASED PAYMENTS
The Company uses the Black-Scholes option pricing model in determining the fair value of Options issued to employees. In estimating fair value, the Company is required to make certain assumptions
and estimates such as the expected life of the options, volatility of the Company’s future share price, the risk-free rate, future dividend yields and estimated forfeiture rates at the initial grant date.
ESTIMATED USEFUL LIVES AND DEPRECIATION/AMORTIZATION OF PROPERTY AND EQUIPMENT, AS WELL AS INTANGIBLE ASSETS
Depreciation and amortization of property and equipment, as well as intangible assets, are dependent upon estimated useful lives which are determined through the exercise of judgment. Estimated
useful lives are assessed at the end of each reporting period for any changes in the expected life of the asset and consumption of economic benefits from the use of the asset. Amortization as well as depreciation commences when the asset is first
put into use. The expected life of any intangible assets with a finite life are assessed at the end of each reporting period.
LEASES
Judgment is used in determining the value of the Company’s right-of-use assets and lease liabilities. The value determined for the Company’s right-of-use assets and lease liabilities can be
materially different based on the discount rate selected to present value the future lease payments as well as the likelihood of the Company exercising extensions, termination, and/or purchase options. The discount rate used to present value the
future lease payments over the life of the lease is based on the Company’s incremental borrowing rate at inception of the lease. This rate is determined by the Company using judgment.
In determining the value of the Company’s right-of-use assets and lease liabilities, the Company assesses future business plans to determine whether to include certain extension options noted in
the lease agreement.
If there is no interest rate implicit in the lease agreement, the Company uses a discount rate that would be charged to a similar borrower, with similar risk characteristics, in a mortgage loan
to purchase the leased facility. This discount rate is used to present value the future lease payments in determining the right-of-use asset and lease liability values at inception of the leases.
47
Management’s Discussion and Analysis
REVENUE RECOGNITION
Under IFRS 15 Revenue from Contracts with Customers, judgment is required in recognizing revenue when variable consideration is present in a contract. In certain supply agreements, the Company
stands ready to accept returns on cannabis sales, indicating the possibility of variable consideration.
Judgment is used by the Company in determining which of the above two methods of revenue recognition should be used when recognizing revenue from cannabis sales. Moreover, estimates are used by
the Company in determining the amount of revenue to recognize upon delivery and acceptance of cannabis inventory to a customer.
CHANGES IN ACCOUNTING POLICIES INCLUDING INITIAL ADOPTION
The Company’s significant accounting policies under IFRS are contained in the Interim Financial Statements (refer to Note 2 to the Interim Financial Statements). Certain of these policies involve
critical accounting estimates as they require management to make particularly subjective or complex judgments, estimates and assumptions about matters that are inherently uncertain and because of the likelihood that materially different amounts
could be reported under different conditions or using different assumptions.
The following new accounting standards applied or adopted during the twelve months ended December 31, 2021, had impact on the Annual Financial Statements:
IFRS 3, “BUSINESS COMBINATIONS”:
In October 2018, the IASB issued an amendment to the definition of a “business” in IFRS 3, “Business Combinations” (the “2018 Amendment”). The 2018
Amendment is intended to assist entities in determining whether a transaction should be accounted for as a business combination or as an acquisition of an asset.
The 2018 Amendment consists of the following:
1. Clarification that to meet the definition of a business, an integrated set of activities and assets must include, as a minimum, an input and a substantive process that
together significantly contribute to the ability to create output.
2. Removal of the reference to the assessment whether market participants are capable of acquiring the business and continuing to operate it and produce outputs by integrating
the business with their own inputs and processes.
3. Introduction of additional guidance and examples to assist entities in assessing whether the acquired processes are substantive.
4. Narrowing the definitions of “outputs” and “business” by focusing on goods and services provided to customers.
5. Introducing an optional concentration test that permits a simplified assessment of whether an acquired set of activities and assets is not a business.
The 2018 Amendment is to be applied prospectively to all business combinations and asset acquisitions for which the acquisition date is on or after the beginning of the first annual reporting
period beginning on or after January 1, 2020, with earlier application permitted. The 2018 Amendment is not expected to have a material impact on the Company in the current or future reporting periods.
48
Management’s Discussion and Analysis
AMENDMENT TO IAS 1, “PRESENTATION OF FINANCIAL STATEMENTS”:
In January 2020, the IASB issued an amendment to IAS 1, “Presentation of Financial Statements” (the “2020 Amendment”) regarding the criteria for
determining the classification of liabilities as current or non-current.
The 2020 Amendment includes the following clarifications:
| ● |
What is meant by a right to defer settlement;
|
| ● |
That a right to defer must exist at the end of the reporting period;
|
| ● |
That classification is unaffected by the likelihood that an entity will exercise its deferral right; and
|
| ● |
That only if an embedded derivative in a convertible liability is itself an equity instrument would the terms of a liability not impact its classification.
|
The 2020 Amendment is effective for annual periods beginning on or after January 1, 2023 and must be applied retrospectively.
The Company is evaluating the possible impact of the 2020 Amendment on its current loan agreements.
FINANCIAL INSTRUMENTS
Financial instruments are measured either at fair value or at amortized cost. The table below lists the valuation methods used to determine fair value of each financial instrument.
|
Financial Instruments Measured at Fair Value
|
Fair Value Method
|
|
|
Derivative assets1
|
Black & Scholes model (Level 3 category)
|
|
|
Warrants liability1
|
Black & Scholes model (Level 3 category)
|
|
|
Investment in affiliates
|
Market comparable (Level 3 category)
|
|
|
Financial Instruments Measured at
Amortized Cost |
||
|
Cash and cash equivalents, trade receivables and other account receivables
|
Carrying amount (approximates fair value due to short-term nature)
|
|
|
Loans receivable
|
Amortized cost (effective interest method)
|
|
|
Trade payables, other accounts payable and accrued expenses
|
Carrying amount (approximates fair value due to short-term nature)
|
Note:
| 1. |
Finance expense (income) include fair value adjustment of warrants, investments, and derivative assets measured at fair value, for the nine months ended September 30, 2022 and 2021, amounted to $5,891 and $21,169, respectively.
|
The Group’s exposure to risk for its use of financial instruments are discussed in the Risk Factors.
49
Management’s Discussion and Analysis
PROCEDURES AND INTERNAL CONTROL OVER FINANCIAL REPORTING
In accordance with National Instrument 52-109 – Certification of Disclosure in Issuers’ Annual and Interim Filings (“NI
52-109”) and Rule 13a-15 under the United States Securities Exchange Act of 1934, as amended (the “Exchange Act”), the establishment and maintenance of the Company’s disclosure controls and
procedures (“DC&P”) and internal control over financial reporting (“ICFR”) is the responsibility of management.
Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for
external purposes in accordance with applicable IFRS. Internal control over financial reporting should include those policies and procedures that establish the following:
| ● |
maintenance of records in reasonable detail, that accurately and fairly reflect the transactions and dispositions of assets;
|
| ● |
reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with applicable IFRS;
|
| ● |
receipts and expenditures are only being made in accordance with authorizations of management or the Board; and
|
| ● |
reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial instruments.
|
NI 52-109 requires the CEO and CFO to certify that they are responsible for establishing and maintaining DC&P and ICFR for the Company and have concluded that as at September 30, 2022, those
internal controls have been designed and are effective in providing reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with IFRS.
The Company maintains a set of DC&P designed to provide reasonable assurance that information required to be publicly disclosed is recorded, processed, summarized and reported on a timely
basis. As required by NI 52-109 and Exchange Act Rule 13a-15(b), an evaluation of the design and operation of our DC&P was completed as of September 30, 2022 under the supervision and with the participation of management, including our CEO
and CFO using the criteria set forth in the Internal Control - Integrated Framework (2013), issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based upon this evaluation, our CEO and CFO concluded that as at September
30, 2022, the Company’s DC&P and ICFR were effective.
There have been no changes to the Company’s ICFR during the nine months ended September 30, 2022 that have materially affected, or are likely to materially affect, the Company’s ICFR.
50
Management’s Discussion and Analysis
LIMITATIONS OF DISCLOSURE CONTROLS AND PROCEDURES AND INTERNAL CONTROL OVER FINANCIAL REPORTING
The Company’s management, including the CEO and CFO, believe that due to inherent limitations, any DC&P or ICFR, no matter how well designed and operated, can provide only reasonable, not
absolute, assurance of achieving the desired control objectives. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Also,
projections of any evaluation of effectiveness to future periods are subject to the risk that any design will not succeed in achieving its stated goals under all potential future conditions. Accordingly, because of the inherent limitations in a
cost- effective control system, misstatements due to error or fraud may occur and not be detected. Additionally, management is required to use judgment in evaluating controls and procedures.
LIMITATION ON SCOPE OF DESIGN
In accordance with Section 3.3 of National Instrument 52-109 – Certification of Disclosure in Issuers’ Annual and Interim Filings (“NI 52-109”), the
Company has limited the design of its DC&P and ICFR to exclude the controls, policies and procedures of Oranim Plus (the “Excluded Entity”), acquired by the Company or by one of it subsidiaries within
365 days of the end of the interim period ended September 30, 2022.
The Excluded Entity is included in the Interim Financial Statements. On a combined basis, the Excluded Entity’s’ contribution to the Interim Financial Statements was approximately 4% of total
revenues. Additionally, as at September 30, 2022, the current assets and current liabilities of the Excluded Entity, on a combined basis, represented approximately 6% and 4% of the Company’s consolidated current assets and current liabilities,
respectively. Combined non-current assets, which includes intangible assets and goodwill from these acquisitions, represented approximately 9% of our consolidated non-current assets. Combined non-current liabilities represented approximately 7%
of our consolidated non-current liabilities. Management of the Company is committed to removing this limitation and integrating the Excluded Entity into the Company’s DC&P and ICFR within the timeframe permitted under NI 52-109.
RESTRUCTURING
Current Israeli law requires prior approval by the IMCA, a unit of the MOH, of the identity of any shareholder owning 5% or more of an Israeli company licensed by the IMCA to engage in
cannabis-related activities in Israel. For a number of reasons, including the opportunity to leverage a network of multiple Israeli licensed producers cultivating under the IMC brand, and in contemplation of a “go-public transaction” to
geographically diversify the Company’s share ownership, IMC Holdings restructured its organization on April 2, 2019 (the “IMC Restructuring”) resulting in the divestiture to Oren Shuster and Rafael Gabay of
its interest in Focus, which is licensed by the IMCA to engage in cannabis-related activity in Israel.
IMC Holdings retains an option with Messrs. Shuster and Gabay to re-acquire the sold interest in Focus Medical at its sole discretion and in accordance with Israeli cannabis regulations, within
10 years of the date of the IMC Restructuring (the “Focus Agreement”). The Focus Agreement sets an aggregate exercise price equal to NIS 765.67 per share of Focus Medical for a total consideration of
NIS 2,756,500, that being equal to the price paid by Messrs. Shuster and Gabay for the acquired interests in Focus Medical at the time of the IMC Restructuring.
51
Management’s Discussion and Analysis
As part of the IMC Restructuring, on April 2, 2019, IMC Holdings and Focus Medical entered into an agreement, as amended on January 1, 2021 (the “IP Agreement”),
which provides for Focus Medical’s obligation to use the IMC brand for the sale of any cannabis plant and/or cannabis product produced by Focus, either alone or together with other sub-contractors engaged by Focus Medical through the IP
Agreement.
Focus Medical is also obligated through a services agreement, as amended on January 1, 2021, (the “Services Agreement”) to use IMC Holdings for certain
management and consulting services including: (a) business development services; (b) marketing services; (c) strategic advisory services; (d) locating potential collaborations on a worldwide basis; and (e) financial analysis services through the
Services Agreement.
Under the IP Agreement, the parties apply an arm’s length royalty as a percentage of the licensees’ net revenues, on a quarterly basis in accordance with a transfer pricing analysis to be updated
from time to time, as consideration for Focus’ use of IMC Holdings’ intellectual property.
Under the Services Agreement, the Parties apply an arm’s length markup on total costs, on a quarterly basis, in accordance with a transfer pricing analysis to be updated from time to time, as
consideration for the provision of such services.
Subsequent to the IMC Restructuring, according to accounting criteria in IFRS 10, the Company is viewed as effectively exercising control over Focus, and therefore, the financial statements of
Focus Medical continue to be consolidated with those of the Company, despite the fact that the Company does not own Focus.
ISRAELI MARKET DEVELOPMENT 2012-2022
According to Israeli Ministry of Health, as of September 2022, there are 119,656 medical cannabis licensed patients in Israel. A monthly prescription of 4,576,000 grams of cannabis were recorded
in September 2022 an increase of 523,000 grams of cannabis from December 2021.3
The chart below reflects the growth in licensed medical cannabis patients in Israel between 2012 to September 2022.4
3 Israel Ministry of Health – licensed patients’ data as of September 2022
https://www.gov.il/BlobFolder/reports/licenses-status-september-2022/he/subjects_cannabis_docs_licenses-status-september-2022.pdf
4 Ministry of Health – licensed patients’ data as of September 2022 -
https://www.gov.il/BlobFolder/reports/licenses-status-september-2022/he/subjects_cannabis_docs_licenses-status-september-2022.pdf
52
Management’s Discussion and Analysis
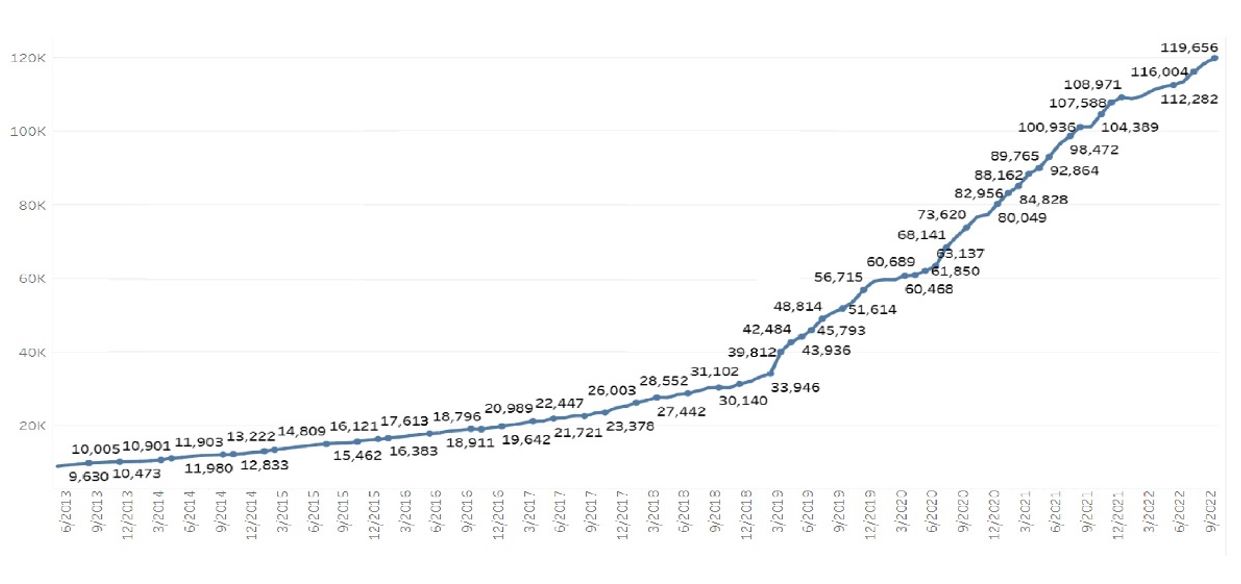
REGULATORY FRAMEWORK IN ISRAEL
In Israel, cannabis is currently defined as a “dangerous drug” according to the Dangerous Drugs Ordinance5 (“DDO”) and the 1961 Single Convention on Narcotic Drugs (“Narcotics Convention”), to which Israel is a signatory. However, both the DDO and the Narcotics Convention allow for the use of
cannabis for medical or research purposes under a supervised and controlled regime. The competent regulatory authority in Israel in all matters concerning the oversight, control and regulation of cannabis for medical production, consumption, and
research in Israel is the IMCA, established by Government Res. No. 3069.6 The production, distribution and consumption of adult-use recreational cannabis products is currently illegal in
Israel.
Patient Medical Consumption
The use of cannabis is allowed for patients and for medical purposes, in respect of certain medical conditions, under a special approval of the MOH. Procedure 1067 of the IMCA sets out a list of medical conditions that are allowed to be treated with medical cannabis products. Such authorized medical conditions are examined and updated from time to
time, and include, among others, cancer, pain, nausea, seizures, muscle spasms, epilepsy, Tourette syndrome, multiple sclerosis, amyotrophic lateral sclerosis, and post-traumatic stress disorder.
5 Cannabis is listed in schedule 1 of the Dangerous Drugs Ordinance [New Version], 1973 [ in English]
https://www.health.gov.il/LegislationLibrary/Samim_01_EN.pdf
6 Israeli Government Res. No. 3609 [in Hebrew], August 7th, 2011 https://www.gov.il/he/departments/policies/2011_des3609
7 Ministry of Health Pharmaceutical Division Policy Number 106 – Licenses for Use of Cannabis https://www.health.gov.il/hozer/CN_106_2019.pdf (in
Hebrew)
53
Management’s Discussion and Analysis
Licensing and Authorization for Commercial Activities in the Medical Cannabis Field
In December 2017, the IMCA issued regulations that standardized the licensing process for any cannabis related activity (the “Road Map”).8 Pursuant to the Road Map, each operation in the medical cannabis field, including the propagation, cultivation, products manufacturing, storage and distribution to licensed pharmacies, and
distribution from licensed pharmacies to licensed patients, requires compliance with the provisions of applicable laws, including the procurement of an appropriate license under the DDO from the IMCA and the maintenance of such license in good
standing. Cannabis licenses may not be transferred, exchanged or assigned without the prior approval of the IMCA. The licenses are valid for a period of up to 3 years and can be renewed with the approval of the IMCA only.
The IMCA has issued a set of directives containing procedures and requirements for applicants for cannabis related activity licenses and has authorized certain entities to issue
official certificates upon compliance with such directives. These directives include (i) Directive 150 (GSP Standard certification); (ii) Directive 151 (GAP Standard certification); (iii) Directive 152 (GMP Standard certification); and (iv)
Directive 153 (GDP Standard certification). Regular and periodic examinations are conducted for licensed entities, in order to ensure compliance with the analytical standards and the level of quality required during each of the phases of
production and distribution of medical cannabis.
Medical Cannabis Imports and Exports
The Narcotics Convention governs the import and export of cannabis between member countries. Since Israel is a member country, any export and import of cannabis is subject to the
Narcotic Convention.
In October 2020, the IMCA issued an updated procedure, titled “Guidelines for Approval of Applications for Importation of Dangerous Drug of Cannabis Type for Medical Use and
for Research” (“Procedure 109”), describing the application requirements for cannabis import licenses for medical and research purposes. Therefore, each import of medical cannabis is to be approved by the
IMCA issuing a specific import permit for each imported shipment, rather than a general license for import. An application for import of medical cannabis can be submitted by an entity licensed by the IMCA for the conduct of medical cannabis
related activity. The Israeli government approved the export of pharmaceutical-grade cannabis and cannabis-based products on January 27, 2019,9 and in December 2020, the IMCA published
guidelines for the medical cannabis export permit application process.10.
Legalization of Adult-Use Recreational Cannabis and CBD for Non-Medical Purposes in Israel
Currently, adult-use recreational cannabis use in Israel and CBD for non-medical use is illegal. In November 2020, an Israeli government committee responsible for
advancing the cannabis market reform published a report supporting and recommending the legalization of adult-use recreational cannabis in Israel. The Israeli parliament dissolved since then without applying the committee’s’ recommendations and
all legislative initiatives were suspended. However, the new government, formed on June 13, 2021, declared, and settled in the coalition agreement, its commitment to legalization of adult-use recreational cannabis. Since the formation of the
new government, several legislative initiatives were filed, including for the decriminalization of the possession of cannabis for individual recreational adult-use and the legalization of CBD for non-medical use. In February 2022, a
Ministry of Health committee contemplated the legality of CBD and published its recommendation that CBD should be excluded from the Drugs Ordinance. The main recommendations of the committee were adopted by the Minister of Health, however, to
date, the Minister has not enacted an order directing that CBD be removed from the Drug Ordinance. On April 1, 2022, new regulations came into force which deemed the previously criminal offences of cannabis
possession and use for self-consumption into administrative offences, which do not impact a criminal record, and limited the penalty to a monetary fine only.
8 Directive 107 - Guidelines for the process of licensing the practice of cannabis for medical use, as amended on October 2020 [Hebrew] - https://www.health.gov.il/hozer/CN_107_2019.pdf
9 Directive 4490 [Hebrew] - https://www.gov.il/he/departments/policies/dec4490_2019
10 Directive 110, December 2020 [Hebrew] - https://www.health.gov.il/hozer/CN_110.pdf
54
Management’s Discussion and Analysis
Previous Regime and Price Control
Until September 2019, under the previous regime, patients licensed for consumption of medical cannabis products by the IMCA received all of their medical cannabis products
authorized under their respective licenses at a fixed monthly price of NIS 370, regardless of each patient’s authorized amount. Since September 2019, under the new regime, licenses to patients were no longer entitling them for such fixed monthly
price. However, some medical cannabis patient licenses granted under the previous regime remain valid, entitling their holders to receive medical cannabis products pursuant to the price controls and supplier restrictions of the former regime. All
licenses under the previous regime expired in Q1 2022.
Regulatory Reform from Licenses to Prescriptions for Medical Treatment of Cannabis
In August 2022, the MOH published a draft outline of the transition reform from licenses to prescriptions for medical treatment of cannabis (the “Proposed Outline”). The Proposed Outline will allow accessibility and significant bureaucratic relief for patients. The main changes proposed in the Proposed Outline are: (i) any specialized
doctor can issue permits without the need for specialized training; (ii) the permits for the use of cannabis will be in the form of prescriptions, and not in the form of licenses from the MOH as the current framework requires; (iii) cannabis
products can be sold in any pharmacy, and not only in pharmacies that have received a special permit from the IMCA and a license from the MOH. The final outline is subject to the approval of the Minister of Health and the approval of the Knesset.
Currently, the required approvals have not yet been received.
REGULATORY FRAMEWORK IN CANADA
The Cannabis Act (Canada), as amended, (the “Cannabis Act”) and the Cannabis Regulations (Canada) made under the
Cannabis Act (the “Cannabis Regulations”) came into force on October 17, 2018, legalizing the sale of adult-use recreational cannabis. The Cannabis Act and Cannabis Regulations establish a licensing and
permitting framework for the production, importation, exportation, testing, packaging, labelling, sending, delivery, transportation, sale, possession and disposal of adult-use recreational cannabis.
On October 17, 2019, amending regulations titled the Regulations Amending the Cannabis Regulations came into force that, among other things, expanded
the scope of the Cannabis Act and Cannabis Regulations to enable the sale of certain categories of cannabis, including cannabis extracts, topicals and edibles, and set THC content limits for certain categories of cannabis products.
55
Management’s Discussion and Analysis
On September 22, 2022, the Government of Canada announced the launch of the legislative review of the Cannabis Act (the “Review”). The Cannabis Act
requires that the Minister of Health cause a review of the Cannabis Act and its administration and operation three years after coming into force. The Minster of Health will table a report in both the Houses of Parliament no later than 18 months
after the start of the review. The Review will include an expert panel that will (i) undertake a factual assessment of the impacts of the Cannabis Act, informed by public engagement process; and (ii) focus on providing advice on areas of the
legislative framework, or its implementation for possible improvement or reform.11
Licensing
The Cannabis Regulations establish six classes of licenses under the Cannabis Act: (i) cultivation licenses, including standard cultivation, micro-cultivation and nursery sub-classes; (ii)
processing licenses, including standard processing and micro-processing sub-classes; (iii) analytical testing licenses; (iv) sales for medical purposes licenses; (v) research licenses; and (vi) cannabis drug licenses. These licenses are valid for
a period of up to five years. License requirements and rules differ depending on the class and/or sub-class of the license.
Security Clearances
Certain people associated with cannabis licensees must hold a valid security clearance issued by Canada’s Minister of Health. For example, in the case of corporations that hold licenses for
cultivation, processing or sale, directors, officers and other individuals who exercise, or are in positions to exercise, direct control over the corporation are required to hold such a security clearance. Under the Cannabis Regulations, the
Minister may refuse to grant security clearances to individuals with organized crime associations or past convictions for, or in association with, drug trafficking, corruption or violent offences. Individuals who have a history of nonviolent,
lower-risk criminal activity (for example, simple possession of cannabis or small-scale cultivation of cannabis plants) are not precluded by legislation from participating in the legal cannabis industry, and the granting of security clearance to
such individuals is at the discretion of the Minister.
Cannabis Tracking System
Under the Cannabis Act, the Minister is authorized to establish and maintain a national cannabis tracking system. Accordingly, Health Canada introduced the Cannabis Tracking and Licensing System,
whereby license holders are required to use this online system to submit monthly tracking reports, new license applications and license renewal requests, among other things. The purpose of this system is to track cannabis throughout the supply
chain to help prevent diversion of cannabis into, and out of, the legal market. The Cannabis Act provides the Minister with the authority to make a ministerial order that would require licensees to report specific information about their
authorized activities with cannabis, in the form and manner specified by the Minister.
11 Government of Canada Launches Legislative Review of the Cannabis Act, (September 22, 2022) see
https://www.canada.ca/en/health-canada/news/2022/09/government-of-canada-launches-legislative-review-of-the-cannabis-act.html; and (September 22, 2022) see Cannabis Act Legislative Review see
https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/laws-regulations/cannabis-act-legislative-review.html
56
Management’s Discussion and Analysis
Cannabis Products
The Cannabis Act and Cannabis Regulations, as amended, set out the requirements for the sale of dried cannabis, fresh cannabis, cannabis plants, cannabis seeds, cannabis edibles, cannabis
extracts and cannabis topicals. Among other requirements, THC content limits are prescribed depending on the product category.
Packaging & Labelling
The Cannabis Regulations set out detailed requirements pertaining to the packaging and labelling of cannabis products that seek to promote informed consumer choice and allow for the safe handling
and transportation of cannabis, while also reducing the appeal of cannabis to youth and promoting safe consumption. These requirements include plain packaging for cannabis products and packaging that is tamper-proof and child-resistant. The
Cannabis Regulations further require package labels to include, among other information, the class of cannabis and the name, phone number and email of the licensed cultivator or processor, the standardized cannabis symbol and information
pertaining to the THC and CBD content. Specific requirements vary depending on the product category of cannabis.
Promotion
The Cannabis Act prohibits the promotion of cannabis, cannabis accessories and cannabis-related services unless authorized by the Cannabis Act through certain exceptions prescribed in the
Cannabis Act and the Cannabis Regulations.
Medical Cannabis
In addition to governance of adult-use recreational cannabis activities, the Cannabis Regulations also govern the regulatory framework associated with medical cannabis in Canada. Prior to the
coming into force of the Cannabis Act and Cannabis Regulations, the sale of medical cannabis was permitted under the Access to Cannabis for Medical Purposes Regulations (Canada) (“ACMPR”). Although the
ACMPR was replaced by the Cannabis Act and Cannabis Regulations, the new rules were not significantly different from the previous rules; changes were made to improve patient access, ensure consistency with adult-use recreational cannabis rules,
and reduce the risk of abuse within the medical access system.
57
Management’s Discussion and Analysis
Provincial and Territorial Regulatory Framework
While the Cannabis Act provides for the regulation of adult-use cannabis production by the federal government, provincial and territorial governments maintain authority to regulate other aspects
of adult-use recreational cannabis activities such as sale and distribution, minimum age requirements, and places where cannabis can be consumed. The following chart summarizes the basic adult-use
recreational cannabis regimes in place as of the date of this MD&A:
|
Province or Territory
|
Minimum Age to Purchase Adult-Use Recreational Cannabis Products
|
Private and/or Public Operated Retailers
|
Online Sales
|
|
Alberta
|
18
|
Private and Public
|
Yes (Public only)
|
|
British Columbia
|
19
|
Private and Public
|
Yes (Public only)
|
|
Manitoba
|
19
|
Private
|
Yes
|
|
New Brunswick
|
19
|
Private and Public
|
Yes
|
|
Newfoundland and Labrador
|
19
|
Private and Public
|
Yes (Public only)
|
|
Nova Scotia
|
19
|
Public
|
Yes
|
|
Ontario
|
19
|
Private and Public
|
Yes (Public only)
|
|
Prince Edward Island
|
19
|
Public
|
Yes
|
|
Quebec
|
21
|
Public
|
Yes
|
|
Saskatchewan
|
19
|
Private
|
Yes
|
|
Northwest Territories
|
19
|
Private and Public
|
Yes (Public only)
|
|
Nunavut
|
19
|
Private and Public
|
Yes
|
|
Yukon
|
19
|
Private and Public
|
Yes (Public only)
|
REGULATORY FRAMEWORK IN GERMANY
On March 10, 2017, the German federal government enacted bill Bundestag-Drucksache 18/8965 – Law amending narcotics and other regulations that amended existing narcotics legislation to recognize
cannabis as a form of medicine and allow for the importation and domestic cultivation of medical cannabis products. Under the updated legislation, cannabis is listed in Annex 3 to the Federal Narcotics Act (“BtMG”)
as a “marketable narcotic suitable for prescription”. Legalization in Germany applies only to cannabis for medicinal purposes under state control in accordance with the Narcotic Convention. Currently, the production, distribution, exportation and
importation of medical cannabis products in Germany is legal, subject to regulations and licensing requirements, while operations involving adult-use recreational cannabis products remain illegal. Nevertheless, current German government has
declared in the coalition agreement its intention to open up the German market also in the adult-use recreational market. Recently, a report of the Federal Ministry of Health (“BMG”), allegedly coordinated
with the responsible ministries, was published in the German media. However, the BMG denied the content of this report shortly afterwards. The BMG confirmed that the key points would not be adopted by the federal cabinet until all ministries
concerned had agreed on them.
Before a proposal for a law would follow, contact would be made with the European Union (“EU”) level. The draft law will then be submitted to the EU
Commission for notification. In the Company’s view, it is unlikely that there will be significant progress with the EU on these developments in 2022.
58
Management’s Discussion and Analysis
Medical cannabis in Germany must comply with the corresponding monographs of the German and European pharmacopoeia. Currently, there are only (non-harmonised) national pharmacopoeial monographs
for cannabis flowers (e.g. in the German Pharmacopoeia (Deutsches Arzneibuch (DAB)) and cannabis extracts (DAB) in the EU. The Committee on Herbal Medicinal Products (HMPC) as the European Medicines Agency’s (EMA) committee responsible for
compiling and assessing scientific data on herbal substances, preparations and combinations, announced that in view of uniform EU quality requirements (including with respect to import and export of cannabis), three new European Pharmacopoeia
(Ph. Eur.) Cannabis monographs that are in preparation and may be of importance in the future:
| ● |
Cannabi-s flos (3028),
|
| ● |
Cannabis extractum siccum (3068),
|
| ● |
Cannabis extractum spissum (3069).
|
All BtMG permit applications must specify the strains and estimated quantities of medical cannabis involved and any subsequent changes must be reported to the Federal Opium Agency of Germany.
Unlike cannabis, CBD is not subject to German narcotics laws, unless it is synthetic CBD that has been included as a substance that can be prescribed and marketed in Annex 3 of the BtMG, which
may or may not be subject to German drug laws depending on its use and dosage. Annex 1 of the Ordinance on the Prescription of Medicinal Products stipulates that CBD is in principle subject to prescription but does not specify a minimum quantity
or a specific dosage form. However, a distinction must be made between consumable products that naturally contain CBD and those that are infused with CBD extract; the European Commission considers the latter to be a type of “food” and has
recently indicated that all current novel food applications have at least insufficient data on safety and therefore none of the applications can currently lead to approval. In light of the above, various products containing CBD can be found in
the German market. There are currently various court decisions that problematize CBD in food (specifically food supplements) and in cosmetics (specifically: mouth oil). On the one hand, CBD is regarded as a medicinal substance and/or as a novel
food subject to authorization and therefore unsuitable for use in a foodstuff, and on the other hand as unsuitable for cosmetic use in the mouth, as CBD would ultimately be consumed in this case (like a foodstuff).
Cultivation in Germany and Distribution of Medical Cannabis Cultivated in Germany
The Federal Opium Agency of Germany’s Federal Institute for Drugs and Medical Devices (“BfArM”) formed a cannabis division (the “Cannabis Agency”) to oversee cultivation, harvesting, processing, quality control, storage, packaging and distribution to wholesalers, pharmacists and manufacturers. The Cannabis Agency also regulates pricing of German-produced
medical cannabis products and serves as an intermediary of medical cannabis product sales between manufacturers, wholesalers and pharmacies on a non-profit basis. In late 2018, the Cannabis Agency issued a call for tenders to award licenses for
local medical cannabis cultivation and distribution of German-cultivated medical cannabis products (the “German Local Tender”). The Cannabis Agency would serve as an intermediary in the supply chain between
such cultivation and distribution. In April 2019, three licenses for local cultivation were granted. In consequence three companies in Germany cultivate on behalf of the Cannabis Agency of the BfArM. Each license permitted the holder to grow up
to 200kg per year for total production of 2,600kg per year collectively from the 13 cultivation lots and 10,400kg over the four-year license period. In July 2021, the BfArM launched the state sale of cannabis grown in Germany. Since then,
pharmacies have been able to purchase medical cannabis in pharmaceutical drug quality for the supply of patients from the BfArM via the portal www.cannabisagentur.de. The sale from the BfArM to pharmacies is at a price of 4.30 euros per gram.
59
Management’s Discussion and Analysis
The Cannabis Agency has no influence on the actual retail price of medical cannabis products and is not responsible for the import of medical cannabis products and will therefore neither purchase
nor distribute imported medical cannabis products. As a wholesaler, the Cannabis Agency sells German-based medical cannabis products in its own name.
Import volumes and procedures
The current regime permits the importation of cannabis plants and plant parts for medicinal purposes under state control subject to the requirements under the Narcotic Convention, according to
which, Germany must estimate the expected demand of medical cannabis products for medical and research purposes for the following year and report such estimates to the International Narcotics Control Board.
As a prerequisite to obtaining a German import license, the supplier must grow and harvest in compliance with EU-GACP-Guidelines and manufacture in compliance with EU-GMP-Guidelines and
certifications, or alternatively, it is a pure EU-GACP product and the EU-GMP manufacturing steps then take place in Germany. All medical cannabis products imported to Germany must derive from plant material cultivated in a country whose
regulations comply with the Narcotic Convention and must comply with the relevant monographs described in the German and European pharmacopeias. While these requirements also apply to the exportation of medical cannabis products, the current
German regime does not allow domestically cultivated medical cannabis products to be directly sold to commercial entities other than the Cannabis Agency.
Dispensing Exclusively via Pharmacies
Medical cannabis products imported pursuant to an import license under the BtMG and AMG/BtMG permits are sold exclusively to pharmacies for final dispensing to patients on a prescription basis as
‘magistral preparations’, a term used in Europe to refer to medical products prepared in a pharmacy in accordance to a medical prescription for an individual patient. Magistral preparations require certain manufacturing steps in the pharmacy.
Such manufacturing steps of the pharmacist typically include the testing and dosing of pre-packaged cannabis inflorescences (typically referred to as “floss”), medical cannabis products for oral administration (dronabinol), medical cannabis
products for inhalation upon evaporation, and medical cannabis-infused teas. In addition to magistral preparations, medical cannabis products are also marketable as pre-packaged, licensed drugs (e.g. Sativex®).
NO U.S. CANNABIS-RELATED ACTIVITIES
The Group does not engage in any U.S. cannabis-related activities as defined in Canadian Securities Administrators Staff Notice 51-352 (Revised) – Issuers with
U.S. Marijuana-Related Activities.
RISK FACTORS
The Company has implemented risk management governance processes that are led by the Board, with the active participation of management, and updates its assessment of its business risks on an
annual basis. Notwithstanding, it is possible that the Company may not be able to foresee all the risks that it may have to face. The market in which IM Cannabis currently competes is complex, competitive and changing rapidly, and its business is
subject to risks inherent in a high growth, heavily regulated enterprise, and the Company has identified certain risks pertinent to the Group’s business that may have affected or may affect the Group’s business, financial conditions, results of
operations and cash flows, as further described throughout this MD&A and under “Risk Factors” in the Annual Information Form. For additional risk factors, readers are directed to the Company’s most recent Annual Information Form, which is (a)
available under the Company’s issuer profile on SEDAR at www.sedar.com, and (b) incorporated into and forms part of the Company’s annual report on Form-40-F filed with the Electronic Data Gathering,
Analysis, and Retrieval system (“EDGAR”) at www.sec.gov/edgar. Sometimes new risks emerge, and management may not be able to predict all of them or be able to
predict how they may cause actual results to be different from those contained in forward looking statements. Readers of this MD&A should not rely upon forward looking statements as a prediction of future results.
60
Management’s Discussion and Analysis
CREDIT RISK
The maximum credit exposure as of September 30, 2022, is the carrying amount of cash and cash equivalents, accounts receivable and other current assets. The Group does not have significant credit
risk with respect to customers. All cash and cash equivalents are placed with major Israeli financial institutions.
Loan receivable credit risk is managed by each loan separately according to the Group’s policy, procedures and control relating to the borrower’s credit risk management. At the end of each
period, the individual loan values are assessed based on a credit risk analysis. As of September 30, 2022, the Group had one loan outstanding with a total balance of approximately $2,680. As security on the Biome Loan, the Obligors hold approximately 745,000 Common Shares which cannot be sold without the proceeds from any sale of the security being provided to the Company as repayment for the Biome Loan until the balance is fully discharged. For
more information, see “Corporate Highlights and Events - Key Highlights for the Quarter Ended September 30, 2022” section of the MD&A.
The expected credit loss analysis is generally based on management’s understanding of the borrower’s experience/integrity, financial health, business plans, capacity, products,
customers, contracts, competitive advantages/disadvantages, and other pertinent factors when assessing credit risk. This would also include the assessment of the borrower’s forecasts as well as taking into consideration any security and/or
collateral the Company has on the outstanding balance.
As security on the loan receivable to Biome Grow Inc., the borrower holds approximately 745,000 Common Shares which cannot be sold without the proceeds from any sale being provided to the Company
as repayment for the loan until the balance is fully discharged.
As of September 30, 2022, the Company assessed the overall risk of the Biome Loan receivable balance and concluded that no
expected credit loss under IFRS 9 was required.
LIQUIDITY RISK
The Company’s liquidity risk is the risk that the Company will not be able to meet its financial obligations as they become due. As of September 30, 2022, the Group’s financial liabilities with
liquidity risk consist of trade payables and other accounts payable which have contractual maturity dates within one year, and lease liabilities. The Group manages its liquidity risk by reviewing its capital requirements on an ongoing basis.
Based on the Group’s working capital position as at September 30, 2022, management considers liquidity risk to be low.
CURRENCY RATE RISK
As at September 30, 2022, a portion of the Group’s financial assets and liabilities held in NIS, Euros, Canadian and U.S. dollars consist of cash and cash equivalents. The
Group’s objective in managing its foreign currency risk is to minimize its net exposure to foreign currency cash flows by transacting, to the greatest extent possible, with third parties as applicable. The Group does not currently use foreign
exchange contracts to hedge its exposure of its foreign currency cash flows, as management has determined that this risk is not significant at this point of time.
61
Management’s Discussion and Analysis
SHARE PRICE RISK
The Company’s investments in unlisted shares are sensitive to the market price risk arising from uncertainties about the future value of these investments. The Company manages
the price risk through diversification and tight management attention.
The Board reviews and approves all decisions related to investments in shares.
At the reporting date, the Group’s exposure to investments in unlisted shares measured at fair value was $2,413.
INFLATION RISK
Global economies are currently experiencing elevated inflation which could curtail levels of economic activity, including in the Company’s primary production markets. This inflation is
predominantly driven by costs of goods as input costs continue to increase as a result of several external factors including but not limited to general uncertainties caused by the Ukraine war, global supply chain constraints and rising energy
prices. As such, delivery and distribution costs, utility costs and other necessary supplies at an economic cost cannot be assured. These are integral requirements for the Company’s business, and it is reasonable to expect that inflation, supply
shortages or increases in demand could impact the Company’s future economic performance and competitiveness, as it may entail a meaningful increase in costs for various goods and services that the Company may not be able to pass onto patients or
customers. In addition, the operations of the Company could be affected should interest rates, inflation or unemployment levels reach levels that curtail consumer trends and spending and, consequently, impact the sales and profitability of the
Company. The Company may not be able to effectively or successfully address such risks and uncertainties or successfully implement operating strategies to mitigate the impact of such risks and uncertainties. In the event that the Company fails to
do so, such failure could materially harm the Company’s business.
TAX REMITTANCE
The Company is subject to the provisions of the ITA12 and to review by CRA13. The Company files its annual tax compliance based on its interpretation of the ITA and CRA’s guidance. There is no
certainty that the returns and tax position of the Company will be accepted by CRA as filed. Any difference between the Company’s tax filings and CRA’s final assessment could impact the Company’s results and financial position.
As at September 30, 2022, the Company’s financial statements included a tax liability of $6,333 and a tax indemnification asset of $1,798. The indemnification asset, intended to cover certain
statutory tax obligations arising from the Trichome Transaction to the CRA, consists of: (1) 695,954 Common Shares; (2) The Company is a party to an indemnification agreement (the “Indemnification Agreement”)
with certain directors and officers of the Company and Trichome to cover certain tax liabilities, interest and penalties arising from the Trichome Transaction; (3) the chairman of the Board entered into a security pledge agreement with the
Company to secure the obligations under the Indemnification Agreement, consisting of certain securities of the Company owned by him; (4) the chairman of the Board transferred the Company a cash amount of $3,250 (the “Indemnification Asset”).
62
Management’s Discussion and Analysis
As of the date hereof, the chairman of the Board is in default of his obligations under the Indemnification Agreement. Accordingly, the Company enforced its security pursuant to the security
pledge agreement to satisfy the tax liabilities, interest and penalties arising from the Trichome Transaction. On April 24, 2022, notices of exercise of its right of exclusive control over the accounts in which are held the financial assets
securing the obligations under the Indemnification Agreement were issued.
There can be no assurance that the Indemnification Asset will be sufficient to satisfy the requisite payments to the CRA. Additionally, there can be no assurance that the directors and officers
whom are party to the indemnification agreement will make sufficient payments to the Company and/or CRA or make the payments at all.
There can be no assurance that income tax laws or the interpretation thereof in any of the jurisdictions in which the Company operates will not be changed or interpreted or administered in a
manner which adversely affects the Company and its shareholders. In addition, there is no assurance that CRA will agree with the manner in which the Company calculates taxes payable or that any of the other tax agencies will not change their
administrative practices to the detriment of the Company or its shareholders.
CONSOLIDATION OF CERTAIN FINANCIAL RESULTS UNDER IFRS 10 AND MAINTENANCE OF COMMON CONTROL
The Company complies with IFRS 10, which applies a single consolidation model using a definition of “control” that requires an investor (as defined in IFRS 10) to consolidate an investee (as
defined in IFRS 10) where: (i) the investor has power over the investee; (ii) the investor has exposure or rights to variable returns from involvement with the investee; and (iii) the investor can use its power over the investee to affect the
amount of the investor’s returns.
Subsequent to the restructuring of IMC Holdings on April 2, 2019, the Company analyzed the terms of the contractual agreements with Focus Medical in accordance with IFRS 10 to conclude whether it
should continue to consolidate the accounts of Focus Medical in its financial statements.
Under IFRS 10, consolidation occurs when an investor can exercise control over an investee. Control is achieved through voting rights or other evidence of power. Where there are no direct
holdings, under IFRS 10, an investor (as defined in IFRS 10) should consider other evidence of power and ability to unilaterally direct an investee’s (as defined in IFRS 10) relevant activities. In view of the contractual agreements and the
guidance in IFRS 10, notwithstanding that the Company has no direct or indirect ownership of Focus Medical, it has sufficient rights to unilaterally direct the relevant activities (a concept known as “de facto control”), mainly due to the
following:
| (a) |
the Company receiving economic benefits from Focus Medical (and the terms of the contractual agreements between the Company and Focus Medical cannot be changed without the approval of the Company);
|
| (b) |
the Company having the option to purchase the divested 74% interest in Focus Medical held by Oren Shuster, the CEO, director and a promoter of the Company, and Rafael Gabay, a former director and a promoter of
the Company;
|
63
Management’s Discussion and Analysis
| (c) |
Messrs. Shuster and Gabay each being a director of Focus Medical (while Mr. Shuster concurrently being a director, officer and substantial shareholder of the Company and Mr. Gabay concurrently being a
substantial shareholder of the Company); and
|
| (d) |
the Company providing management and support activities to Focus Medical through a services agreement
|
Accordingly, under IFRS 10, the Company has “de facto control” over Focus Medical, and therefore consolidates the financial results of Focus Medical in the Company’s financial statements.
Any failure of the Company or Messrs. Oren Shuster and Rafael Gabay to maintain “de facto control” over Focus Medical as defined under IFRS 10 could alter the Company’s consolidation model,
potentially resulting in a material adverse effect on the business, results of operations and financial condition of the Company.
For the period ended September 30, 2022, the Company analyzed the terms of the definitive agreements with HW Shinua in accordance with the accounting criteria set out in IFRS 10. Viewed as
effectively exercising control over HW Shinua, the Company consolidated the financial results of HW Shinua as of the date of signing the definitive acquisition agreement. The definitive agreement for the acquisition of HW Shinua provides the
Company with the power to unilaterally make all decisions regarding the financial and operating policies of HW Shinua and the right to obtain all related economic benefits.
POSSIBLE DIRECT INVOLVEMENT IN THE ISRAELI CANNABIS INDUSTRY
According to current Israeli regulatory medical cannabis framework, any engagement in Cannabis Activities requires receiving the applicable license from the “IMCA”, an agency operated by the
Israeli Ministry of Health, which requires, among other things, pre-approvals by the IMCA (the “IMCA Pre-Approval Requirement”) of the directors, officers and shareholders holding 5% or more of the shares of the license applicant (“Material
Holders”), and of all directors, officers and shareholders that become Material Holders following the grant of the applicable license. Therefore, if the Company will be considered by the IMCA as directly engaged in Cannabis Activities the
aforementioned approvals by the IMCA might apply, on future securityholdings, as described above.
Furthermore, any failure of the Company or its shareholders to comply with the IMCA Pre-Approval Requirement may impact the Group’s ability to continue operating in compliance with any licenses
to engage in Cannabis Activities or to renew such licenses. Any inability of the Group to maintain licenses for Cannabis Activities in good standing may result in a material adverse effect on the Group’s business, financial condition, results of
operations and prospects.
COMPANY’S ABILITY TO CONTINUE AS A GOING CONCERN
The Group’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outlays for operating expenses and capital expenditures,
including cost saving plans and restructuring actions taken in 2022. The Company’s board of directors approved a cost saving plan, implemented in whole or in part, to allow the Company to continue its operations and meet its cash obligations. The
cost saving plan consists of cost reduction due to efficiencies and synergies, which include mainly the following steps: discontinuing operation of loss-making activities, reduction in payroll and headcount, reduction in compensation paid to key
management personnel, operational efficiencies and reduced capital expenditures. The Company had experienced lack of timely execution of its cost saving plan, mainly in respect of the Trichome Group.
64
Management’s Discussion and Analysis
These conditions raise substantial doubt about the Company’s ability to continue as a going concern. The interim condensed consolidated financial statements do not include any adjustments
relating to the recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
CONFLICT AND POLITICAL INSTABILITY IN EASTERN EUROPE
The first part of 2022 has seen significantly higher levels of volatility in global markets due to market participants’ reactions to, and uncertainty surrounding, the magnitude and timing of
government and central bank action to be taken in response to heightened inflation, as well as Russia’s invasion of Ukraine. This volatility has resulted in a decline in the level of activity in the financial markets. Continued market volatility
or uncertainty related to actions taken or to be taken by central banks, a decline in the global macroeconomic outlook, including as a result of Russia’s invasion of Ukraine and the threat, or outbreak of more widespread armed conflict in Eastern
Europe would cause financial market activity to continue to decrease, which would negatively affect the Group’s revenues and capital markets activity.
CAUTION CONCERNING FORWARD-LOOKING INFORMATION
Certain statements in this MD&A may contain “forward-looking statements” or “forward-looking information,” within the meaning of applicable Canadian and United States securities legislation
(collectively referred to herein as “forward-looking statements”). All statements other than statements of fact may be deemed to be forward-looking statements, including statements with regard to expected financial performance, strategy and
business conditions. The words “believe”, “plan”, “intend”, “estimate”, “expect”, “anticipate”, “continue”, or “potential”, and similar expressions, as well as future or conditional verbs such as “will”, “should”, “would”, and “could” often
identify forward-looking statements. These statements reflect management’s current expectations and plans with respect to future events and are based on information currently available to management including based on reasonable assumptions,
estimates, internal and external analysis and opinions of management considering its experience, perception of trends, current conditions and expected developments as well as other factors that management believes to be relevant as at the date
such statements are made. No assurance can be given that the expectations in any forward-looking statement will prove to be correct and, as such, the forward-looking statements included in this MD&A should not
be unduly relied upon. Forward-looking statements is by its nature prospective and requires IM Cannabis to make certain assumptions and is subject to inherent risks and uncertainties. All forward-looking statements are provided as of the
date of this MD&A. The Company does not undertake to update any such forward-looking statements whether as a result of new information, future events or otherwise, except as required by law.
FORWARD LOOKING STATEMENTS
This MD&A and the documents incorporated by reference herein contain certain statements which contain “forward-looking statements” or “forward-looking
information” within the meaning of Canadian and United States securities legislation (each a “forward-looking statement”). All statements, other than statements of historical fact included in this MD&A, including information that address
activities, events or developments that the Company expects or anticipates will or may occur in the future, are forward-looking statements. The use of any of the words “anticipates”, “plans”, “contemplates”, “continues”, “estimates”, “expects”,
“intends”, “proposes”, “might”, “may”, “will”, “shall”, “projects”, “should”, “could”, “would”, “believe”, “predict”, “forecast”, “pursue”, “potential”, “capable”, “budget” and similar expressions are intended to identify forward-looking
statements. Forward-looking statements in this MD&A may include, without limitation, forward-looking statements pertaining to:
| ● |
the Company’s business objectives and milestones and the anticipated timing of execution;
|
65
Management’s Discussion and Analysis
| ● |
the performance of the Company’s business, strategies and operations;
|
| ● |
the intention to expand the business, operations and potential activities of the Company;
|
| ● |
geographic diversification and brand recognition;
|
| ● |
preparations to target, upon legalization, new cannabis markets;
|
| ● |
expectations relating to the number of patients in Israel licensed by the MOH to consume medical cannabis;
|
| ● |
the future impact of the acquisitions of the Israeli Pharmacies and the Panaxia Transaction;
|
| ● |
the expansion of its Israeli sales channels, distribution, delivery and storage capacity, and reach to medical cannabis patients;
|
| ● |
the future product portfolios of the Group and the Company’s ability to export its products, strains and genetics to Israel and Germany;
|
| ● |
the opportunity and ability to expand in Germany and export to new, legal adult-use recreational cannabis markets in Europe;
|
| ● |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets in the jurisdictions in which the Company operates;
|
| ● |
the growth of the Company’s brands in the respective jurisdictions;
|
| ● |
the Company’s retail presence, distribution capabilities and data-driven insights;
|
| ● |
the competitive conditions of the industry, including the Company’s ability to maintain or grow its market share;
|
| ● |
cannabis licensing in the jurisdictions in which the Company operates;
|
| ● |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany;
|
| ● |
expectations regarding the renewal and/or extension of the Group’s licenses;
|
| ● |
the Group’s anticipated operating cash requirements and future financing needs;
|
| ● |
the Group’s expectations regarding its revenue, expenses, profit margins and operations;
|
| ● |
the anticipated Gross Margins, EBITDA and Adjusted EBITDA from the Company’s operations;
|
| ● |
the expected increase in revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions
|
| ● |
statements relating to the Company exiting the Canadian cannabis market to focus Israel, Germany and Europe;
|
| ● |
the Company’s ability to achieve profitability in 2023;
|
| ● |
the completion of the Consolidation;
|
| ● |
expectations related to demand and momentum in the Company’s Israeli operations;
|
| ● |
the restructuring of the Trichome Group under CCAA and the continuation of the Company’s Canadian operations through the Trichome Group until a sale or other restructuring transaction is completed;
|
| ● |
cost savings from restructurings;
|
| ● |
future opportunities for the Company in Israel, particularly in the retail and distribution segments of the cannabis market;
|
66
Management’s Discussion and Analysis
| ● |
future expansion and growth opportunities for the Company in Germany and Europe and the timing of such;
|
| ● |
statements relating to the timing, terms and completion of the Consolidation, including the ratio of pre-consolidation Common Shares to post-consolidation Common Shares that will be effected, non-issuance of fractional shares, and the
contents and delivery of the letter of transmittal following completion of the Consolidation;
|
| ● |
the Group’s reliance on third party suppliers and partners and its ability to enter into additional supply agreements to provide sufficient quantities of medical cannabis to fulfil the Group’s obligations; and
|
| ● |
contractual obligations and commitments.
|
With respect to the forward looking-statements contained in this MD&A, the Company has made assumptions regarding, among other things:
| ● |
the anticipated increase in demand for medical and adult-use recreational cannabis in the markets in which the Company operates;
|
| ● |
the Company’s satisfaction of international demand for its products;
|
| ● |
the Company’s ability to implement its growth strategies and leverage synergies of acquisitions;
|
| ● |
the Company’s ability to reach patients through e-commerce and brick and mortar retail;
|
| ● |
the development and introduction of new products;
|
| ● |
the ability to import and the supply of premium and indoor grown cannabis products from third- party suppliers and partners;
|
| ● |
the changes and trends in the cannabis industry;
|
| ● |
the Company’s ability to maintain and renew or obtain required licenses, permits or authorization related to its domestic and international operations;
|
| ● |
the Company’s ability to rely on the export of, creation and maintenance of and maintain a consistent supply of imported cannabis from suppliers and partners;
|
| ● |
the ability to maintain cost-efficiencies and network of suppliers to maintain purchasing capabilities;
|
| ● |
the effectiveness of its products for medical cannabis patients and adult-use recreational consumers;
|
| ● |
future cannabis pricing and input costs;
|
| ● |
cannabis production yields;
|
| ● |
the Company being able to continue to drive growth from suppliers and partners into Israel, Germany and Europe; and
|
| ● |
the Company’s ability to market its brands and services in Israel, Germany and Europe successfully to its anticipated customers.
|
Readers are cautioned that the above lists of forward-looking statements and assumptions are not exhaustive. Since forward-looking statements address future events and conditions, by their very nature they involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated or implied by such forward-looking statements due to a number of factors and risks. These include:
| ● |
general business risk and liability, including claims or complaints in the normal course of business;
|
67
Management’s Discussion and Analysis
| ● |
any failure of the Company to maintain “de facto” control over Focus Medical and/or HW Shinua in accordance with IFRS 10;
|
| ● |
regulatory authorities in Israel viewing the Company as the deemed owner of more than 5% of Focus Medical or licensed entities in contravention of Israeli regulations;
|
| ● |
limitations on stockholdings of the Company in connection with its direct engagement in the Israeli medical cannabis market;
|
| ● |
the ability and/or need to obtain additional financing for continuing operations;
|
| ● |
the lack of control over the Company’s investees;
|
| ● |
the risk of defaulting on existing debt;
|
| ● |
the Company’s ability to continue as a going concern;
|
| ● |
the ability of the Company to access future financing if needed or on terms acceptable to the Company;
|
| ● |
the failure of the Company to comply with applicable regulatory requirements in a highly regulated industry;
|
| ● |
unexpected changes in governmental policies and regulations affecting the production, distribution, manufacture or use of medial cannabis in any jurisdictions in which the Company currently operates or intends to operate;
|
| ● |
the Company’s ability to continue to meet the listing requirements of the CSE and the NASDAQ;
|
| ● |
the Israeli government deciding to abandon the decriminalization or legalization of adult-use recreational cannabis;
|
| ● |
any change in the political environment which would negatively affect the prospect of decriminalization or legalization of adult-use recreational cannabis in Israel;
|
| ● |
any unexpected failure of Focus Medical to maintain in good standing or renew its licenses;
|
| ● |
any adverse outcome of the Construction Proceedings;
|
| ● |
any unexpected failure of Adjupharm to maintain in good standing or renew any of its Adjupharm Licenses;
|
| ● |
any unexpected failure of TJAC to maintain in good standing or renew any of the TJAC Licenses or MYM License;
|
| ● |
the reliance on the Canadian Facilities to conduct medical cannabis activities;
|
| ● |
any unexpected failure of TJAC and/or MYM to maintain their facilities in good standing with all applicable regulations, including all required licenses and permits and under the TJAC;
|
| ● |
the Group’s ability to maintain ancillary business licenses, permits and approvals required to operate effectively;
|
| ● |
the ability of the Company to integrate acquisitions into the Company’s operations and realize the anticipated benefits and synergies of each such acquisition and the timing thereof and the focus of management on such integration;
|
| ● |
the interpretation of Company’s acquisitions of companies or assets by tax authorities or regulatory bodies, including but not limited to the change of control of licensed entities;
|
| ● |
the ability of the Group to deliver on their sales commitments or growth objectives;
|
| ● |
the Group’s reliance on third-party supply agreements and its ability to enter into additional supply agreements to provide sufficient quantities of medical cannabis to fulfil the Group’s obligations;
|
| ● |
the Group’s possible exposure to liability, the perceived level of risk related thereto, and the anticipated results of any litigation or other similar disputes or legal proceedings involving the Group, including but not limited to the
Construction Proceedings and the class action proceedings described herein;
|
68
Management’s Discussion and Analysis
| ● |
the impact of increasing competition;
|
| ● |
any lack of merger and acquisition opportunities;
|
| ● |
inconsistent public opinion and perception regarding the use of cannabis;
|
| ● |
engaging in activities considered illegal under US federal law related to cannabis;
|
| ● |
political instability and conflict in the Middle East, Eastern Europe and Ukraine;
|
| ● |
adverse market conditions;
|
| ● |
unexpected disruptions to the operations and businesses of the Group as a result of the COVID-19 global pandemic or other disease outbreaks including a resurgence in the cases of COVID-19;
|
| ● |
the inherent uncertainty of production quantities, qualities and cost estimates and the potential for unexpected costs and expenses;
|
| ● |
the Group’s ability to sell its products;
|
| ● |
currency fluctuations;
|
| ● |
inflationary risks;
|
| ● |
any change in accounting practices or treatment affecting the consolidation of financial results;
|
| ● |
the costs of inputs;
|
| ● |
reliance on management; and
|
| ● |
the loss of key management and/or employees.
|
Readers are cautioned that the foregoing list of risk factors is not exhaustive. Additional information on these and other factors that could affect the business, operations or financial results of the Company are detailed under the headings “Risk Factors” and “Contingent Liabilities and Commitments” of this MD&A. The Company and management caution readers not to place undue reliance on any forward-looking statements, which speak only as of the date made. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it can give no assurance that such expectations will prove to have been correct. The Company and management assume no obligation to update or revise them to reflect new events or circumstances except as required by applicable securities laws.
Additional information about the assumptions, risks and uncertainties of the Company’s business and material factors or assumptions on which information contained in forward-looking statements is
based is provided in the Company’s disclosure materials, including in this MD&A under “Legal and Regulatory – Risk Factors” and the Company’s current annual information form under “Risk Factors”, filed with the securities regulatory authorities in Canada and which can be viewed online under the Company’s profile on the System for Electronic Document Analysis and Retrieval (“SEDAR”) at www.sedar.com.
All forward-looking statements in this MD&A is qualified by these cautionary statements.
FINANCIAL OUTLOOK
The forward-looking statements in this MD&A contain statements in respect of estimated revenues. The Company and its management believe that the estimated revenues are reasonable as of the
date hereof and are based on management’s current views, strategies, expectations, assumptions and forecasts, and have been calculated using accounting policies that are generally consistent with the Company’s current accounting policies. These
estimates are considered financial outlooks under applicable securities legislation. These estimates and any other financial outlooks or future-oriented financial information included herein have been approved by management of the Company as of
the date hereof. The Company disclaims any intention or obligation to update or revise any future-oriented financial information, whether as a result of new information, future events or otherwise, except as required by securities legislation.
Readers are cautioned that actual results may vary materially as a result of a number of risks, uncertainties, and other factors, many of which are beyond the Group’s control. See the risks and uncertainties discussed in the “Risk Factors”
section and elsewhere in this MD&A and other risks detailed from time to time in the publicly filed disclosure documents of the Company.
ADDITIONAL INFORMATION
Additional information relating to the Company, including the Company’s Annual Information Form, is available on the Company’s profile on SEDAR at www.sedar.com and on EDGAR at www.sec.gov/edgar.
- - - - - - - - - - - - - - - - - - - - - -