SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
|
REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR 12(g) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended |
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period from ____________________ to ____________________ |
|
SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
(Exact name of Registrant as specified in its charter)
(Jurisdiction of incorporation or organization)
(Address of principal executive offices)
(Name, Telephone, E-Mail and/or Facsimile number and Address of Company Contact Person)
|
Title of each class
|
Trading Symbol
|
Name of each exchange on which registered
|
||
|
|
|
|
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|
|
|
Emerging growth company
|
|
U.S. GAAP ☐
|
|
Other ☐
|
| 5 | ||
| 6 | ||
|
11 | ||
|
11 | ||
|
11 | ||
|
A. |
Reserved. |
11 |
|
B. |
Capitalization and Indebtedness |
11 |
|
C. |
Reasons for the Offer and Use of Proceeds
|
11 |
|
D. |
Risk Factors |
11 |
|
39 | ||
|
A. |
History and Development of the Company
|
39 |
|
B. |
Business Overview |
49 |
|
C. |
Organizational Structure |
63 |
|
D. |
Property, Plants and Equipment |
63 |
|
64 | ||
|
77 | ||
|
A. |
Directors and Senior Management |
77 |
|
B. |
Compensation |
79 |
|
C. |
Board Practices |
89 |
|
D. |
Employees |
95 |
|
E. |
Share Ownership |
96 |
|
F. |
Disclosure of a registrant’s action
to recover erroneously awarded compensation |
96 |
|
96 | ||
|
A. |
Major Shareholders |
96 |
|
B. |
Related Party Transactions |
98 |
|
C. |
Interests of Experts and Counsel
|
99 |
|
100 | ||
|
A. |
Consolidated Statements and Other Financial
Information |
100 |
|
B. |
Significant Changes |
105 |
|
105 | ||
|
A. |
Offer and Listing Details |
105 |
|
B. |
Plan of Distribution |
105 |
|
C. |
Markets |
105 |
|
D. |
Selling Shareholders |
105 |
|
E. |
Dilution |
105 |
|
F. |
Expenses of the Issue |
105 |
|
105 | ||
|
A. |
Share Capital |
105 |
|
B. |
Memorandum and Articles of Association
|
105 |
|
C. |
Material Contracts |
109 |
|
D. |
Exchange Controls |
109 |
|
E. |
Taxation |
|
|
F. |
Dividends and Paying Agents |
116 |
|
G. |
Statement by Experts |
116 |
|
H. |
Documents on Display |
116 |
|
I. |
Subsidiary Information |
117 |
|
J. |
Annual Report to Security Holders
|
117 |
|
117 | ||
|
117 | ||
|
117 | ||
|
117 | ||
|
117 | ||
|
A. |
Disclosure Controls and Procedures
|
117 |
|
B. |
Management’s Annual Report on Internal
Control Over Financial Reporting |
118 |
|
C. |
Attestation Report of Registered Public
Accounting Firm |
118 |
|
D. |
Changes in Internal Controls Over Financial
Reporting |
118 |
|
118 | ||
|
119 | ||
|
119 | ||
|
119 | ||
|
119 | ||
|
119 | ||
|
119 | ||
|
120 | ||
|
120 | ||
|
120 | ||
|
120 | ||
|
122 | ||
|
122 | ||
|
123 |
|
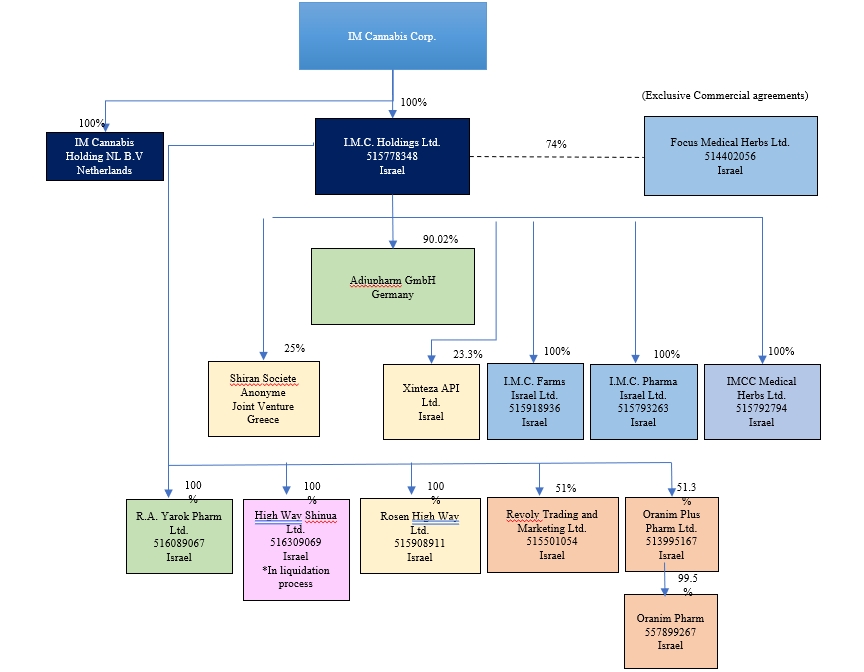
Legal
Entity |
Jurisdiction |
Relationship
with the Company |
|
I.M.C.
Holdings Ltd. (“IMC Holdings”) |
Israel
|
Wholly-owned
subsidiary |
|
I.M.C.
Pharma Ltd. (“IMC Pharma”) |
Israel
|
Wholly-owned
subsidiary of IMC Holdings |
|
I.M.C
Farms Israel Ltd. (“IMC Farms”) |
Israel
|
Wholly-owned
subsidiary of IMC Holdings |
|
Focus
Medical Herbs Ltd.* (“Focus”) |
Israel
|
Private
company over which IMC Holdings exercises “de facto control” under IFRS 10 |
|
R.A.
Yarok Pharm Ltd. (“Pharm Yarok”) |
Israel
|
Wholly-owned
subsidiary of IMC Holdings |
|
Rosen
High Way Ltd. (“Rosen High Way”) |
Israel
|
Wholly-owned
subsidiary of IMC Holdings |
|
Revoly
Trading and Marketing Ltd. dba Vironna Pharm (“Vironna”) |
Israel
|
Subsidiary
of IMC Holdings |
|
Oranim
Plus Pharm Ltd.** (“Oranim
Plus” and together with IMC Pharma, IMC Farms, Pharm Yarok, Rosen High Way, Vironna and Oranim Plus, the “Israeli
Subsidiaries“) |
Israel
|
Subsidiary
of IMC Holdings |
|
Adjupharm GmbH (“Adjupharm”)
|
Germany |
Subsidiary of IMC
Holdings |
|
Trichome Financial
Corp.** |
Canada |
Wholly-owned subsidiary
|
| • |
the Company’s business objectives and milestones and the anticipated timing of execution; |
| • |
the performance of the Company’s business, strategies and operations; |
| • |
the Company’s intentions to expand the business, operations and potential activities of the Company; |
| • |
the Company’s plans to expand its sales channels, distribution, delivery and storage capacity, and reach to medical cannabis
patients; |
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets in the jurisdictions
in which the Company operates; |
| • |
the competitive conditions of the industry, including the Company’s ability to maintain or grow its market share and maintain
its competitive advantages; |
| • |
statements relating to the Company’s commitment to responsible growth and compliance with the strictest regulatory environments;
|
| • |
the Company’s focus on providing premium cannabis products to medical patients in the jurisdictions in which the Company conducts
business and any other jurisdiction in which the Company may conduct business in the future; |
| • |
the Company’s plans to amplify its commercial and brand power to become a global high-quality cannabis player; |
| • |
the Company’s primary goal of sustainably increasing revenue in its core markets; |
| • |
the demand and momentum in the Company’s Israeli and Germany operations; |
| • |
how the Company intends to position its brands; |
| • |
the efficiencies and synergies of the Company as a global organization with domestic expertise in Israel and Germany; |
| • |
expectations that providing high-quality, reliable supply to the Company’s customers and patients will lead to recurring sales;
|
| • |
expectations related to the Company’s introduction of new Stock Keeping Units (“SKUs”);
|
| • |
anticipated cost savings from the reorganization of the Company and the completion thereof upon the timelines disclosed herein;
|
| • |
geographic diversification and brand recognition and the growth of the Company’s brands in the jurisdictions that the Company
operates in or may expand to; |
| • |
expectations related to the Company’s ability to address the ongoing needs and preferences of medical cannabis patients;
|
| • |
the Company’s retail presence, distribution capabilities and data-driven insights; |
| • |
the future impact of the Regulations Amendment (as defined herein) regarding the transition reform from licenses to prescriptions
for medical treatment of cannabis; |
| • |
the Company’s continued partnerships with third party suppliers and partners and the benefits thereof; |
| • |
the Company’s ability to achieve profitability in 2024; |
| • |
the number of patients in Israel licensed by the Israeli Ministry of Health (“MOH”)
to consume medical cannabis; |
| • |
expectations relating to the number of patients paying out-of-pocket for medical cannabis products in Germany; |
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany; |
| • |
expectations related to the demand and the ability of the Company to source premium and ultra-premium cannabis products exclusively
and competition in this product segment; |
| • |
the anticipated impact of inflation and liquidity on the Company’s performance; |
| • |
expectations with respect to the Company’s operating budget and the assumptions related thereto; |
| • |
expectations relating to the Company as a going concern and its ability to conduct business under the ordinary course of operations;
|
| • |
expectations related to the collection the payment awarded in the Judgment and the chances of the claim advancing or the potential
outcome of the Test Kits Appeal (as defined herein); |
| • |
the continued listing of the common shares in the capital of the Company (“Common Shares”)
on the Nasdaq Stock Market (“Nasdaq”) and Canadian Securities Exchange (“CSE”);
|
| • |
cannabis licensing in the jurisdictions in which the Company operates; |
| • |
the renewal and/or extension of the Company’s licenses; |
| • |
the Company’s anticipated operating cash requirements and future financing needs; |
| • |
the Company’s expectations regarding its revenue, expenses, profit margins and operations; |
| • |
the expected increase in revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
future opportunities for the Company in Israel, particularly in the retail and distribution segments of the cannabis market;
|
| • |
future expansion and growth opportunities for the Company in Germany and Europe and the timing of such; |
| • |
contractual obligations and commitments.; and |
| • |
the Company completing the Potential Transaction with Kadimastem (each as defined herein). |
| • |
the Company has the ability to achieve its business objectives and milestones under the stated timelines; |
| • |
the Company will succeed in carrying out its business, strategies and operations; |
| • |
the Company will realize upon its intentions to expand the business, operations and potential activities of the Company; |
| • |
the Company will expand its sales channels, distribution, delivery and storage capacity, and reach to medical cannabis patients;
|
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis in the jurisdictions
in which the Company operates; |
| • |
the competitive conditions of the industry will be favorable to the Company, and the Company has the ability to maintain or grow
its market share and maintain its competitive advantages; |
| • |
the Company will commit to responsible growth and compliance with the strictest regulatory environments; |
| • |
the Company will remain focused on providing premium cannabis products to medical patients in the jurisdictions in which the Company
conducts business and any other jurisdiction in which the Company may conduct business in the future; |
| • |
the Company has the ability to amplify its commercial and brand power to become a global high-quality cannabis player; |
| • |
the Company will maintain its primary goal of sustainably increasing revenue in its core markets; |
| • |
the demand and momentum in the Company’s Israeli and Germany operations will be favorable to the Company; |
| • |
the Company will carry out its plans to position its brands as stated; |
| • |
the Company’s Company has the ability to realize upon the stated efficiencies and synergies the Company as a global organization
with domestic expertise in Israel and Germany; |
| • |
providing a high-quality, reliable supply to the Company’s customers and patients will lead to recurring sales; |
| • |
the Company will introduce of new SKUs; |
| • |
the Company will realize the anticipated cost savings from the reorganization; |
| • |
the Company has the ability to achieve geographic diversification and brand recognition and the growth of the Company’s brands
in the jurisdictions that the Company operates in or may expand to; |
| • |
the Company’s has the ability to address the ongoing needs and preferences of medical cannabis patients; |
| • |
the Company has the ability to realize upon its retail presence, distribution capabilities and data-driven insights; |
| • |
the future impact of the Regulations Amendment will be favorable to the Company; |
| • |
the Company will maintain its partnerships with third parties, suppliers and partners; |
| • |
the Company has the ability to achieve profitability in 2024; |
| • |
the accuracy of number of patients in Israel licensed by the MOH to consume medical cannabis; |
| • |
the accuracy of the number of patients paying out-of-pocket medical cannabis products in Germany; |
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany will occur; |
| • |
the Company has the ability to source premium and ultra-premium cannabis products exclusively and competition in this product segment;
|
| • |
the anticipated impact of inflation and liquidity on the Company’s performance will be as forecasted; |
| • |
the accuracy with respect to the Company’s operating budget and the assumptions related thereto; |
| • |
the Company will remain as going concern; |
| • |
a favorable outcome with respect to the collection the payment awarded in the Judgment and the chances of the claim advancing or
the potential outcome of the Test Kits Appeal; |
| • |
the Company’s Common Shares will remain listed on the Nasdaq and CSE; |
| • |
the Company’s ability to maintain cannabis licensing in the jurisdictions in which the Company operates; |
| • |
the Company has the ability to obtain the renewal and/or extension of the Company’s licenses; |
| • |
the Company has the ability to meet operating cash requirements and future financing needs; |
| • |
the Company will meet or surpass its expectations regarding its revenue, expenses, profit margins and operations; |
| • |
the Company will increase its revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
the Company has the ability to capitalize on future opportunities for the Company in Israel, particularly in the retail and distribution
segments of the cannabis market; |
| • |
the Company will carry out its future expansion and growth opportunities for the Company in Germany and Europe and the timing of
such; |
| • |
the Company will fulfill its contractual obligations and commitments; and |
| • |
the Company will complete the Proposed Transaction with Kadimastem. |
| • |
the Company’s inability to achieve its business objectives and milestones under the stated timelines; |
| • |
the Company inability to carry out its business, strategies and operations; |
| • |
the Company’s inability to realize upon its intentions to expand the business, operations and potential activities of the Company;
|
| • |
the Company will not expand its sales channels, distribution, delivery and storage capacity, and reach to medical cannabis patients;
|
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets will be
unfavorable to the Company in the jurisdictions in which the Company operates; |
| • |
the competitive conditions of the industry will be unfavorable to the Company, and the Company’s inability to maintain or grow
its market share and maintain its competitive advantages; |
| • |
the Company will not commit to responsible growth and compliance with the strictest regulatory environments; |
| • |
the Company’s inability to remain focused on providing premium cannabis products to medical patients in the jurisdictions in
which the Company conducts business and any other jurisdiction in which the Company may conduct business in the future; |
| • |
the Company inability to amplify its commercial and brand power to become a global high-quality cannabis player; |
| • |
the Company will not maintain its primary goal of sustainably increasing revenue in its core markets; |
| • |
the demand and momentum in the Company’s Israeli and Germany operations will be unfavorable to the Company; |
| • |
the Company will not carry out its plans to position its brands as stated; |
| • |
the Company’s inability to realize upon the stated efficiencies and synergies of the Company as a global organization with
domestic expertise in Israel and Germany; |
| • |
providing a high-quality, reliable supply to the Company’s customers and patients will not lead to recurring sales; |
| • |
the Company will not introduce of new SKUs; |
| • |
the Company’s inability to realize upon the anticipated cost savings from the reorganization; |
| • |
the Company’s inability to achieve geographic diversification and brand recognition and the growth of the Company’s brands
in the jurisdictions that the Company operates in or may expand to; |
| • |
the Company’s inability to address the ongoing needs and preferences of medical cannabis patients; |
| • |
the Company’s inability to realize upon its retail presence, distribution capabilities and data-driven insights; |
| • |
the future impact of the Regulations Amendment will be unfavorable to the Company; |
| • |
the Company will not maintain its partnerships with third party suppliers and partners; |
| • |
the Company’s inability to achieve profitability in 2024; |
| • |
the inaccuracy of number of patients in Israel licensed by the MOH to consume medical cannabis; |
| • |
the inaccuracy of the number of patients paying out-of-pocket for medical cannabis products in Germany; |
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany will not occur;
|
| • |
the Company’s ability to source premium and ultra-premium cannabis products exclusively and competition in this product segment;
|
| • |
the anticipated impact of inflation and liquidity on the Company’s performance will not be as forecasted; |
| • |
the inaccuracy with respect to the Company’s operating budget and the assumptions related thereto; |
| • |
the Company will not remain as going concern; |
| • |
an unfavorable outcome of the negotiations or the Construction Proceedings; |
| • |
an unfavorable outcome with respect to the collection the payment awarded in the Judgment and the chances of the claim advancing
or the potential outcome of the Test Kits Appeal; |
| • |
the Company’s Common Shares will not remain listed on the Nasdaq and CSE; |
| • |
the Company’s inability to maintain cannabis licensing in the jurisdictions in which the Company operates; |
| • |
the Company’s inability to obtain the renewal and/or extension of the Company’s licenses; |
| • |
the Company’s inability to meet operating cash requirements and future financing needs; |
| • |
the Company will not meet or surpass its expectations regarding its revenue, expenses, profit margins and operations; |
| • |
the Company will not increase its revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
the Company’s ability to capitalize on future opportunities for the Company in Israel, particularly in the retail and distribution
segments of the cannabis market; |
| • |
the Company will not carry out its future expansion and growth opportunities for the Company in Germany and Europe and the timing
of such; |
| • |
the Company will not fulfill its contractual obligations and commitments; and |
| • |
the Company will not complete the Proposed Transaction with Kadimastem. |
| 1) |
the Company receiving economic benefits from Focus (and the terms of the Commercial Agreements (as defined herein) cannot be changed
without the approval of the Company); |
| 2) |
the Company having the option to purchase the divested 74% interest in Focus held by Oren Shuster, the Chief Executive Officer, director
and a promoter of the Company, and Rafael Gabay, a former consultant director, a former consultant and a promoter of the Company;
|
| 3) |
Messrs. Shuster and Gabay each being a director of Focus (while Mr. Shuster concurrently being a director, officer and substantial
shareholder of the Company and Mr. Gabay concurrently being a substantial shareholder of the Company); and |
| 4) |
the Company providing management and support activities to Focus through the Services Agreement (as defined herein). |
| ● |
diversion of management time and focus from operating our business to addressing acquisition integration challenges; |
| ● |
coordination of research and development and sales and marketing functions; |
| ● |
retention of employees from the acquired company; |
| ● |
cultural challenges associated with integrating employees from the acquired company into our organization; |
| ● |
integration of the acquired company's accounting, management information, human resources, and other administrative systems;
|
| ● |
the need to implement or improve controls, procedures, and policies at a business that prior to the acquisition may have lacked effective
controls, procedures, and policies; |
| ● |
potential write-offs of intangible assets or other assets acquired in transactions that may have an adverse effect on our operating
results in a given period; |
| ● |
liability for activities of the acquired company before the acquisition, including patent and trademark infringement claims, violations
of laws, commercial disputes, tax liabilities, and other known and unknown liabilities; and |
| ● |
litigation or other claims in connection with the acquired company, including claims from terminated employees, consumers, former
stockholders, or other third parties. |
| • |
Shai Shemesh, resigned as Chief Financial Officer of the Company and Itay Vago, was appointed as Chief Financial Officer of the Company
to fill the vacancy created by Shai Shemesh’s resignation. |
| • |
Rinat Efrima, resigned as Chief Executive Officer of IMC Holdings and Eyal Fisher was appointed as the General Manager of IMC Holdings
to fill the vacancy created by Ms. Efrima’s resignation. Mr. Fisher previously held the position of Sales Director of IMC Holdings
prior to his appointment as General Manager. |
| • |
Yael Harrosh resigned as Chief Legal and Operations Officer of the Company and Michal Lebovitz was appointed as General Counsel of
the Company to fill the vacancy created by Mr. Harrosh’s resignation. |
| • |
approve the Proposed Transaction; |
| • |
approve the Spin-Out; |
| • |
a change of name of the Company as directed by Kadimastem and acceptable to the applicable regulatory authorities effective upon
Closing; and |
| • |
reconstitution of the Board. |
| • |
the execution of a definitive agreement; |
| • |
completion of mutually satisfactory due diligence; |
| • |
completion of the share consolidation; and |
| • |
receipt of all required regulatory, corporate and third party approvals, including approvals by governing regulatory bodies, the
shareholders of IMC and Kadimastem, applicable Israeli governmental authorities, and the fulfilment of all applicable regulatory requirements
and conditions necessary to complete the Proposed Transaction. |

|
The Top-Shelf
Collection – IMC’s premium product line, which offers indoor-grown, high-THC cannabis flowers with strains such as
Lemon Rocket, Diesel Drift, Tropicana Gold, Lucy Dreamz, Santa Cruz, Or’enoz, and Banjo. Inspired by the 1970’s cannabis culture
in America, the Top-Shelf Collection targets the growing segment of medical patients who are cannabis culture enthusiasts. |
 |





|
BLKMKT™, the Company’s
second Canadian brand, super premium product line with indoor grown, hand-dried and hand-trimmed high-THC cannabis flowers. The BLKMKT™
includes JEALOUSY, BACIO GLTO, PNPL P, PARK FIRE OG, UPSIDE DOWN C. In Q4 2023, the Company relaunched JEALOUSY and BACIO GLTO.
|
 |








|
Revenues
from Continuing operations - By Product Type | ||||
|
Financial Year |
Medical Cannabis Products |
Adult-Use Recreational Cannabis Products |
Other Products |
Total |
|
2023 |
$44,246 |
-
|
$4,559 |
$48,804 |
|
2022 |
$48,384 |
-
|
$5,951 |
$54,335 |
|
2021 |
$26,449 |
-
|
$7,604 |
$34,053 |
| • |
The intellectual property agreement dated April 2, 2019 and as amended on January 1, 2021, between IMC Holdings and Focus (the “IP
Agreement”) and the Services Agreement dated April 2, 2019 and as amended on January 1, 2021, between IMC Holdings and Focus
(the “Services Agreement” and together with the IP Agreement, the “Commercial
Agreements”), whereby IMC Holdings derives economic benefit from Focus and whereby Focus (i) uses the IMC brand on an exclusive
basis for the sale of cannabis products; and (ii) engages IMC Holdings to provide certain management and consulting services. As a result
of the Company’s commercial relationship with Focus, it is dependent on Focus maintaining its licenses, as well as any ancillary
licenses required to carry on its operations in the Israeli medical cannabis industry. |
| • |
Supply agreements with third party cannabis cultivators and suppliers to meet the Israeli market’s demand for the Company’s
products. |
| • |
Purchase orders received from time to time for the sale of the Company’s products to pharmacies or distributors, either in
association with Focus or through the Company’s direct trading house operations. |
| • |
Ongoing retail purchases of the Company’s products sold at the Israeli Pharmacies by Israeli medical cannabis patients.
|
|
Israel |
Germany |
Adjustments |
Total |
|||||||||||||||||||||||||||||
|
2023 |
2022 |
2023 |
2022 |
2023 |
2022 |
2023 |
2022 |
|||||||||||||||||||||||||
|
Revenues |
$ |
43,316 |
$ |
50,500 |
$ |
5,488 |
$ |
3,835 |
$ |
- |
$ |
- |
$ |
48,804 |
$ |
54,335 |
||||||||||||||||
|
Segment income (loss) |
$ |
(6,627 |
) |
$ |
(23,606 |
) |
$ |
(1,615 |
) |
$ |
(3,225 |
) |
$ |
- |
$ |
- |
$ |
(8,242 |
) |
$ |
(26,831 |
) | ||||||||||
|
Unallocated corporate expenses |
$ |
- |
$ |
- |
$ |
- |
$ |
- |
$ |
(4,550 |
) |
$ |
(3,960 |
) |
$ |
(4,550 |
) |
$ |
(3,960 |
) | ||||||||||||
|
Total operating (loss) income |
$ |
(6,627 |
) |
$ |
(23,606 |
) |
$ |
(1,615 |
) |
$ |
(3,225 |
) |
$ |
(4,550 |
) |
$ |
(3,960 |
) |
$ |
(12,792 |
) |
$ |
(30,791 |
) | ||||||||
|
Depreciation, amortization & impairment
|
$ |
2,823 |
$ |
6,747 |
$ |
173 |
$ |
200 |
$ |
- |
$ |
- |
$ |
2,996 |
$ |
6,947 |
||||||||||||||||
|
Israel |
Germany |
Adjustments |
Total |
|||||||||||||||||||||||||||||
|
2023 |
2022 |
2023 |
2022 |
2023 |
2022 |
2023 |
2022 |
|||||||||||||||||||||||||
|
Revenues |
$ |
9,375 |
$ |
13,136 |
$ |
1,323 |
$ |
1,325 |
$ |
- |
$ |
- |
$ |
10,698 |
$ |
14,461 |
||||||||||||||||
|
Segment income (loss) |
$ |
(3,653 |
) |
$ |
(10,280 |
) |
$ |
(580 |
) |
$ |
(517 |
) |
$ |
- |
$ |
- |
$ |
(4,233 |
) |
$ |
(10,797 |
) | ||||||||||
|
Unallocated corporate income (expenses)
|
$ |
- |
$ |
- |
$ |
- |
$ |
- |
$ |
(932 |
) |
$ |
90 |
$ |
(932 |
) |
$ |
90 |
||||||||||||||
|
Total operating (loss) income |
$ |
(3,653 |
) |
$ |
(10,280 |
) |
$ |
(580 |
) |
$ |
(517 |
) |
$ |
(932 |
) |
$ |
90 |
$ |
(5,165 |
) |
$ |
(10,707 |
) | |||||||||
|
Depreciation, amortization & impairment
|
$ |
684 |
$ |
4,957 |
$ |
47 |
$ |
48 |
$ |
- |
$ |
- |
$ |
731 |
$ |
5,005 |
||||||||||||||||
| ● |
Revenues from continuing operations for the year ended December 31, 2023 and 2022 were $48,804 and $54,335, respectively, representing
a decrease of $5,531 or 10%. Revenues for the three months ended December 31, 2023, and 2022 were $10,698 and $14,461, respectively,
representing a decrease of $3,763 or 26%. The decrease in revenues is primarily attributed to the effects of the Israel – Hamas
war and the different challenges it caused from a business perspective effecting the Company sells and also its operation activities such
as longer importing cycles (transportation of goods, approvals from relevant authorities etc.) |
| ● |
Revenues from the Israeli operation were attributed to the sale of medical cannabis through the Company’s agreement with Focus
Medical and the revenues from the Israeli Pharmacies the Company owns, mostly from cannabis products. |
| ● |
In Germany, Company revenues were attributed to the sale of medical cannabis through Adjupharm. |
| ● |
Total dried flower sold for the year ended December 31, 2023, was 8,609 kg at an average selling price of $5.14 per gram compared
to 6,794kg for the same period in 2022 at an average selling price of $7.12 per gram, mainly attributed to the inventory life cycle, discounts
given and increased competition in the segment. Total dried flower sold for the three months ended December 31, 2023, was 2,082kg at an
average selling price of $4.52 per gram compared to 2,334kg for the three months ended December 31, 2022, at an average selling price
of $5.19 per gram. The decreased is mainly attributed to the Israel – Hamas war effect. |
|
Less than one year |
1 to 5 years |
6 to 10 years |
> 10 years |
|||||||||||||
|
Contractual Obligations |
$ |
12,618 |
$ |
1,293 |
- |
- |
||||||||||
|
Payments Due by Period |
||||||||||||||||||||
|
Contractual Obligations
|
Total |
Less than one year |
1 to 3 years |
4 to 5 years |
After 5 years |
|||||||||||||||
|
Debt |
$ |
12,513 |
$ |
12,119 |
$ |
394 |
$ |
- |
$ |
- |
||||||||||
|
Finance Lease Obligations |
$ |
1,398 |
$ |
499 |
$ |
814 |
$ |
85 |
$ |
- |
||||||||||
|
Total Contractual Obligations
|
$ |
13,911 |
$ |
12,618 |
$ |
1,208 |
$ |
85 |
$ |
- |
||||||||||
|
Name |
Position(s) with the
Company |
Other Directorships
|
Date of Initial Appointment
|
|
Oren Shuster |
Chief Executive Officer and Director
|
IMC Holdings; Focus; Pharm Yarok; Rosen High Way; IMC Pharma; IMC Farms; Ewave Group Ltd (“Ewave”) and its subsidiaries |
October 11, 2019 |
|
Marc Lustig |
Executive Chairman and Director |
PharmaCielo Ltd.
Cresco Labs Inc.
Aequus Pharmaceuticals Inc.
22 Capital Corp.
BriaCell Therapeutics Corp. |
October 11, 2019 |
|
Moti Marcus(2)(3)(4)
|
Director |
Nil |
September 12, 2022 |
|
Einat Zakariya(2)(3)(4)
|
Director |
HYGEAR Inc. |
September 12, 2022 |
|
Brian Schinderle(2)(3)
|
Director |
Nil |
February 22, 2021 |
|
Uri Birenberg |
Chief Financial Officer |
Nil |
October 10, 2023 |
|
Eyal Fisher |
Chief Executive Officer of IMC Holdings
and each of the Israeli Subsidiaries |
Nil |
March 8, 2023 |
|
Michal Lebovitz Nissimov |
General Counsel |
Nil |
April 14, 2023 |
|
Richard Balla |
Chief Executive Officer of Adjupharm
|
Nil |
October 11, 2019 |
| (1) |
Information furnished by the respective individual. |
| (2) |
Member of the Audit Committee. |
| (3) |
Member of Compensation Committee. |
| (4) |
Member of the Governance and Nomination Committee. |
| (1) |
Oren Shuster, CEO and a director of the Company; |
| (2) |
Uri Birenberg, CFO of the Company; |
| (3) |
Itay Vago, former CFO of the Company; |
| (4) |
Shai Shemesh, former CFO of the Company; |
| (5) |
Marc Lustig, Executive Chairman and a director of the Company; |
| (6) |
Eyal Fisher, CEO of the IMC Holdings and each of the Israeli Subsidiaries; and |
| (7) |
Richard Balla, CEO of Adjupharm. |
| 1. |
base salary; |
| 2. |
cash bonuses; and/or |
| 3. |
long-term incentives. |
| 1. |
Base Salary |
| 2. |
Cash Bonuses |
| 3. |
Long Term Incentives |
| (a) |
the maximum number of RSUs available for grant to any one person under the RSU Plan and any other Securities Based Compensation Arrangements
of the Company in a 12-month period is 5% of the total number of Common Shares then outstanding on a non-diluted basis; and |
| (b) |
the maximum number of Common Shares issuable to insiders of the Company (as a group) under the RSU Plan, together with any other
Common Shares issuable under any other Securities Based Compensation Arrangements, shall not exceed at any time or within any 12-month
period, 10% of the issued and outstanding Common Shares on a non-diluted basis at the time of grant. |
| (a) |
increase the number of Common Shares which may be issued pursuant to the RSU Plan, other than by virtue of a change in Common Shares,
whether by reason of a stock dividend, consolidation, subdivision or reclassification which adjustment may be made by the Board or Compensation
Committee for the number of Common Shares available under the RSU Plan and the number of Common Shares subject to RSUs; |
| (b) |
amend the definition of “Participant” under the RSU Plan which would have the potential of narrowing, broadening or increasing
insider participation; |
| (c) |
amendments to cancel and reissue RSUs; |
| (d) |
amendments to the list of amendments to the RSU Plan or RSUs requiring requisite regulatory and shareholder approval and those subject
to requisite regulatory approval (where required) but not subject to shareholder approval; |
| (e) |
amendments that extend the term of an RSU; |
| (f) |
amendments to the participation limits including: the maximum number of shares issuable under the RSU Plan, limitations on grants
of RSUs to any one person in a 12-month period, grants within a one year period to insiders, and the number of shares issuable to a person
providing investor relations activities in any 12-month period; and |
| (g) |
amendments to the RSU Plan that would permit RSUs, or any other right or interest of a RSU Participant under the RSU Plan, to be
assigned or transferred, other than for normal estate settlement purposes. |
| (a) |
amendments of a housekeeping nature; |
| (b) |
amendments to the vesting provisions of a RSU or the RSU Plan; |
| (c) |
amendments to the definitions, other than such definitions noted above; |
| (d) |
amendments to reflect changes to applicable securities laws; and |
| (e) |
amendments to ensure that the RSUs granted under the RSU Plan will comply with any provisions respecting income tax and other laws
in force in any country or jurisdiction of which a RSU Participant to whom a RSU has been granted may from time to time be a resident,
citizen or otherwise subject to tax therein. |
|
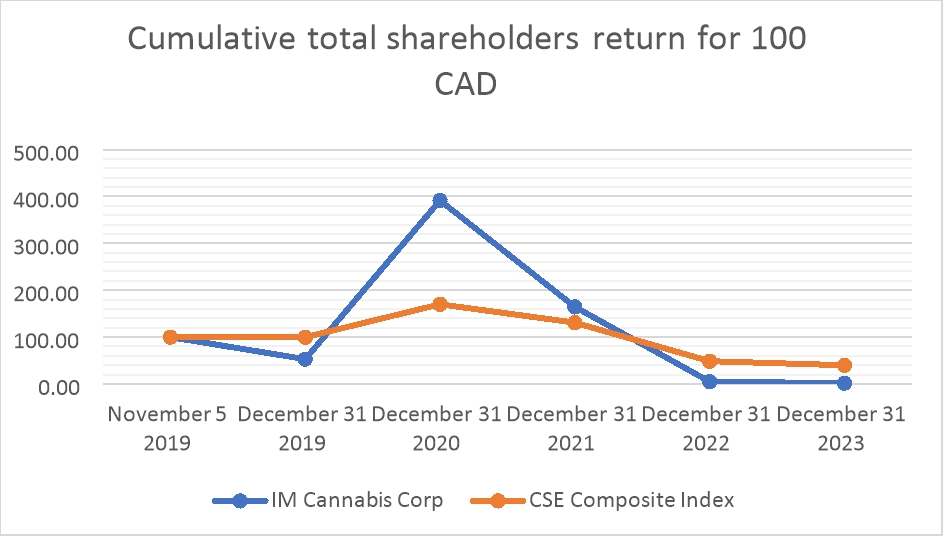
November 5, 2019
|
December 2019
|
December 2020
|
December 2021
|
December 2022
|
December 2023
| |
|
IM Cannabis Corp.
|
$100.00 |
$53.97 |
$398.41 |
$167.46 |
$5.16 |
$1.83 |
|
CSE Composite Index
|
$100.00 |
$99.81 |
$170.10 |
$130.97 |
$48.67 |
$40.95 |
| 1. |
Brian Schinderle (Chair); |
| 2. |
Moti Marcus; and |
| 3. |
Einat Zakariya, |
|
Name and Principal Position
|
Year |
Salary
($)(1)
|
Share-Based Awards
($) |
Option-Based Awards
($)(8)
|
Non-Equity Incentive
Plan Compensation
($) |
Pension Value
($) |
All Other
Compensation ($) |
Total
Compensation ($) | |
|
Annual Incentive Plans
|
Long-Term Incentive
Plans | ||||||||
|
Oren Shuster
CEO
and Director |
2023 |
476,266(2)
|
Nil |
331,802 |
Nil |
Nil |
Nil |
Nil |
818,068 |
|
2022 |
506,244(2)
|
Nil |
1,110,057 |
Nil |
Nil |
Nil |
Nil |
1,616,301 | |
|
2021 |
515,731(2)
|
Nil |
1,388,455 |
121,000 |
Nil |
Nil |
Nil |
2,025,186 | |
|
Uri Birenberg(3)
CFO
|
2023 |
73,558 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
73,558 |
|
2022 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil | |
|
2021 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil | |
|
Itay Vago(4)
Former
CFO |
2023 |
180,968 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
180,968 |
|
2022 |
53,219 |
Nil |
508 |
Nil |
Nil |
Nil |
Nil |
53,727 | |
|
2021 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil | |
|
Shai Shemesh(5)
Former
CFO |
2023 |
135,934 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
135,934 |
|
2022 |
321,950 |
Nil |
307,636 |
Nil |
Nil |
Nil |
Nil |
629,586 | |
|
2021 |
300,607 |
Nil |
408,653 |
82,500 |
Nil |
Nil |
Nil |
791,760 | |
|
Marc Lustig
Executive
Chairman and Director(6) |
2023 |
129,920 |
79,959 |
Nil |
Nil |
Nil |
Nil |
Nil |
209,879 |
|
2022 |
282,480 |
558,538 |
50,089 |
Nil |
Nil |
Nil |
Nil |
891,107 | |
|
2021 |
264,000 |
1,286,498 |
329,846 |
Nil |
Nil |
Nil |
Nil |
1,880,344 | |
|
Eyal Fisher(7)
CEO of the IMC Holdings
and each of the Israeli Subsidiaries |
2023 |
216,998 |
Nil |
1,693 |
Nil |
Nil |
Nil |
Nil |
216,998 |
|
2022 |
215,586 |
Nil |
4,381 |
Nil |
Nil |
Nil |
Nil |
217,279 | |
|
2021 |
144,506 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
144,506 | |
|
Richard Balla
CEO of Adjupharm
|
2023 |
175,385 |
Nil |
Nil |
87,692 |
Nil |
Nil |
30,895 |
293,972 |
|
2022 |
164,186 |
Nil |
37 |
Nil |
Nil |
Nil |
29,066 |
193,289 | |
|
2021 |
176,674 |
Nil |
7,799 |
121,087 |
Nil |
Nil |
38,083 |
343,643 | |
| (1) |
Each of Messrs. Shuster, Birenberg, Vago, Shemesh and Fisher received their compensation in NIS and Mr. Balla received his compensation
in Euros. All salaries were converted to CDN pursuant to the average Bank of Canada rate for the applicable fiscal year. |
| (2) |
Oren Shuster, through Ewave, entered into a consulting agreement with the Company pursuant to which he is paid NIS 108,350 plus VAT
per month (approximately $39,544 plus tax per month) in consideration of his CEO services provided to Company. Mr. Shuster did not earn
consideration for his role as a director of the Company during the fiscal years ended December 31, 2023, 2022 and 2021. |
| (3) |
Mr. Birenberg was appointed as CFO of the Company effective October 10, 2023. |
| (4) |
Mr. Vago was appointed as CFO of the Company effective March 8, 2023 and resigned effective October 10, 2023, but stayed on to assist
the transition until November 15, 2023. |
| (5) |
Mr. Shemesh resigned as the CFO of the Company effective March 8, 2023. |
| (6) |
Mr. Lustig does not earn consideration for his role as a director of the Company; however, Mr. Lustig, through L5 Capital, entered
into a consulting agreement with the Company pursuant to which he is paid $5,250 per month in consideration of his Executive Chairman
services provided to the Company. |
| (7) |
Mr. Fisher was appointed as CEO of the IMC Holdings and each of the Israeli Subsidiaries effective March 15, 2023. |
| (8) |
The Company used the Black-Scholes pricing model as the methodology to calculate the grant date fair value, and relied on the following
the key assumptions and estimates for each calculation under the following assumptions: (i) risk free interest rate of 0.42% to 1.97%
(ii) expected dividend yield of 0%; (iii) expected volatility of 76.28% to 82.31%; and (iv) a term of 5 to 10 years. The Black-Scholes
pricing model was used to estimate the fair value as it is the most accepted methodology. |
|
Option-based
Awards |
Share-based
Awards | ||||||
|
Name
|
Number
of securities underlying unexercised Options
(#)(1)
|
Option
exercise price
($)
|
Option
expiration date |
Value
of unexercised
in-the-money Options ($)
|
Number
of shares or units of shares that have not vested
(#)
|
Market
or payout value of share-based awards that have not vested(4)
($)
|
Market or payout value
of vested share-based awards not paid out or distributed
($) |
|
Oren Shuster |
6,250
75,000
50,000 |
40.00
58.70
16.00 |
June 9, 2025
May 19, 2026
January 4, 2029 |
Nil
Nil
Nil |
Nil |
Nil |
Nil |
|
Marc Lustig |
67,500 |
16.00 |
September 11, 2029 |
Nil |
Nil |
Nil |
Nil |
|
Eyal Fisher |
1,000 |
27.30 |
April 4, 2027 |
Nil |
Nil |
Nil |
Nil |
|
Richard Balla |
9,000 |
16.00 |
July 30, 2029 |
Nil |
Nil |
Nil |
Nil |
| (1) |
Each Option entitles the holder to purchase one Common Share. |
|
Name
|
Option-based
awards – Value vested during the year
($)
|
Share-based
awards – Value vested during the year ($) |
Non-equity
incentive plan compensation – Value earned during the year
($)
|
|
Oren Shuster |
932,254 |
Nil |
Nil |
|
Marc Lustig |
Nil |
481,494 |
Nil |
|
Richard Balla |
Nil |
Nil |
87,692 |
|
Eyal Fisher |
2,171 |
Nil |
Nil |
|
Name(1)
|
Fees earned
($)(2) |
Share-based awards
($) |
Option-based awards ($)(3) |
Non-equity incentive
plan compensation ($) |
Pension value ($) |
All other compensation ($) |
Total
($) |
|
Brian Schinderle |
73,700(4) |
Nil |
61,616 |
Nil |
Nil |
Nil |
135,316 |
|
Moti Marcus |
76,700(45) |
Nil |
19,963 |
Nil |
Nil |
Nil |
96,663 |
|
Einat Zakariya |
73,200 |
Nil |
19,963 |
Nil |
Nil |
Nil |
93,163 |
| (1) |
Each of Mr. Marcus and Ms. Zakariya received their compensation in NIS and Mr. Schinderle received his compensation in USD. All salaries
were converted to CDN pursuant to the average Bank of Canada rate for the applicable fiscal year. |
| (2) |
Each director is entitled to a $13,750 payment per quarter for their role as a director of the Company. For each Audit Committee
meeting, the Chair receives a $1,500 payment and each other member receives a $1,000 payment and for each of the Compensation Committee
and Governance and Nomination Committee meetings, the Chair receives a $1,200 payment and each other member receives a $700 payment.
|
| (3) |
The Company used the Black-Scholes pricing model as the methodology to calculate the grant date fair value, and relied on the following
the key assumptions and estimates for each calculation under the following assumptions: (i) risk free interest rate of 0.42% to 3.03%
(ii) expected dividend yield of 0%; (iii) expected volatility of 78.7% to 82.01%; and (iv) a term of 5 to 10 years. The Black-Scholes
pricing model was used to estimate the fair value as it is the most accepted methodology. |
| (4) |
Mr. Schinderle receives compensation through Solidum Capital Advisors LLC. |
| (5) |
Mr. Marcus receives compensation through Marcus Management Services Ltd. |
|
Option-based
Awards |
Share-based
Awards | ||||||
|
Name
|
Number
of securities underlying unexercised Options(1)
(#)
|
Option
exercise price
($)
|
Option
expiration date |
Value
of unexercised
in-the-money Options ($)
|
Number
of shares or units of shares that have not vested
(#)
|
Market
or payout value of share-based awards that have not vested
($)
|
Market or payout value
of vested share-based awards not paid out or distributed
($) |
|
Brian Schinderle |
9,000 |
100.00 |
February 28, 2026 |
Nil |
Nil |
Nil |
Nil |
|
Moti Marcus |
9,000 |
6.00 |
September 19, 2027 |
Nil |
Nil |
Nil |
Nil |
|
Einat Zakariya |
9,000 |
6.00 |
September 19, 2027 |
Nil |
Nil |
Nil |
Nil |
| (1) |
Each Option entitles the holder to purchase one Common Share. |
|
Name
|
Option-based
awards – Value vested during the year
($)
|
Share-based
awards – Value vested during the year ($) |
Non-equity
incentive plan compensation – Value earned during the year
($)
|
|
Brian Schinderle |
207,000 |
Nil |
Nil |
|
Moti Marcus |
14,601 |
Nil |
Nil |
|
Einat Zakariya |
14,601 |
Nil |
Nil |
|
Name of Director |
Board |
Audit Committee |
Compensation Committee |
Governance and Nomination Committee |
|
Oren Shuster |
100% (17/17) |
- |
- |
- |
|
Marc Lustig |
94.12% (16/17) |
- |
- |
- |
|
Moti Marcus |
100% (17/17) |
100% (6/6) |
100% (1/1) |
- |
|
Einat Zakariya |
100% (17/17) |
83.33% (5/6) |
100% (1/1) |
- |
|
Brian Schinderle |
100% (17/17) |
100% (6/6) |
100% (1/1) |
- |
| (a) |
overseeing that the day-to-day business affairs of the Company are appropriately managed and taking steps to maintain and enhance
an effective senior management team reporting to the CEO; |
| (b) |
recommending to the Board the Company’s financial and operating goals and objectives and, following approval by the Board thereof,
consistently striving to achieve such goals and objectives; |
| (c) |
formulating, and presenting to the Board for approval, long-term business plans, strategies and policies having the objective of
maximizing the Company’s long-term success and the creation of shareholder value; |
| (d) |
together with other senior management as are appropriate, developing and recommending to the Board annual business plans and budgets
that support the Company’s long term business plans and strategies; |
| (e) |
developing and implementing, with senior management of the Company, plans, strategies, budgets and policies necessary to achieve
the goals and objectives of the Company; |
| (f) |
supervising, maintaining and deploying the Company’s resources – human, financial or otherwise – with the purpose
and objective of achieving the Company’s operating goals and objectives; |
| (g) |
keeping the Board informed in a timely and candid manner of the progress of the Company towards the achievement of its strategic
and operational goals and objectives and of all material deviations from the goals, objectives, plans, strategies, budgets or policies
established by the Board; |
| (h) |
overseeing, evaluating and taking steps to enhance, where necessary, the integrity and reliability of the Company’s internal
controls, including its management information systems and financial reporting, and establishing, maintaining, designing and evaluating
disclosure controls and procedures for the Company; |
| (i) |
identifying and managing business risks faced by the Company, including overseeing the design and implementation of appropriate systems
and procedures to effectively monitor, manage and mitigate such risks; |
| (j) |
ensuring that the Board has regular exposure to the Company’s senior management and overseeing the development and succession
of the Company’s senior management team; |
| (k) |
evaluating the performance of senior management of the Company and making recommendations with respect to their compensation;
|
| (l) |
maintaining a positive and ethical work climate that is conducive to attracting, retaining and motivating a diverse group of top-quality
employees at all levels; |
| (m) |
serving as the Company’s principal spokesperson and ensuring that information communicated to the public fairly portrays the
position of the Company and that timely and continuous disclosure obligations of the Company are met; |
| (n) |
representing the Company in a such a way so as to enhance and maintain the Company’s reputation and to promote positive relationships
with shareholders, suppliers, contractors, clients, service providers, strategic partners, creditors, financial institutions, local communities,
all levels of government and the media; and |
| (o) |
fulfilling all other responsibilities as assigned by the Board, in the manner expected by the Board. |
|
Board Diversity Matrix
– IM Cannabis Corp. | ||||||||
|
Country of Principal
Executive Offices |
Israel |
|||||||
|
Foreign Private Issuer
|
Yes | |||||||
|
Disclosure Prohibited
under Home Country Law |
No | |||||||
|
As of March 29, 2023
|
As of March 28, 2024
| |||||||
|
Total Number of Directors
|
5 |
5 | ||||||
|
Gender Identity
|
Female |
Male |
Non-Binary |
Did Not Disclose Gender
|
Female |
Male |
Non-Binary |
Did Not Disclose Gender
|
|
Directors |
1 |
4 |
- |
- |
1 |
4 |
0 |
0 |
|
Demographic Background
| ||||||||
|
Underrepresented Individual in Home Country
Jurisdiction |
0 |
0 | ||||||
|
LGBTQ+ |
0 |
0 | ||||||
|
Did Not Disclose Demographic Background
|
- |
0 | ||||||
|
Name
|
Independence(1)
|
Financial
Literacy(2) |
|
Moti Marcus (Chair)
|
Independent
|
Financially literate
|
|
Brian Schinderle
|
Independent
|
Financially literate
|
|
Einat Zakariya
|
Independent
|
Financially literate
|
| 1. |
Within the meaning of subsection 1.4 of NI 52-110 and as determined under Exchange Act Rule 10A-3 and Rule 5605(a)(2) of the Nasdaq
Stock Market Rules. |
| 2. |
Within the meaning of subsection 1.6 of NI 52-110. |
|
2023 |
2022(5)
| |
|
Audit Fees |
$297 |
$427 |
|
Audit-related Fees(1)
|
$28 |
$61 |
|
Tax Fees(2)(3)
|
$68 |
$81 |
|
All Other Fees(4)
|
$13 |
- |
|
Total |
$406 |
$569 |
| 1. |
Consist of fees for professional services and expenses relating to the audit of the annual financial statements and review of our
quarterly financial information. Fees charged for assurance and related services reasonably related to the performance of an audit, and
not included under “Audit Fees”. |
| 2. |
Consist of fees for professional services and expenses reasonably relating to the audit of the annual financial statements or review
of our quarterly financial information and are not reported as “Audit Fees”. Fees charged for tax compliance, tax advice and
tax planning services. |
| 3. |
Consist of fees for tax-related services related primarily to tax consulting and tax planning. |
| 4. |
Fees for services other than disclosed in any other row, including fees related to the review of management’s discussion and
analysis and Sarbanes-Oxley Act procedures. |
| 5. |
Amounts stated do not include 2022 audit fees of $331 related to deconsolidated Trichome. |
| (i) |
matters of governance; and |
| (ii) |
the nomination of directors to the Board. |
| 1. |
Einat Zakariya (Chair); |
| 2. |
Oren Shuster; and |
| 3. |
Moti Marcus. |
|
Year |
Full Time |
Part Time |
Total | |||
|
Fiscal 2021 |
283 |
- |
363 | |||
|
Fiscal 2022 |
153 |
- |
153 | |||
|
Fiscal 2023 |
95 |
- |
95 |
|
Year |
Israel |
Germany |
Canada |
Total | ||||
|
Fiscal 2021 |
112 |
15 |
236 |
363 | ||||
|
Fiscal 2022 |
126 |
27 |
- |
153 | ||||
|
Fiscal 2023 |
77 |
18 |
- |
95 |
| • |
each person, or group of affiliated persons, known by us to beneficially own five percent (5%) or more of any class of our shares;
|
| • |
each of our Named Executive Officers; |
| • |
each of our directors; and |
| • |
all of our directors and executive officers as a group. |
|
Name of Beneficial Holder
|
Number of Common Shares
Beneficially Held |
Number of Common Shares
Underlying Options |
Option Exercise Price ($) |
Option Expiration Date
|
Restricted Share Units
|
Warrants |
Total Convertible Securities
|
Percentage of Common
Shares Beneficially Held Undiluted |
Percentage of Common
Shares Beneficially Held Partially Diluted |
|
Oren Shuster(1)
|
1,872,870 |
6,250
75,000
50,000 |
40.00
58.70
16.00 |
June 9, 2025 May 19, 2026 January 4, 2029 |
Nil |
856,704 |
987,954 |
13.98% |
19.89% |
|
Marc Lustig(2)
|
930,635 |
67,500 |
16.00 |
November 9, 2029 |
55,000 |
633,860 |
756,360 |
6.95% |
11.92% |
|
Moti Marcus |
Nil |
9,000 |
6.00 |
September 19, 2027 |
Nil |
Nil |
9,000 |
N/A |
0.07% |
|
Einat Zakariya |
61,200 |
9,000 |
6.00 |
September 19, 2027 |
Nil |
Nil |
9,000 |
0.46% |
0.52% |
|
Brian Schinderle |
Nil |
9,000 |
N/A |
N/A |
Nil |
Nil |
9,000 |
N/A |
0.07% |
|
Uri Birenberg |
Nil |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
N/A |
|
Michal Lebovitz Nissimov |
Nil |
3,000 |
1.10 |
May 16, 2028 |
Nil |
Nil |
3,000 |
N/A |
0.02% |
|
Richard Balla |
5,250 |
9,000 |
16.00 |
July 30, 2029 |
Nil |
Nil |
9,000 |
0.04% |
0.11% |
|
Rafael Gabay(3) |
1,173,716 |
Nil |
N/A |
N/A |
Nil |
303,295 |
303,295 |
8.76% |
10.78% |
|
Luminera Derm Ltd.(4) |
757,172 |
Nil |
Nil |
Nil |
Nil |
Nil |
757,172 |
5.65% |
10.70% |
| (5) |
1,872,564 Common Shares and 856,704 Warrants are held by Oren Shuster directly and 153 Common Shares are held indirectly through
Ewave, a privately-held entity of which Mr. Shuster owns and controls 50% of the outstanding voting. |
| (6) |
300,393 Common Shares and 138,118 Warrants are held by Marc Lustig directly and 630,242 Common Shares and 495,742 Warrants are held
indirectly through L5 Capital, a privately held entity of which Mr. Lustig owns and controls 100% of the outstanding voting. |
| (7) |
1,173,563 Common Shares and 303,295 Warrants are held by Rafael Gabay directly and 153 Common Shares are held indirectly by Ewave,
a privately-held entity of which Mr. Gabay owns and controls 50% of the outstanding voting shares. |
| (8) |
Luminera Derm Ltd., has beneficially own 5% of the outstanding voting rights attached to the outstanding Common Shares of the Company starting
from January 20, 2023. |
| • |
On April 2, 2019, IMC Holdings and Focus entered into an option agreement (the “Focus Agreement”) pursuant to which IMC
Holdings acquired an option to purchase, at its sole discretion and in compliance with Israeli cannabis regulation, all of the ordinary
shares held by Messrs. Shuster and Gabay held in Focus at a price equal to NIS 765.67 per ordinary share until April 2029. On November
30, 2023, IMC Holdings sent a request letter to IMCA to approve IMC Holding’s exercise of the option and on February 25, 2024, IMCA's
approval was obtained. Effective February 27, 2024, IMC Holdings acquired 74% of the ordinary shares of Focus. |
| • |
The Company is a party to indemnification agreements with certain directors and officers of the Company and Trichome to cover certain
tax liabilities, interest and penalties arising from the Company’s acquisition of all of issued and outstanding securities of Trichome
and certain of its subsidiaries. |
| • |
The Stalking Horse Purchase Agreement constituted a related party transaction as L5 Capital is an entity controlled by Marc Lustig,
who was a director of Trichome and the Executive Chairman of the Board. On March 8, 2023, the Company announced that the SISP approved
by the Court did not result in any bids for the going-concern business of Trichome; however, L5 Capital advised that it would not complete
the proposed transaction contemplated by the Stalking Horse Share Purchase Agreement. |
| • |
On January 16, 2023, the Company closed of the first tranche of the Concurrent Offering comprised of an aggregate of 1,159,999 Units
for aggregate gross proceeds of US$1,500. The Concurrent Offering was led by insiders of the Company. The units offered under the Concurrent
Offering were sold under similar terms as the Life Offering. |
| • |
On January 20, 2023, the Company closed the second tranche of the LIFE Offering comprised of 102,152 Life Units for an aggregate
subscription price of approximately US$128. The second tranche of the LIFE Offering was comprised of a single subscription by the Executive
Chairman of the Company whose subscription price was satisfied by the settlement of approximately US$128 in debt owed by the Company to
him for certain consulting services previously rendered to the Company. |
| • |
On February 16, 2023, the Company closed the fifth and final tranche of the LIFE Offering. Marc Lustig, the Executive Chairman of
the Company subscribed for 29,548 Life Units in the fifth tranche at an aggregate subscription price of US$37. Marc Lustig’s subscription
price was satisfied by the settlement of US$37 in debt owed by the Company to the director for certain consulting services previously
rendered by the director to the Company. |
| • |
Pursuant to the consulting agreement between the Company and L5 Capital, the Company issued 50,414 Common Shares as a result of the
vested RSUs according to the agreed vesting schedule. The Common Shares were issued on May 5, 2023. In July 24, 2023, an additional 4,585
Common Shares were issued as a result of the vested RSUs according to the agreed vesting schedule. |
| • |
On October 12, 2023, Oren Shuster, the CEO loaned an amount of NIS 500 (approximately $170) to IMC Holdings. The participation of
the CEO constituted a “related party transaction”, as such term is defined in MI 61-101 and would require the Company to receive
minority shareholder approval for and obtain a formal valuation for the subject matter of, the transaction in accordance with MI 61-101,
prior to the completion of such transaction. However, in completing the loan, the Company has relied on exemptions from the formal valuation
and minority shareholder approval requirements of MI 61-101, in each case on the basis that the fair market value of the CEO’s loan
did not exceed 25% of the market capitalization of the Company, as determined in accordance with MI 61-101. |
| 1. |
The contractual party of the company was not Stroakmont. The contract with Stroakmont was only concluded as a sham transaction to
cover up a contract with a company named Uniclaro GmbH (“Uniclaro”). Therefore, Stroakmont
is not the real purchaser rather than Uniclaro. |
| 2. |
The company allegedly placed an order with Uniclaro for a total of 4.3 million Clongene COVID-19 tests, of which Uniclaro claims
to have a payment claim against the company for a partial delivery of 380,400 Clongene COVID-19 tests in the total amount of EUR 942.
Uniclaro has assigned this alleged claim against the company to Stroakmont Trading GmbH, and Stroakmont Trading GmbH has precautionary
declared a set-off against the company’s claim. |
| 1. |
under the age of 18 years; |
| 2. |
found by a court, in Canada or elsewhere, to be incapable of managing the individual’s own affairs, unless a court, in Canada
or elsewhere, subsequently finds otherwise; |
| 3. |
an undischarged bankrupt; or |
| 4. |
convicted in or out of the Province of British Columbia of an offence in connection with the promotion, formation or management of
a corporation or unincorporated business, or of an offence involving fraud, unless: |
| a. |
the court orders otherwise; |
| b. |
5 years have elapsed since the last to occur of: |
| i. |
the expiration of the period set for suspension of the passing of sentence without a sentence having been passed; |
| ii. |
the imposition of a fine; |
| iii. |
the conclusion of the term of any imprisonment; and |
| iv. |
the conclusion of the term of any probation imposed; or |
| c. |
a pardon was granted or issued, or a record suspension ordered, under the Criminal Records Act (Canada) and the pardon or record
suspension, as the case may be, has not been revoked or ceased to have effect. |
| a. |
Focus Agreement. For more information, please see the section entitled “B. Related Party Transactions”. |
| b. |
Services Agreement. For more information, please see the section entitled “Economic Dependence”. |
| c. |
IP Agreement. For more information, please see the section entitled “Economic Dependence”. |
| d. |
First LIFE Warrant Indenture; For more information, please see the section entitled “Life Offering”. |
| e. |
Second LIFE Warrant Indenture; For more information, please see the section entitled “Life Offering”. |
| f. |
Third LIFE Warrant Indenture; For more information, please see the section entitled “Life Offering”. |
| g. |
The Loan Agreement. For more information, please see the section entitled “Potential Reverse Merger with Kadimastem”.
|
| • |
an individual who is a citizen or resident of the U.S.; |
| • |
a corporation (or other entity taxable as a corporation for U.S. federal income tax purposes) organized under the laws of the U.S.,
any state thereof or the District of Columbia; |
| • |
an estate whose income is subject to U.S. federal income taxation regardless of its source; or |
| • |
a trust that (1) is subject to the primary supervision of a court within the U.S. and the control of one or more U.S. persons for
all substantial decisions or (2) has a valid election in effect under applicable Treasury Regulations to be treated as a U.S. person.
|
|
Consolidated Financial
Statements for the Years Ended December 31, 2023 and 2022 |
|
Independent Auditors’ Reports |
|
Consolidated Statements of Financial Position |
|
Consolidated Statements of Net Loss and Comprehensive Loss
|
|
Consolidated Statements of Changes in Shareholders’ Equity
|
|
Consolidated Statements of Cash Flows |
|
Notes to the Consolidated Financial Statements |
|
Description |
Page | |
|
Consolidated Financial Statements and Notes |
F-1 - F-77 |
|
No. Item |
Description of Exhibit
|
| 15.2* | |
|
101.INS* |
XBRL Instant Document |
|
101.SCH* |
XBRL Taxonomy Extension Schema Document
|
|
101.CAL* |
XBLR Taxonomy Extension Calculation Linkbase
Document |
|
101.DEF* |
XBRL Taxonomy Extension Definition Linkbase
|
|
101.LAB* |
XBRL Taxonomy Extension Label Linkbase
|
|
101.PRE* |
XBRL Taxonomy Extension Presentation Linkbase
|
|
104* |
Cover Page Interactive Data File –
(formatted as Inline XBRL and contained in Exhibit 101) |
|
Date: March 28, 2024
|
IM Cannabis Corp.
By: /s/
Uri Birenberg
Name: Uri Birenberg Title: Chief Financial Officer |

|
Page
|
|
|
Report of Independent Registered Public Accounting Firm (PCAOB ID:
|
F-2
|
|
F-3 – F-4
|
|
|
F-5 – F-6
|
|
|
F-7 – F- 8
|
|
|
F-9– F-10
|
|
|
F-11 – F-72
|
 |
Kost Forer Gabbay & Kasierer
144 Menachem Begin Road, Building A,
Tel-Aviv 6492102, Israel
|
Tel: +972-3-6232525
Fax: +972-3-5622555
ey.com
|
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Group is not required to have, nor were we engaged to perform, an audit of its over financial reporting. As part of our audit we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Group’s internal control over financial reporting. Accordingly, we express no such opinion.
|
/s/ KOST FORER GABBAY & KASIERER |
|
|
|
A Member of Ernst & Young Global
|
|
We have served as the Company's auditor since 2018.
|
|
|
|
March 28, 2024
|
|
December 31,
|
|||||||||||
|
Note
|
2023
|
2022
|
|||||||||
|
ASSETS
|
|||||||||||
|
CURRENT ASSETS:
|
|||||||||||
|
Cash and cash equivalents
|
$
|
|
$
|
|
|||||||
|
Trade receivables
|
6
|
|
|
||||||||
|
Advances to suppliers
|
|
|
|||||||||
|
Other accounts receivable
|
7
|
|
|
||||||||
|
Inventory
|
9
|
|
|
||||||||
|
|
|
||||||||||
|
NON-CURRENT ASSETS:
|
|||||||||||
|
Property, plant and equipment, net
|
10
|
|
|
||||||||
|
Investments in affiliates
|
15c
|
|
|
|
|||||||
|
Right-of-use assets, net
|
12
|
|
|
||||||||
|
Deferred tax assets, net
|
17
|
|
|
||||||||
|
Intangible assets, net
|
11
|
|
|
||||||||
|
Goodwill
|
11
|
|
|
||||||||
|
|
|
||||||||||
|
Total assets
|
$
|
|
$
|
|
|||||||
|
December 31,
|
|||||||||||
|
Note
|
2023
|
2022
|
|||||||||
|
LIABILITIES AND EQUITY
|
|||||||||||
|
CURRENT LIABILITIES:
|
|||||||||||
|
Trade payables
|
14
|
$
|
|
$
|
|
||||||
|
Credit from banks and others
|
13
|
|
|
||||||||
|
Other accounts payable and accrued expenses
|
15
|
|
|
||||||||
|
Accrued purchase consideration liabilities
|
5
|
|
|
||||||||
|
PUT Option liability
|
|
||||||||||
|
Current maturities of operating lease liabilities
|
12
|
|
|
||||||||
|
|
|
||||||||||
|
NON-CURRENT LIABILITIES:
|
|||||||||||
|
Warrants measured at fair value
|
17
|
|
|
||||||||
|
Operating lease liabilities
|
12
|
|
|
||||||||
|
Credit from banks and others
|
|
|
|||||||||
|
Employee benefit liabilities, net
|
16
|
|
|
||||||||
|
Deferred tax liability, net
|
19
|
|
|
||||||||
|
|
|
||||||||||
|
Total liabilities
|
|
|
|||||||||
|
EQUITY ATTRIBUTABLE TO EQUITY HOLDERS OF THE COMPANY:
|
20
|
||||||||||
|
Share capital and premium
|
|
|
|||||||||
|
Translation reserve
|
|
|
|||||||||
|
Reserve from share-based payment transactions
|
|
|
|||||||||
|
Accumulated deficit
|
(
|
)
|
(
|
)
|
|||||||
|
Total equity attributable to shareholders of the Company
|
|
|
|||||||||
|
Non-controlling interests
|
(
|
)
|
|
||||||||
|
Total equity
|
|
|
|||||||||
|
Total equity and liabilities
|
$
|
|
$
|
|
|||||||
|
March 28, 2024
|
/s/ Marc Lustig | /s/ Oren Shuster | /s/ Uri Birenberg | |||
|
Date of approval of the
|
Marc Lustig
|
Oren Shuster
|
Uri Birenberg
|
|||
|
financial statements
|
Chairman of the Board
|
Chief Executive Officer
|
Chief Financial Officer
|
|
Year ended December 31,
|
|||||||||||||||
|
Note
|
2023
|
2022
|
*) 2021
|
|
|||||||||||
|
Revenues
|
21
|
$
|
|
$
|
|
$
|
|
||||||||
|
Cost of revenues
|
21
|
|
|
|
|||||||||||
|
Gross profit before fair value adjustments
|
|
|
|
||||||||||||
|
Fair value adjustments:
|
|||||||||||||||
|
Unrealized change in fair value of biological assets
|
|
(
|
)
|
|
|||||||||||
|
Realized fair value adjustments on inventory sold in the year
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Total fair value adjustments
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Gross profit after fair value adjustments
|
|
|
|
||||||||||||
|
General and administrative expenses
|
21
|
|
|
|
|||||||||||
|
Selling and marketing expenses
|
21
|
|
|
|
|||||||||||
|
Restructuring expenses
|
1
|
|
|
|
|||||||||||
|
Share-based compensation
|
20
|
|
|
|
|||||||||||
|
Total operating expenses
|
|
|
|
||||||||||||
|
Operating loss
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Finance income
|
|
|
|
||||||||||||
|
Finance expenses
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Finance income (expense), net
|
|
|
|
||||||||||||
|
Loss before income taxes
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Income tax expense (benefit)
|
18
|
|
(
|
)
|
|
||||||||||
|
Net loss from continuing operations
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Net loss from discontinued operations, net of tax
|
25
|
|
(
|
)
|
(
|
)
|
|||||||||
|
Net loss
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Year ended December 31,
|
|||||||||||||||
|
Note
|
2023
|
2022
|
*) 2021
|
|
|||||||||||
|
Other comprehensive income that will not be reclassified to profit or loss in subsequent periods:
|
|||||||||||||||
|
Remeasurement gain on defined benefit plans
|
|
|
|
||||||||||||
|
Exchange differences on translation to presentation currency
|
(
|
)
|
(
|
)
|
|
||||||||||
|
Total other comprehensive income that will not be reclassified to profit or loss in subsequent periods
|
(
|
)
|
(
|
)
|
|
||||||||||
|
Other comprehensive income that will be reclassified to profit or loss in subsequent periods:
|
|||||||||||||||
|
Adjustments arising from translating financial statements of foreign operation
|
|
(
|
)
|
|
|||||||||||
|
Total other comprehensive income (loss)
|
(
|
)
|
(
|
)
|
|
||||||||||
|
Total comprehensive loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Net loss attributable to:
|
|||||||||||||||
|
Equity holders of the Company
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Non-controlling interests
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||
|
Total comprehensive loss attributable to:
|
|||||||||||||||
|
Equity holders of the Company
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Non-controlling interests
|
$
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||
|
Earnings (loss) per share attributable to equity holders of the Company from continuing operations:
|
22
|
||||||||||||||
|
Basic earnings (loss) per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
|||||||
|
Diluted loss per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Loss per share attributable to equity holders of the Company from discontinued operations:
|
|||||||||||||||
|
Basic and diluted loss per share (in CAD)
|
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||
|
Loss per share attributable to equity holders of the Company from net loss:
|
|||||||||||||||
|
Basic earnings (loss) per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Diluted loss per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
IM CANNABIS CORP. AND ITS SUBSIDIARIES
|
Share capital and premium
|
Treasury Stock
|
Reserve from share-based payment transactions
|
Translation reserve
|
Accumulated deficit
|
Total
|
Non-controlling interests
|
Total
equity
|
|||||||||||||||||||||||||
|
Balance as of January 1, 2021
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
|
$
|
|
|||||||||||||||
|
Net loss
|
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Total other comprehensive income
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Total comprehensive income (loss)
|
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Issuance of common shares, net of issuance costs of $
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Purchase of treasury common shares
|
|
(
|
)
|
|
|
|
(
|
)
|
|
(
|
)
|
|||||||||||||||||||||
|
Exercise of warrants and compensation options
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Exercise of options
|
|
|
(
|
)
|
|
|
|
|
|
|||||||||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Expired options
|
|
|
(
|
)
|
|
|
|
|
|
|||||||||||||||||||||||
|
Balance as of December 31, 2021
|
|
(
|
)
|
|
|
(
|
)
|
|
|
|
||||||||||||||||||||||
|
Net loss
|
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Total other comprehensive income (loss)
|
|
|
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Total comprehensive loss
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||||
|
Issuance of treasury common shares
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Issuance of shares, net of issuance costs of $
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Exercise of options
|
|
|
(
|
)
|
|
|
|
|
|
|||||||||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Expired options
|
|
|
(
|
)
|
|
|
|
|
|
|||||||||||||||||||||||
|
Balance as of December 31, 2022
|
|
|
|
|
(
|
)
|
|
|
|
|||||||||||||||||||||||
|
Share capital and premium*)
|
Reserve from share-based payment transactions
|
Translation reserve
|
Accumulated deficit
|
Total
|
Non-controlling interests
|
Total
equity
|
||||||||||||||||||||||
|
Balance as of December 31, 2022
|
|
|
|
(
|
)
|
|
|
|
||||||||||||||||||||
|
Net loss
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||
|
Total other comprehensive income (loss)
|
|
|
(
|
)
|
|
(
|
)
|
|
(
|
)
|
||||||||||||||||||
|
Total comprehensive loss
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||
|
Issuance of treasury common shares
|
|
|
|
|
|
|
|
|||||||||||||||||||||
|
Issuance of shares, net of issuance costs of $
|
||||||||||||||||||||||||||||
|
Exercise of options
|
||||||||||||||||||||||||||||
|
Other comprehensive income Classification |
( |
) | ( |
) | ( |
) | ( |
) | ||||||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|||||||||||||||||||||
|
Expired options
|
|
(
|
)
|
|
|
|
|
|
||||||||||||||||||||
|
Balance as of December 31, 2023
|
|
|
|
(
|
)
|
|
(
|
)
|
|
|||||||||||||||||||
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Cash provided from operating activities:
|
||||||||||||
|
Net loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Adjustments for non-cash items:
|
||||||||||||
|
Unrealized gain on changes in fair value of biological assets
|
|
(
|
)
|
(
|
)
|
|||||||
|
Fair value adjustment on sale of inventory
|
|
|
|
|||||||||
|
Fair value adjustment on warrants, investments, and accounts receivable
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Depreciation of property, plant and equipment
|
|
|
|
|||||||||
|
Amortization of intangible assets
|
|
|
|
|||||||||
|
Depreciation of right-of-use assets
|
|
|
|
|||||||||
|
Impairment of goodwill
|
|
|
|
|||||||||
|
Impairment of property, plant and equipment
|
|
|
|
|||||||||
|
Impairment of intangible assets
|
|
|
|
|||||||||
|
Impairment of right-of-use assets
|
|
|
|
|||||||||
|
Finance income, net
|
|
|
|
|||||||||
|
Deferred tax payments (benefit), net
|
|
(
|
)
|
|
||||||||
|
Share-based payments
|
|
|
|
|||||||||
|
Share based acquisition costs related to business combination
|
|
|
|
|||||||||
|
Revaluation of other accounts receivable
|
|
|
|
|||||||||
|
Restructuring expenses
|
|
|
|
|||||||||
|
Loss from revaluation of investments
|
|
|
|
|||||||||
|
|
|
(
|
)
|
|||||||||
|
Changes in non-cash working capital:
|
||||||||||||
|
Increase (decrease) in trade receivables, net
|
|
|
(
|
)
|
||||||||
|
Increase (decrease) in other accounts receivable and advances to suppliers
|
|
|
|
|||||||||
|
Decrease in biological assets, net of fair value adjustments
|
|
|
|
|||||||||
|
Increase (decrease) in inventory, net of fair value adjustments
|
|
|
(
|
)
|
||||||||
|
Increase (decrease) in trade payables
|
(
|
)
|
|
|
||||||||
|
Changes in employee benefit liabilities, net
|
(
|
)
|
(
|
)
|
|
|||||||
|
Increase in other accounts payable and accrued expenses
|
(
|
)
|
|
|
||||||||
|
|
|
(
|
)
|
|||||||||
|
Taxes paid
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Net cash used in operating activities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Cash flows from investing activities:
|
||||||||||||
|
Purchase of property, plant and equipment
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds from sales of property, plant and equipment
|
|
|
|
|||||||||
|
Proceeds from loans receivable
|
|
|
|
|||||||||
|
Purchase of intangible assets
|
|
|
(
|
)
|
||||||||
|
Acquisition of businesses, net of cash acquired
|
|
|
(
|
)
|
||||||||
|
Deconsolidation of subsidiary (see Note 25)
|
|
(
|
)
|
|
||||||||
|
Investments in financial assets
|
|
|
(
|
)
|
||||||||
|
Proceeds from sale of investment
|
|
|
|
|||||||||
|
Proceeds from (investment in) restricted deposits
|
|
|
|
|||||||||
|
Investments in associates
|
(
|
)
|
(
|
)
|
|
|||||||
|
Net cash used in investing activities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Cash provided by financing activities:
|
||||||||||||
|
Proceeds from issuance of share capital, net of issuance costs
|
|
|
|
|||||||||
|
Proceeds from issuance of warrants measured at fair value
|
|
|
|
|||||||||
|
Proceeds from exercise of warrants
|
|
|
|
|||||||||
|
Proceeds from exercise of options
|
|
|
|
|||||||||
|
Repayment of lease liability
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Payment of lease liability interest
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds from loans
|
|
|
|
|||||||||
|
Repayment of loans
|
(
|
)
|
(
|
)
|
|
|||||||
|
Interest paid
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds from discounted checks
|
|
|
|
|||||||||
|
Net cash provided by financing activities
|
|
|
|
|||||||||
|
Effect of foreign exchange on cash and cash equivalents
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Increase (decrease) in cash and cash equivalents
|
(
|
)
|
(
|
)
|
|
|||||||
|
Cash and cash equivalents at beginning of year
|
|
|
|
|||||||||
|
Cash and cash equivalents at end of year
|
$
|
|
$
|
|
$
|
|
||||||
|
Supplemental disclosure of non-cash activities:
|
||||||||||||
|
Right-of-use asset recognized with corresponding lease liability
|
$
|
|
$
|
|
$
|
|
||||||
|
Conversion of warrant and compensation options into common shares
|
$
|
|
$
|
|
$
|
|
||||||
|
Issuance of shares in payment of purchase consideration liability
|
$
|
|
$
|
|
$
|
|
||||||
|
Issuance of shares in payment of debt settlement to a non-independent director of the company
|
$
|
|
$
|
|
$
|
|
||||||
IM CANNABIS CORP. AND ITS SUBSIDIARIES
| NOTE 1:- |
GENERAL
|
| a. |
Corporate information:
|
| NOTE 1:- |
GENERAL (Cont.)
|
As of December 31, 2023, the Group's cash and cash equivalents totaled $
| NOTE 1:- |
GENERAL (Cont.)
|
| NOTE 1:- |
GENERAL (Cont.)
|
| NOTE 1:- |
GENERAL (Cont.)
|
| NOTE 1:- |
GENERAL (Cont.)
|
| b. |
Approval of consolidated financial statements:
|
| c. |
Definitions:
|
|
The Company, or IMCC
|
-
|
IM Cannabis Corp.
|
|
The Group
|
-
|
IM Cannabis Corp., its Subsidiaries
|
|
Subsidiaries
|
-
|
Companies that are controlled by the Company (as defined in IFRS 10) and whose accounts are consolidated with those of the Company
|
|
CAD or $
|
-
|
Canadian Dollar
|
|
NIS
|
-
|
New Israeli Shekel
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES |
| a. |
Basis of presentation:
|
| - |
Financial instruments which are presented at fair value through profit or loss.
|
| - |
Biological assets which are presented at fair value less cost to sell up to the point of harvest.
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| b. |
Consolidated financial statements:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
|
Percentage ownership
|
||||||||
|
Subsidiaries
|
2023
|
2022
|
||||||
|
I.M.C. Holdings Ltd. (”IMC”)
|
|
%
|
|
%
|
||||
|
Focus Medical Herbs Ltd. ("Focus") *)
|
|
%
|
|
%
|
||||
|
I.M.C. Pharma Ltd.
|
|
%
|
|
%
|
||||
|
I.M.C.C. Medical Herbs Ltd. ***)
|
|
%
|
|
%
|
||||
|
I.M.C Farms Israel Ltd. ("IMC Farms") ***)
|
|
%
|
|
%
|
||||
|
I.M.C Ventures Ltd. ("IMC Ventures") ***)
|
|
%
|
|
%
|
||||
|
I.M.C - International Medical Cannabis Portugal Unipessoal Lda) ***)
|
|
|
||||||
|
Adjupharm GmbH (“Adjupharm”)
|
|
%
|
|
%
|
||||
|
R.A. Yarok Pharm Ltd. (“Pharm Yarok”)
|
|
%
|
|
%
|
||||
|
Rosen High Way Ltd. (“Rosen High Way”)
|
|
%
|
|
%
|
||||
|
High Way Shinua Ltd. (“HW Shinua”)
|
|
%
|
|
%
|
||||
|
Revoly Trading and Marketing Ltd. (“Vironna”)
|
|
%
|
|
%
|
||||
|
Oranim Plus Pharm LTD.
|
|
%
|
|
%
|
||||
|
Oranim Pharm
|
|
%
|
|
%
|
||||
|
Trichome Financial Corp. (“Trichome”)
|
)
|
)
|
||||||
|
Trichome Financial Cannabis GP Inc.
|
)
|
)
|
||||||
|
Trichome Financial Cannabis Manager Inc.
|
)
|
)
|
||||||
|
Trichome Asset Funding Corp.
|
)
|
)
|
||||||
|
Trichome JWC Acquisition Corp. (“TJAC”)
|
)
|
)
|
||||||
|
Trichome Retail Corp.
|
)
|
)
|
||||||
|
MYM Nutraceuticals Inc. (“MYM”)
|
)
|
)
|
||||||
|
SublimeCulture Inc.
|
)
|
)
|
||||||
|
CannaCanada Inc.
|
)
|
)
|
||||||
|
MYM International Brands Inc.
|
)
|
)
|
||||||
|
Highland Grow Inc.
|
)
|
)
|
||||||
| *) |
The Company does not hold directly interest or voting rights in Focus. The Company's wholly-owned subsidiary holds an option to buy the ownership of the
|
| **) |
Deconsolidated effective November 7, 2022, when Trichome filed to commence proceedings under the Companies’ Creditors Arrangement Act (CCAA) (see Note 1).
|
| ***) |
Dissolved as of December 31, 2023.
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| c. |
Business combinations and goodwill:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| d. |
Functional currency, presentation currency and foreign currency:
|
| 1. |
Functional currency and presentation currency:
|
| 2. |
Transactions, assets and liabilities in foreign currency:
|
| e. |
Cash equivalents:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| f. |
Inventories:
|
| g. |
Biological assets:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| h. |
Revenue recognition:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| a) |
The reason for the bill-and-hold arrangement is substantive (for example, the customer has requested the arrangement);
|
| b) |
The product is identified separately as belonging to the customer;
|
| c) |
The product currently is ready for physical delivery to the customer;
|
| d) |
The Group does not have the ability to use the product by selling it or delivering it to another customer.
|
| i. |
Taxes on income:
|
| 1. |
Current taxes:
|
| 2. |
Deferred taxes:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| j. |
Non-current assets or disposal group held for sale and discontinued operations:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| k. |
Leases:
|
|
NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| l. |
Property, plant and equipment, net:
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
|
%
|
Mainly %
|
|||||
|
Buildings
|
|
|
||||
|
Greenhouse production equipment
|
|
|
||||
|
Greenhouse structure
|
|
|
||||
|
Motor vehicles
|
|
|
||||
|
Computer, software and equipment
|
|
|
||||
|
Leasehold improvements
|
See below
|
See below
|
| m. |
Intangible assets:
|
|
Years
|
|||
|
Customer relationship
|
|
||
|
Brand name
|
|
||
|
Other intangibles
|
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| n. |
Impairment of non-financial assets:
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| o. |
Financial instruments:
|
| 1. |
Financial assets:
|
| - |
The Group’s business model for managing financial assets; and
|
| - |
The contractual cash flow terms of the financial asset.
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| 2. |
Financial liabilities:
|
| 3. |
Issue of a unit of securities:
|
| 4. |
Put option granted to non-controlling interests:
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| p. |
Fair value measurement:
|
|
Level 1
|
-
|
quoted prices (unadjusted) in active markets for identical assets or liabilities.
|
|
Level 2
|
-
|
inputs other than quoted prices included within Level 1 that are observable directly or indirectly.
|
|
Level 3
|
-
|
inputs that are not based on observable market data (valuation techniques which use inputs that are not based on observable market data).
|
| q. |
Provisions:
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| r. |
Share-based payment transactions:
|
| s. |
Earnings (loss) per share:
|
| NOTE 2:- |
SIGNIFICANT ACCOUNTING POLICIES (Cont.)
|
| 1. |
Amendment to IAS 1, "Disclosure of Accounting Policies":
|
| 2. |
Amendment to IAS 12, "Income Taxes":
|
| NOTE 3:- |
SIGNIFICANT ACCOUNTING JUDGMENTS, ESTIMATES AND ASSUMPTIONS USED IN THE PREPARATION OF THE FINANCIAL STATEMENTS
|
| a. |
Judgments:
|
| - |
Determining the fair value of share-based payment transactions:
|
| - |
Discount rate for a lease liability:
|
| b. |
Estimates and assumptions:
|
| - |
Legal claims:
|
| NOTE 3:- |
SIGNIFICANT ACCOUNTING ESTIMATES AND ASSUMPTIONS USED IN THE PREPARATION OF THE FINANCIAL STATEMENTS (Cont.)
|
| - |
Deferred tax assets:
|
| - |
Impairment of goodwill:
|
| - |
Determining the fair value of an unquoted financial assets and liabilities:
|
| - |
Loss of control of subsidiary:
On November 7, 2022, Trichome filed a petition with the Superior Court of Ontario for protection under the Companies’ Creditors Arrangement Act (“CCAA”) in order to restructure its business and financial affairs. Management applied judgement in assessing whether this event represented a loss of control of Trichome. On filing of CCAA, which included a request for an order to approve a sale and investment solicitation process and to approve a stalking horse agreement of purchase and sale, management concluded that the Company ceased to have the power to direct the relevant activity of Trichome because substantive rights were granted to other parties through the CCAA proceedings that restricted the decision making ability of the Company to the extent that the Company was unable to demonstrate power over Trichome. As a result, the Company accounted for a loss in control and Trichome was deconsolidated on November 17, 2022 (see Note 1 and Note 25).
|
| NOTE 4:- |
DISCLOSURE OF NEW STANDARDS IN THE PERIOD PRIOR TO THEIR ADOPTION
|
| a. |
Amendments to IAS 21, "The Effects of Changes in Foreign Exchange Rates":
|
| NOTE 5:- |
BUSINESS COMBINATIONS
|
| NOTE 6:- |
TRADE RECEIVABLES
|
| NOTE 7:- |
OTHER ACCOUNTS RECEIVABLE
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Prepaid expenses
|
$
|
|
$
|
|
||||
|
Government authorities
|
|
|
||||||
|
Related parties (see Note 24)
|
|
|
||||||
|
Non-independent director – L5 Capital (See note 1)
|
|
|
||||||
|
Other receivables
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 8:- |
BIOLOGICAL ASSETS
|
|
Balance at of January 1, 2022
|
$
|
|
||
|
Production costs capitalized
|
|
|||
|
Changes in fair value less cost to sell due to biological transformation
|
|
|||
|
Transferred to inventory upon harvest
|
(
|
)
|
||
|
Restructuring disposal
|
(
|
)
|
||
|
Foreign exchange translation
|
|
|||
|
Deconsolidation of Trichome (see Note 25)
|
(
|
)
|
||
|
Balance at of December 31, 2022
|
|
|||
|
Balance at of December 31, 2023
|
|
| 1. |
Selling price per gram - calculated as the weighted average historical selling price for all strains of cannabis sold by the Group, which is expected to approximate future selling prices.
|
| 2. |
Post-harvest costs - calculated as the cost per gram of harvested cannabis to complete the sale of cannabis plants post-harvest, consisting of the cost of direct and indirect materials, depreciation and labor as well as labelling and packaging costs.
|
| NOTE 8:- |
BIOLOGICAL ASSETS (Cont.)
|
| 3. |
Attrition rate - represents the weighted average percentage of biological assets which are expected to fail to mature into cannabis plants that can be harvested.
|
| 4. |
Average yield per plant - represents the expected number of grams of finished cannabis inventory which are expected to be obtained from each harvested cannabis plant.
|
| 5. |
Stage of growth - represents the weighted average number of weeks out of the average weeks growing cycle that biological assets have reached as of the measurement date. The growing cycle is approximately 12 weeks.
|
|
December 31,
|
10% change as at
December 31,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Average selling price per gram of dried cannabis (in CAD)
|
|
$
|
|
|
$
|
|
||||||||||
|
Average post-harvest costs per gram of dried cannabis (in CAD)
|
|
$
|
|
|
$
|
|
||||||||||
|
Attrition rate
|
|
|
%
|
|
|
%
|
||||||||||
|
Average yield per plant (in grams)
|
|
|
|
|
||||||||||||
|
Average stage of growth
|
|
|
%
|
|
|
%
|
||||||||||
| NOTE 9:- |
INVENTORY
|
|
December 31, 2023
|
||||||||||||
|
Capitalized costs
|
Fair valuation adjustment, net
|
Carrying value
|
||||||||||
|
Work in progress:
|
||||||||||||
|
Bulk cannabis
|
$
|
|
$
|
|
$
|
|
||||||
|
Finished goods:
|
||||||||||||
|
Packaged dried cannabis
|
|
|
|
|||||||||
|
Other products
|
|
|
|
|||||||||
|
Balance as of December 31, 2023
|
|
|
|
|||||||||
|
December 31, 2022
|
||||||||||||
|
Capitalized costs
|
Fair valuation adjustment, net
|
Carrying value
|
||||||||||
|
Work in progress:
|
||||||||||||
|
Bulk cannabis
|
$
|
|
$
|
|
$
|
|
||||||
|
Finished goods:
|
||||||||||||
|
Packaged dried cannabis
|
|
|
|
|||||||||
|
Other products
|
|
|
|
|||||||||
|
Balance as of December 31, 2022
|
$
|
|
$
|
|
$
|
|
||||||
| NOTE 10:- |
PROPERTY, PLANT AND EQUIPMENT, NET
|
|
Buildings and improvements
|
Production equipment and furniture
|
Greenhouse structure
|
Computer, software and equipment
|
Motor vehicles
|
Total
|
|||||||||||||||||||
|
Cost:
|
||||||||||||||||||||||||
|
Balance at January 1, 2022
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
Additions during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||
|
Foreign currency translation
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||
|
Balance at December 31, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
Additions during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Foreign currency translation
|
(
|
)
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|
||||||||||||||||||
|
Accumulated depreciation:
|
||||||||||||||||||||||||
|
Balance at January 1, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
Depreciation during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Impairment
|
|
|
|
|
|
|
||||||||||||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||
|
Foreign currency translation
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||
|
Balance at December 31, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
Depreciation during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Foreign currency translation
|
(
|
)
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|
||||||||||||||||||
|
Depreciated cost at December 31, 2023
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
Depreciated cost at December 31, 2022
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
| NOTE 11:- |
GOODWILL AND INTANGIBLE ASSETS, NET
|
|
Cultivations and processing license *)
|
Customer relationships
|
Brand
|
Goodwill
|
Other
|
Total
|
|||||||||||||||||||
|
Cost:
|
||||||||||||||||||||||||
|
Balance at January 1, 2022
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
PPA adjustments during measurement period
|
|
|
|
(
|
)
|
|
|
|||||||||||||||||
|
Disposals
|
(
|
)
|
|
|
|
|
(
|
)
|
||||||||||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
(
|
)
|
|
(
|
)
|
(
|
)
|
|||||||||||||
|
Foreign currency translation adjustments
|
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||
|
Balance at December 31, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
PPA adjustments during measurement period
|
|
|
|
|
|
|
||||||||||||||||||
|
Disposals
|
|
(
|
)
|
|
|
|
(
|
)
|
||||||||||||||||
|
Foreign currency translation adjustments
|
|
(
|
)
|
|
|
|
|
(
|
)
|
|||||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|
||||||||||||||||||
|
Accumulated amortization:
|
||||||||||||||||||||||||
|
Balance at January 1, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortization recognized in the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Impairment
|
|
|
|
|
|
|
||||||||||||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
|
|
(
|
)
|
(
|
)
|
||||||||||||||
|
Balance at December 31, 2022
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortization recognized in the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortized cost at December 31, 2023
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
Amortized cost at December 31, 2022
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
| *) |
The licenses consist of GMP and GDP licenses.
|
| NOTE 11:- |
GOODWILL AND INTANGIBLE ASSETS, NET (Cont.)
|
For the year ended December 31, 2023, the Company did not recorded a goodwill and intangible assets impairment.
| NOTE 12:- |
RIGHT-OF-USE ASSETS
|
|
buildings
|
Motor vehicles
|
Total
|
||||||||||
|
Cost:
|
||||||||||||
|
Balance at January 1, 2022
|
$
|
|
$
|
|
$
|
|
||||||
|
Additions during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Disposals during the year
|
(
|
)
|
|
(
|
)
|
|||||||
|
Termination of leases
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Currency translation adjustments
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Balance at December 31, 2022
|
|
|
|
|||||||||
|
Additions during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Balance at December 31, 2023
|
|
|
|
|||||||||
|
Accumulated depreciation:
|
||||||||||||
|
Balance at January 1, 2022
|
|
|
|
|||||||||
|
Additions during the year:
|
||||||||||||
|
Depreciation and amortization
|
|
|
|
|||||||||
|
Termination of leases
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Impairment
|
|
|
|
|||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Currency translation adjustments
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Balance at December 31, 2022
|
|
|
|
|||||||||
|
Additions during the year:
|
||||||||||||
|
Depreciation and amortization
|
|
|
|
|||||||||
|
Currency translation adjustments
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Balance at December 31, 2023
|
|
|
|
|||||||||
|
Depreciated cost at December 31, 2023
|
|
|
|
|||||||||
|
Depreciated cost at December 31, 2022
|
$
|
|
$
|
|
$
|
|
||||||
| NOTE 13:- |
CREDIT FROM BANKS AND OTHERS
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Short-term credit from banks
|
$
|
|
$
|
|
||||
|
Short-term credit from others
|
|
|
||||||
|
Check receivables
|
|
|||||||
|
$
|
|
$
|
|
|||||
| 1. |
In January 2022, Focus entered into a revolving credit facility with an Israeli bank, Bank Mizrahi (the “Mizrahi Facility”). The Mizrahi Facility is guaranteed by Focus assets. Advances from the Mizrahi Facility will be used for working capital needs. The Mizrahi Facility has a total commitment of up to NIS
|
| NOTE 13:- |
CREDIT FROM BANKS AND OTHERS (Cont.)
|
| 2. |
On October 11, 2022, IMC Holdings entered into a loan agreement with A.D.I. Car Alarms Stereo Systems Ltd (“ADI” and the “ADI Agreement”), to borrow a principal amount of NIS
|
| 3. |
On July 3rd, 2023, the Company entered into a short-term loan agreement with a non-financial institution in the amount of NIS
|
| 4. |
During October 2023, the Company entered into a series of short-term loans for aggregate gross proceeds of NIS
|
| NOTE 14:- |
TRADE PAYABLES
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Open accounts in Israel
|
$
|
|
$
|
|
||||
|
Open accounts abroad
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 15:- |
OTHER PAYABLES AND ACCRUED EXPENSES
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Accrued expenses
|
$
|
|
$
|
|
||||
|
Employees and payroll accruals
|
|
|
||||||
|
Government authorities
|
|
|
||||||
|
Related parties
|
|
|
||||||
|
Advances from customers
|
|
|
||||||
|
Liabilities for restructuring
|
|
|
||||||
|
Other payables
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 16:- |
EMPLOYEE BENEFIT LIABILITIES, NET
|
|
NOTE 17:- |
FINANCIAL INSTRUMENTS
|
| a. |
Management believes that the carrying amount of cash and cash equivalents, trade receivables, other accounts receivable, loans receivables, trade payables, bank loans, other account payables and accrued expenses and purchase consideration payable and approximate their fair value due to the short-term maturities of these instruments.
|
| b. |
For the years ended December 31, 2023, 2022 and 2021, the Company recognized a revaluation gain (loss) from remeasurement of warrants in the consolidated statement of profit or loss and other comprehensive income, which unrealized gain is included in finance income (expense). (See also note 20f).
|
| c. |
On December 26, 2019, IMC entered into a share purchase agreement (the “SPA”) with Xinteza API Ltd. ("Xinteza"), a company with a unique biosynthesis technology.
|
| NOTE 17:- |
FINANCIAL INSTRUMENTS (Cont.)
|
| d. |
Financial risk management:
|
|
Less than one year
|
1 to 5 years
|
6 to 10
years
|
>10
years
|
|||||||||||||
|
Lease liabilities
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Bank loans and others
|
|
|
|
|
||||||||||||
|
Total
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Less than one year
|
1 to 5 years
|
6 to 10
years
|
>10
years
|
|||||||||||||
|
Lease liabilities
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Bank loans and others
|
|
|
|
|
||||||||||||
|
Total
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
| NOTE 17:- |
FINANCIAL INSTRUMENTS (Cont.)
|
| e. |
On November 29, 2022, the IMC signed on a convertible loan agreement with Telekana Ltd. (“Telekana”), a Pharmacy for sell of medical Cannabis accordingly IMC will loan a total of $
|
| f. |
Changes in liabilities arising from financing activities:
|
|
Loans
|
Lease liabilities
|
Warrants
|
Total liabilities arising from financing activities
|
|||||||||||||
|
Balance as of January 1, 2022
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Additions for new loans
|
|
|
|
|
||||||||||||
|
Additions for new leases
|
|
|
|
|
||||||||||||
|
Repayments
|
|
(
|
)
|
|
(
|
)
|
||||||||||
|
Effective interest
|
|
|
|
|
||||||||||||
|
Effect of exchange rate differences
|
(
|
)
|
(
|
)
|
|
(
|
)
|
|||||||||
|
Deconsolidation of Trichome
|
(
|
)
|
(
|
)
|
|
(
|
)
|
|||||||||
|
Effect of changes in fair value
|
|
|
(
|
)
|
(
|
)
|
||||||||||
|
Balance as of December 31, 2022
|
|
|
|
|
||||||||||||
|
Additions for new loans
|
|
|
|
|
||||||||||||
|
Additions for new leases
|
|
|
|
|
||||||||||||
|
Receivables checks
|
|
|
|
|
||||||||||||
|
Repayments
|
|
(
|
)
|
|
(
|
)
|
||||||||||
|
Effective interest
|
|
|
|
|
|
|
||||||||||
|
Effect of exchange rate differences
|
(
|
)
|
(
|
)
|
|
|
||||||||||
|
Deconsolidation of Trichome
|
||||||||||||||||
|
Effect of changes in fair value
|
|
|
(
|
)
|
(
|
)
|
||||||||||
|
Balance as of December 31, 2023
|
|
|
|
|
||||||||||||
| NOTE 18:- |
CONTINGENT LIABILITIES, GUARANTEES, COMMITMENTS AND CHARGES
|
| a. |
On August 19, 2019, a cannabis consumer (the “Applicant”) filed a motion for approval of a class action to Tel Aviv - Jaffa District Court (the “Motion”) against 17 companies (the “Parties”) operating in the field of medical cannabis in Israel, including Focus. The Applicant’s argument is that the Parties did not accurately mark the concentration of active ingredients in their products. The personal suit sum for each class member stands at NIS
Due to the current preliminary state of the litigation process and based on the opinion of legal counsel to Focus, the Company’s management believes that it is not reasonably possible to assess the outcome of the proceeding. Therefore, no provision has been recorded in respect thereof.
|
| b. |
On July 11, 2021 the Company was informed that on June 30, 2021, a claim was filed to Beer Sheva Magistrate Court, by the municipal committee presiding over planning and construction in southern Israel against Focus, Focus’ directors and officers, including Oren Shuster and Rafael Gabay, and certain landowners, claiming for inadequate permitting for construction relating to the Focus Facility (the “Construction Proceedings”).
On December 6, 2021 the defendants filed a motion request for dismissal the indictment on the ground of defense of justice. The municipal committee filed its response and after that the defendants filed a response to the municipal committee’s response. As of the date of this letter no decision has yet been made on the application.
|
| NOTE 18:- |
CONTINGENT LIABILITIES, GUARANTEES, COMMITMENTS AND CHARGES (Cont.)
|
| c. |
On November 19, 2021, Adjupharm filed a statement of claim (the “Claim”) to the District Court of Stuttgart (the “Stuttgart Court”) against Stroakmont & Atton Trading GmbH (“Stroakmont & Atton”), its shareholders and managing directors regarding a debt owed by Stroakmont & Atton to Adjupharm in an amount of approximately EUR
|
| 1. |
The contractual party of the Company was not the Stroakmont & Atton. The contract with Stroakmont & Atton was only concluded as a sham transaction in order to cover up a contract with a company named Uniclaro. Therefore, Stroakmont & Atton is not the real purchaser rather than Uniclaro GmbH.
|
| 2. |
The Company allegedly placed an order with Uniclaro GmbH for a total of
|
| NOTE 18:- |
CONTINGENT LIABILITIES, GUARANTEES, COMMITMENTS AND CHARGES (Cont.)
|
| NOTE 18:- |
CONTINGENT LIABILITIES, GUARANTEES, COMMITMENTS AND CHARGES (Cont.)
|
|
According to Uniclaro's statement of claim the lawsuit does not concern the same purchase price and the same Covid-19 rapid tests as in the Stroakmont & Atton Claim. On February 23, 2023, the Company provided its statement of defense to the court. The statement of defense contains similar arguments to reject the allegations in this respect as in the court proceedings in Stuttgart about the counterclaims. As a next step, Uniclaro is allowed to respond to the Company's statement of defense.
At this stage, the Company management cannot assess the chances of the claim advancing or the potential outcome of these proceedings. Therefore, no provision has been recorded in respect of this claim.
|
| NOTE 19:- |
TAXES ON INCOME
|
| a. |
Tax rates applicable to the Group:
|
| 1. |
The Company is subject to tax rates applicable in Canada. The combined federal and provincial rate for 2023 and 2022 is
|
| 2. |
The Israeli subsidiaries are subject to Israeli corporate income tax rate of
|
| 3. |
The German subsidiary is subject to weighted tax rate of approximately
|
| b. |
Carryforward losses for tax purposes:
Carryforward operating tax losses of the Israeli subsidiaries total approximately $
Carryforward operating tax losses of the German subsidiary as of December 31, 2023, amounted to approximately $
|
| NOTE 19:- |
TAXES ON INCOME (Cont.)
|
| c. | Income tax expense (benefit): | |
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Current
|
$
|
|
$
|
|
$
|
|
||||||
|
Deferred, net
|
|
(
|
)
|
|
||||||||
|
Income tax from previous years
|
|
(
|
)
|
(
|
)
|
|||||||
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||
| d. | Deferred taxes: | |
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Deferred tax assets:
|
||||||||
|
Carryforward tax losses and other
|
$
|
|
$
|
|
||||
|
Other deferred tax assets
|
|
|
||||||
|
|
|
|||||||
|
Deferred tax liabilities:
|
||||||||
|
Intangible assets
|
|
|
||||||
|
Other
|
|
|
||||||
|
|
|
|||||||
|
Deferred tax liabilities, net
|
$
|
(
|
)
|
$
|
(
|
)
|
||
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Non-current assets
|
$
|
|
$
|
|
||||
|
Non-current liabilities
|
$
|
|
$
|
|
||||
| NOTE 19:- |
TAXES ON INCOME (Cont.)
|
| e. |
Reconciliation of tax expense (benefit) and the accounting loss multiplied by the Company's domestic tax rate for:
|
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Loss before income tax
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Statutory tax rate in Canada
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Increase (decrease) in income tax due to:
|
||||||||||||
|
Non-deductible expenses (non-taxable income), net for tax purposes
|
(
|
)
|
|
(
|
)
|
|||||||
|
Effect of different tax rate of subsidiaries
|
|
|
|
|||||||||
|
Adjustments in respect of current income tax of previous years
|
|
(
|
)
|
(
|
)
|
|||||||
|
Recognition (derecognition) of tax benefit in respect of losses of previous years
|
|
|
|
|||||||||
|
Unrecognized tax benefit in respect of loss for the year
|
|
|
|
|||||||||
|
Other adjustments
|
|
(
|
)
|
(
|
)
|
|||||||
|
Income tax expense (benefit)
|
$
|
|
$
|
(
|
)
|
$
|
|
|||||
| NOTE 20:- |
EQUITY
|
| a. |
Composition of share capital:
|
|
December 31, 2023
|
December 31, 2022
|
|||||||||
|
Authorized
|
Issued and outstanding
|
Authorized
|
Issued and outstanding
|
|||||||
|
Number of shares
|
||||||||||
|
Common Shares without par value
|
Unlimited
|
|
Unlimited
|
|
||||||
| NOTE 20:- |
EQUITY (Cont.)
|
| b. |
Capital issuances:
|
| NOTE 20:- |
EQUITY (Cont.)
|
| NOTE 20:- |
EQUITY (Cont.)
|
| c. |
Changes in issued and outstanding share capital:
|
|
Number of shares
|
||||
|
Balance as of January 1, 2022
|
|
|||
|
Common shares issued as a result of options exercises
|
|
|||
|
Common shares issued in settlement of purchase consideration of business combination
|
|
|||
|
Issuance of treasury Common shares
|
|
|||
|
Issuance of Common shares
|
|
|||
|
Balance as of December 31, 2022
|
|
|||
|
Issuance of Common Shares pursuant to debt settlement
|
|
|||
|
Issuance of Common Shares pursuant to vested RSUs
|
|
|||
|
Issuance of Common shares
|
|
|||
|
Balance as of December 31, 2023
|
|
|||
| NOTE 20:- |
EQUITY (Cont.)
|
| d. |
Share option plan:
|
|
Year ended
December 31,
|
||||||
|
2023
|
2022
|
|||||
|
Exercise price (in CAD)
|
$
|
$
|
||||
|
Dividend yield (%)
|
|
|
||||
|
Expected life of share options (years)
|
|
|
||||
|
Volatility (%)
|
|
|
||||
|
Annual risk-free rate (%)
|
|
|
||||
|
Share price (in CAD)
|
$
|
$
|
||||
|
Year ended December 31, 2023
|
||||||||
|
Number of options
|
Weighted average exercise price
|
|||||||
|
in CAD
|
||||||||
|
Options outstanding at the beginning of the year
|
|
|
||||||
|
Options granted during the year
|
|
|
||||||
|
Options exercised during the year
|
|
|
||||||
|
Options forfeited during the year
|
(
|
)
|
(
|
) | ||||
|
Options outstanding at the end of year
|
|
|
||||||
|
Options exercisable at the end of year
|
|
|
||||||
| NOTE 20:- |
EQUITY (Cont.)
|
|
Year ended December 31, 2022
|
||||||||
|
Number of options
|
Weighted average exercise price
|
|||||||
|
in CAD
|
||||||||
|
Options outstanding at the beginning of the year
|
|
|
||||||
|
Options granted during the year
|
|
|
||||||
|
Options exercised during the year
|
(
|
)
|
|
|||||
|
Options forfeited during the year
|
(
|
)
|
|
|||||
|
Options outstanding at the end of year
|
|
|
||||||
|
Options exercisable at the end of year
|
|
|
||||||
|
Number of RSU
|
||||
|
Outstanding at the beginning of the year
|
|
|||
|
Granted during the year
|
|
|||
|
Outstanding at the end of the year
|
|
|||
|
Exercisable at the end of year
|
|
|||
| e. |
Other convertible securities:
|
| NOTE 20:- |
EQUITY (Cont.)
|
| f. |
On May 10, 2021, the Company completed an overnight marketed offering (the “Offering”) of
|
|
Issue date
|
||||||||||||
|
May 2023
|
February 2023
|
May 2021
|
||||||||||
|
Expected volatility
|
|
%
|
|
%
|
|
%
|
||||||
|
Share price (Canadian Dollar)
|
|
|
|
|||||||||
|
Expected life (in years)
|
|
|
|
|||||||||
|
Risk-free interest rate
|
|
%
|
|
%
|
|
%
|
||||||
|
Expected dividend yield
|
|
%
|
|
%
|
|
%
|
||||||
|
Fair value:
|
||||||||||||
|
Per Warrant (Canadian Dollar)
|
$
|
|
$
|
|
$
|
|
||||||
|
Total Warrants (Canadian Dollar in thousands)
|
$
|
|
$
|
|
$
|
|
||||||
| NOTE 21:- |
ADDITIONAL INFORMATION TO PROFIT OR LOSS ITEMS
|
| a. |
Additional information on revenues:
|
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Israel
|
|
|
|
|||||||||
|
Foreign countries
|
|
|
|
|||||||||
|
|
|
|
||||||||||
| b. |
Cost of sales and services:
|
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Salaries
|
|
|
|
|||||||||
|
Materials
|
|
|
|
|||||||||
|
Professional fees
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Others Cost of sales expenses
|
|
|
|
|||||||||
|
|
|
|
||||||||||
| c. |
Selling and marketing expenses:
|
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Salaries
|
|
|
|
|||||||||
|
Selling and Marketing
|
|
|
|
|||||||||
|
Professional fees
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Other Selling and marketing expenses
|
|
|
|
|||||||||
|
|
|
|
||||||||||
| d. |
General and administrative expenses:
|
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Salaries
|
|
|
|
|||||||||
|
Insurance
|
|
|
|
|||||||||
|
Professional fees
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Impairment
|
|
|
|
|||||||||
|
Other General and Administration
|
|
|
|
|||||||||
|
|
|
|
||||||||||
| NOTE 22:- |
NET LOSS PER SHARE
|
|
Year ended December 31,
|
||||||||||||||||
|
2023
|
2022
|
|||||||||||||||
|
Weighted number of shares (in thousands)
|
Net loss attributable to equity holders of the Company
|
Weighted number of shares (in thousands)
|
Net loss attributable to equity holders of the Company
|
|||||||||||||
|
For the computation of basic net earnings from continuing operations
|
|
$
|
(
|
)
|
|
$
|
(
|
)
|
||||||||
|
Effect of potential dilutive Ordinary shares - warrants
|
|
|
|
(
|
)
|
|||||||||||
|
For the computation of diluted net earnings from continuing operations (*)
|
|
$
|
(
|
)
|
|
$
|
(
|
)
|
||||||||
|
For the computation of basic and diluted net earnings from discontinued operations (*)
|
|
|
|
$
|
(
|
)
|
||||||||||
| *) |
For 2023 and 2022, potentially dilutive securities (share options) were excluded from the calculation of diluted earnings per share as they are antidilutive.
|
| **) |
Including the effect of Share Consolidation (See Note 20a).
|
| NOTE 23:- |
OPERATING SEGMENTS
|
|
Israel
|
Germany
|
Adjustments
|
Total
|
|||||||||||||
|
Year ended December 31, 2023
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss
|
(
|
)
|
(
|
)
|
|
(
|
)
|
|||||||||
|
Unallocated corporate expenses
|
(
|
)
|
(
|
)
|
||||||||||||
|
Total operating loss
|
(
|
)
|
||||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Israel
|
Germany
|
Adjustments
|
Total
|
|||||||||||||
|
Year ended December 31, 2022
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
|||||
|
Unallocated corporate expenses
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||||
|
Total operating loss
|
$
|
(
|
)
|
|||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
|
$ |
|
|||||||
|
Israel
|
Germany
|
Adjustments
|
Total
|
|||||||||||||
|
Year ended December 31, 2021
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
|||||
|
Unallocated corporate expenses
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||||
|
Total operating loss
|
$
|
(
|
)
|
|||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
| NOTE 24:- |
BALANCES AND TRANSACTIONS WITH INTERESTED AND RELATED PARTIES
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Other accounts receivables
|
$
|
|
$
|
|
||||
|
Other accounts payables
|
$
|
|
$
|
|
||||
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
General and administrative and Interest expenses
|
$
|
|
$
|
|
$
|
|
||||||
| b. | Compensation of key management personnel of the Group: |
|
Year ended December 31,
|
||||||||||||
|
2023
|
2022
|
2021
|
||||||||||
|
Payroll and related expenses
|
$ |
$
|
|
$
|
|
|||||||
|
Share-based compensation
|
$ |
$
|
|
$
|
|
|||||||
|
Professional fees *)
|
$ |
$
|
|
$
|
|
|||||||
| *) |
Includes payments to shareholders for the years ended 2023, 2022 and 2021 of $
|
| NOTE 25:- |
DISCONTINUED OPERATIONS AND DECONSOLIDATION OF TRICHOME
|
| NOTE 25:- |
DISCONTINUED OPERATIONS AND DECONSOLIDATION OF TRICHOME (Cont.)
|
| NOTE 25:- |
DISCONTINUED OPERATIONS AND DECONSOLIDATION OF TRICHOME (Cont.)
|
|
November 6,
2022
|
December 31,
2021
|
|||||||
|
ASSETS
|
||||||||
|
CURRENT ASSETS:
|
||||||||
|
Cash and cash equivalents
|
$
|
|
$
|
|
||||
|
Trade receivables
|
|
|
||||||
|
Other accounts receivable
|
|
|
||||||
|
Loans receivable
|
|
|
||||||
|
Biological assets
|
|
|
||||||
|
Inventories
|
|
|
||||||
|
|
|
|||||||
|
NON-CURRENT ASSETS:
|
||||||||
|
Property, plant and equipment, net
|
|
|
||||||
|
Derivative assets
|
|
|
||||||
|
Right-of-use assets, net
|
|
|
||||||
|
Intangible assets, net
|
|
|
||||||
|
Goodwill
|
|
|
||||||
|
|
|
|||||||
|
Total assets
|
$
|
|
$
|
|
||||
|
LIABILITIES
|
||||||||
|
CURRENT LIABILITIES:
|
||||||||
|
Trade payables
|
$
|
|
$
|
|
||||
|
Bank loans and credit facilities
|
|
|
||||||
|
Other accounts payable and accrued expenses
|
|
|
||||||
|
Current maturities of operating lease liabilities
|
|
|
||||||
|
|
|
|||||||
|
NON-CURRENT LIABILITIES:
|
||||||||
|
Operating lease liabilities
|
|
|
||||||
|
Deferred tax liability, net
|
|
|
||||||
|
|
|
|||||||
|
Total liabilities
|
$
|
|
$
|
|
||||
| NOTE 25:- |
DISCONTINUED OPERATIONS AND DECONSOLIDATION OF TRICHOME (Cont.)
|
|
Period ended
November 6,
2022
|
Year ended
December 31,
2021
|
|||||||
|
Revenues
|
$
|
|
$
|
|
||||
|
Cost of revenues
|
|
|
||||||
|
Gross profit before fair value adjustments
|
|
|
||||||
|
Fair value adjustments:
|
||||||||
|
Unrealized change in fair value of biological assets
|
|
|
||||||
|
Realized fair value adjustments on inventory sold in the period
|
(
|
)
|
(
|
)
|
||||
|
Total fair value adjustments
|
(
|
)
|
|
|||||
|
Gross profit
|
|
|
||||||
|
General and administrative expenses
|
|
|
||||||
|
Impairment of goodwill, intangible assets, right-of-use assets and fixed assets
|
|
|
||||||
|
Selling and marketing expenses
|
|
|
||||||
|
Restructuring expenses
|
|
|
||||||
|
Share-based compensation
|
|
|
||||||
|
Total operating expenses
|
|
|
||||||
|
Operating loss
|
(
|
)
|
(
|
)
|
||||
|
Finance expenses, net
|
(
|
)
|
(
|
)
|
||||
|
Loss before income taxes
|
(
|
)
|
(
|
)
|
||||
|
Income tax expense (benefit)
|
(
|
)
|
|
|||||
|
Net loss from discontinued operations, net of tax
|
$
|
(
|
)
|
$
|
(
|
)
|
||
| NOTE 25:- |
DISCONTINUED OPERATIONS AND DECONSOLIDATION OF TRICHOME (Cont.)
|
|
Period ended
November 6,
2022
|
Year ended
December 31,
2021
|
|||||||
|
Operating activities
|
$
|
(
|
)
|
$
|
(
|
)
|
||
|
Investing activities
|
$
|
(
|
)
|
$
|
(
|
)
|
||
|
Financing activities
|
$
|
(
|
)
|
$
|
|
|||
| *) |
From business combination dated, March 18, 2021.
|
| NOTE 26:- |
SUBSEQUENT EVENTS
|
| a. |
Loan from ADI
|
| b. |
Potential Reverse Merger with Kadimastem
|
| NOTE 26:- |
SUBSEQUENT EVENTS (Cont.)
|
| c. |
NASDAQ Compliance Notice
On January 31, 2024, the Company announced that it has received a 180-calendar day extension, until July 29, 2024, from the Nasdaq Stock Market ("Nasdaq"), to regain compliance with Nasdaq Marketplace Rule 5550(a)(2) (the "Bid Price Rule").
|
| d. |
35 Oak Holdings Ltd – Statement of Complaint
The Company, together with some of the Defendants brought, on February 22, 2024, a preliminary motion to strike out several significant parts of the claim (the "Motion") The Motion has not been scheduled by the court.
At this time, the Company's management is of the view that the Motion has merit and is likely to succeed in at least narrowing the scope of the claim against the Company, and that it may also result in certain of the claims against individuals being dismissed altogether, and if not dismissed narrowed in scope and complexity.
Given the preliminary stage of the action, and that the Company have not yet conducted a full investigation of the factual defenses, it is too early to opine on the merits of the claim or whether it is more likely than not to result in an outflow of funds to the Company and if so, how much.
|
| e. |
Acquisition of Jerusalem’s Leading Medical Cannabis Pharmacy – Oranim Pharm
On January 12, 2024, the Company announced that the final sixth payment of the Oranim Pharmacy Acquisition and the reconciliation between the parties regarding the remaining transaction payments are being rescheduled to April 15, 2024.
Through a new amendment signed January 10, 2024, the sixth (6) payment as well as the reconciliation between the parties regarding all remaining unpaid installments has been postponed to April 15, 2024. All six installments (that remain unpaid) will incur a
|
| f. |
Exercise of Focus Option
On February 26, 2024, IMC Holdings exercised its option and as of that date, the Company holds
|
F - 72