Exhibit 99.3

|
Management’s Discussion and Analysis
|
|
TABLE OF CONTENTS
|
3
|
|
|
4
|
|
|
4
|
|
|
4
|
|
|
7
|
|
|
8
|
|
|
12
|
|
|
16
|
|
|
17
|
|
|
20
|
|
|
26
|
|
|
27
|
|
|
28
|
|
|
28
|
|
|
30
|
|
|
30
|
|
|
34
|
|
|
34
|
|
|
36
|
|
|
36
|
|
|
49
|
|
|
51
|
|
|
61
|
|
|
74
|
2
|
Management’s Discussion and Analysis
|
|
IM Cannabis Corp. (“IM Cannabis” or the “Company”) is a British Columbia company operating in the international medical
cannabis industry. The Company’s common shares (the “Common Shares”) trade under the ticker symbol “IMCC” on the NASDAQ Capital Market (“NASDAQ”) as of March 1, 2021.
This Management’s Discussion and Analysis (this “MD&A”) reports on the consolidated financial condition and operating results of IM Cannabis for the year
and three months ended March 31, 2026. Throughout this MD&A, unless otherwise specified, references to “we”, “us”, “our” or similar terms, as well as the “Company” and “IM Cannabis” refer to IM Cannabis Corp., together with its subsidiaries, on a
consolidated basis, and the “Group” refers to the Company, its subsidiaries, and Focus Medical Herbs Ltd.
This MD&A should be read in conjunction with the interim condensed consolidated financial statements of the Company and the notes thereto for the three months ended March 31, 2026 and 2025 (the “Interim Financial Statements”), and with the Company's audited annual consolidated financial statements and the notes thereto for the years ended December 31, 2025 and 2024 (the “Annual
Financial Statements”). References herein to “Q1 2026” and “Q1 2025” refer to the three months ended March 31, 2026 and March 31, 2025 respectively, and references to “2025” refer to the year ended December 31, 2025.
The Interim Financial Statements have been prepared by management in accordance with the International Financial Reporting Standards (“IFRS”) as issued by the
International Accounting Standards Board (“IASB”). IFRS requires management to make certain judgments, estimates and assumptions that affect the reported amount of assets and liabilities at the date of the
Interim Financial Statements and the amount of revenue and expenses incurred during the reporting period. The results of operations for the periods reflected herein are not necessarily indicative of results that may be expected for future periods.
The Interim Financial Statements for the three months ended March 31, 2026, include the accounts of the Group, which includes, among others, the following entities:
|
Legal Entity
|
Jurisdiction
|
Relationship with the Company
|
|
I.M.C. Holdings Ltd. (“IMC Holdings”)
|
Israel
|
Wholly-owned subsidiary
|
|
I.M.C. Pharma Ltd. (“IMC Pharma”)
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
Focus Medical Herbs Ltd. (“Focus")
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
R.A. Yarok Pharm Ltd. (“Pharm Yarok”)
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
Rosen High Way Ltd. (“Rosen High Way”)
|
Israel
|
Wholly-owned subsidiary of IMC Holdings
|
|
Rivoly Trading and Marketing Ltd. d/b/a Vironna Pharm (“Vironna”)”
|
Israel
|
Subsidiary of IMC Holdings
|
|
Adjupharm GmbH (“Adjupharm”)
|
Germany
|
Subsidiary of IMC Holdings
|
|
Trichome Financial Corp. (“Trichome") (1)
|
Canada
|
Former wholly-owned subsidiary
|
|
Xinteza API Ltd (“Xinteza”)
|
Israel
|
Subsidiary of IMC Holdings
|
|
Shiran Societe Anonyme (“Greece”)
|
Greece
|
Subsidiary of IMC Holdings
|
|
IM Cannabis Holding NL B.V Netherlands (“IMC Holdings NL”)
|
Netherlands
|
Wholly-owned subsidiary of IMC Holdings
|
| (1) |
Discontinued operations.
|
In this MD&A, unless otherwise indicated, all references: (i) “Company Subsidiaries” are to the Israeli Subsidiaries and Adjupharm, (ii) “Israeli Operations” are to IMC Holdings and the Israeli
Subsidiaries as defined below, (iii) “Trichome” are to Trichome Financial Corp. and its subsidiaries. As of the date of this Interim Report “Israeli Subsidiaries” means
IMC Holdings, IMC Pharma, Focus, Pharm Yarok, Rosen High Way, Vironna and Xinteza.
All dollar figures in this MD&A are expressed in thousands of Canadian Dollars ($), except per share data and unless otherwise noted. All references to “NIS” are to New Israeli Shekels. All
references to “€” or to “Euros” are to Euros. All references to “US$” or to “U.S. Dollars” are to United States Dollars. The Company’s shares, options, units, prefunded warrants, warrants and prices are not expressed in thousands. Prices are not
expressed in thousands.
3
|
Management’s Discussion and Analysis
|
|
Certain non-IFRS financial measures are referenced in this MD&A that do not have any standardized meaning under IFRS, including “Gross Margin”, “EBITDA” and “Adjusted EBITDA”. The Company
believes that these non-IFRS financial measures and operational performance measures, in addition to conventional measures prepared in accordance with IFRS, enable readers to evaluate the Company’s operating results, underlying performance and
prospects in a similar manner to the Company’s management. For a reconciliation of these non-IFRS financial measures to the most comparable IFRS financial measures, as applicable, see the “Metrics and Non-IFRS
Financial Measures” section of the MD&A.
NOTE
REGARDING THE COMPANY’S ACCOUNTING PRACTICES
Unless otherwise indicated, all dollar figures are expressed in thousands and all references to: (i) “dollars” or “CAD”
or “$” are to Canadian dollars; (ii) “USD” or “US$” are to United States of America (“U.S.”
or “United States”) dollars; (iii) “NIS” are to New Israeli Shekels; and (iv) “€” or to “Euros”
are to Euros. All intercompany balances and transactions were eliminated on consolidation. Common shares, stock options, units, prefunded warrants, warrants, and prices, are not expressed in thousands. Our reporting currency and functional currency
is the Canadian dollar. The CAD to USD exchange rate as of March 31, 2026 was 0.71779.
EXECUTIVE
SUMMARY
OVERVIEW – CORPORATE
STRUCTURE
The Company was incorporated on March 7, 1980, under the name “Nirvana Oil & Gas Ltd.” pursuant to the Business Corporations Act (British Columbia). The Company's changed its name to “Nirvana
Industries Ltd.” on October 6, 1986; then to “Consolidated Nirvana Industries Ltd.” on February 22, 1989; then to “Navasota Resources Ltd.” on June 2, 1995, then to Anglo Aluminum Corp.” on January 25, 2010; then to Navasota Resources Inc. on July
12, 2013, and finally to its current name “IM Cannabis Corp.” on October 4, 2019.
The Common Shares began trading on the Canadian Securities Exchange (“CSE”) under the ticker symbol “IMCC” on November 5, 2019, and were subsequently listed on
the Nasdaq Capital Market under the same symbol effective March 1, 2021.
On May 28, 2025, the Company announced that it had applied for a voluntary delisting of its Common Shares from the CSE. The delisting became effective at the close of business on Monday, June 2,
2025, which marked the final trading day of the Company’s securities on the CSE.
The Company’s shares remain listed and continue to trade on Nasdaq under the ticker symbol “IMCC.”
4
|
Management’s Discussion and Analysis
|
|
On October 4, 2019, in connection with the reverse takeover transaction by IMC Holdings, the Company completed a consolidation of its Common Shares on a 2.83:1 basis, changed its name to “IM Cannabis
Corp.” and changed its business from mining to the international medical cannabis industry.
On February 12, 2021, in connection with its Nasdaq listing application, the Company completed a consolidation of its Common Shares on a 4:1 basis.
On November 17, 2022, in connection with regaining compliance with Nasdaq’s continued listing standards, the Company completed a 10:1 consolidation of its Common Shares, which was approved by
shareholders at the Company’s annual and special meetings of shareholders held on October 20, 2022.
On July 12, 2024, in connection with regaining compliance with Nasdaq’s continued listing standards, the Company completed a 6:1 consolidation of its Common Shares. The exercise price and/or
conversion price and number of Common Shares issuable under any of the Company's outstanding convertible securities were proportionately adjusted in connection with the July 2024 Consolidation.
OVERVIEW
– CURRENT OPERATIONS IN ISRAEL AND GERMANY
The Company operates in the medical cannabis sector in Israel and Germany. The Company’s activities in these jurisdictions include sourcing, importation, distribution and sale of medical cannabis
products in compliance with applicable regulatory requirements.
Israel
In Israel, the Company operates through IMC Holdings and its consolidated subsidiary Focus, which holds an IMCA license permitting the importation and supply of medical cannabis products. The
Company’s operations in Israel primarily consist of importing medical cannabis products from approved suppliers and distributing those products to pharmacies and patients in accordance with Israeli regulations. The Company does not currently operate
large-scale cultivation facilities in Israel and relies primarily on imported products that meet applicable quality and regulatory standards.
The Company’s Israeli operations include brand management, regulatory compliance, logistics coordination and relationships with pharmacies and prescribing physicians. Revenue in Israel is generated
from the sale of medical cannabis products to pharmacies and other authorized distributors.
Germany
In Germany, the Company operates through its German subsidiaries, which are authorized to import and distribute medical cannabis products under applicable German and European Union regulations. The
Company’s German operations focus on sourcing EU-GMP compliant medical cannabis products from approved suppliers and distributing such products to licensed pharmacies throughout Germany.
The German market is regulated by the BfArM, and the Company’s import volumes are subject to regulatory requirements, including compliance with applicable pharmaceutical standards. Revenue in Germany
is generated from the sale of imported medical cannabis products to pharmacies and other authorized customers.
The Company’s operations in both jurisdictions are subject to evolving regulatory frameworks, including changes in prescribing practices, import authorizations, product specifications and
distribution models, which may affect its revenues, margins and operating results.
5
|
Management’s Discussion and Analysis
|
|
Iron Swords War and other conflicts’ Effect on Gross Profit
On October 7, 2023, the State of Israel was attacked by the terrorist organization Hamas, and as a result, the State of Israel declared a state of war and a large-scale mobilization of reserves (the
“War”). At the same time, a front of fighting also developed in the northern border against the terrorist organization Hezbollah, which led to extensive evacuation of residents. The War is an exceptional event with security and economic implications
whose extent and outcomes are unpredictable. In response to the War, the State of Israel has taken significant steps to ensure the security of its residents, which have a considerable impact on economic and business activities in the country. The
events of the War have led to a reduction in business activity in the economy and a significant slowdown in economic activity, affecting the business operations of entities in various circles of influence, among others due to the closure of factories
in the south and north of the country, damage to infrastructure, long-term mobilization of reservists, and more. Potential fluctuations in commodity prices, foreign exchange rates, availability of materials, availability of manpower, local services,
and difficulties in accessing local resources have affected and are expected to continue to affect entities whose main operations are in Israel. In addition, the state of warfare also affects the activities of entities that rely on foreign workers or
on workers recruited for the purposes of the fighting, international trade, foreign companies in Israel, civil aviation, and more. As a result, the War has significant implications for the economy and imposes a considerable burden on the continuation
of business activity and the functional and operational continuity of the entities.
In November 2024, a ceasefire was reached with the terrorist organization Hezbollah in the north of the country, but the War continued in other areas.
On June 13, 2025, the State of Israel launched operation “Rising Lion” against military targets in Iran, with a focus on the Iranian nuclear project. As a result, a state of emergency was declared in
Israel, causing repercussions and restrictions on the Israeli economy, which included, inter alia, partial or complete closure of businesses, restrictions on gatherings in workplaces and in the education system, as well as a decrease in workforce due
to reserve enlistment and a reduction in number of foreign workers. During the operation, a targeted American strike was carried out against Iran, after which, on June 24, 2025, a ceasefire was reached between the parties.
Following the above, in October 2025 a ceasefire agreement was signed with terrorist organization Hamas in Gaza and as a result, the fighting subsided on most fronts.
On February 28, 2026, Israel and the United States launched a joint attack against Iranian government targets, following which Iran responded with missile fire towards Israel and other countries in
the region. As a result of the aforementioned, the Israeli government declared a special situation on the home front across the entire country, including restrictions on gatherings and a reduction in economic activity except for essential workplaces
until March 26, 2026.
The Company's management is continuously monitoring the developments regarding the War and is acting in accordance with the guidelines of the various authorities. The Company suffered a negative
impact from the War commencing the last quarter of 2023. The Company has experienced damage to its ability to function, affecting various aspects, including employees, supplies, imports, sales, and more.
6
|
Management’s Discussion and Analysis
|
|
OUR GOAL – DRIVE PROFITABLE
REVENUE GROWTH
Our primary goal is to sustainably increase revenue in each of our core markets, to accelerate our path to profitability and long-term shareholder value while actively managing costs and margins.
As part of its ongoing strategic review, the Company has also made a decision to explore the introduction of additional business activities, with the goal of enhancing long-term growth opportunities
and creating further shareholder value.
Given the evolving regulatory environment, particularly in Germany, the timing, scope, and impact of our plans are uncertain, and actual results may differ materially from current expectations.
HOW WE
PLAN TO ACHIEVE OUR GOAL – CORE STRATEGIES
Our strategy of sustainable and profitable growth consists of:
| • |
Develop and execute a long-term growth plan in Germany, based on the strong sourcing infrastructure in Israel which is powered by advanced product knowledge and regulatory expertise.
|
| • |
Optimize inventory to meet demand while managing INCB/BfArM import-estimate constraints and aligning products to Ph. Eur. 11.5 specifications. Diversify EU-GMP suppliers (Israel and other countries) to support availability.
|
| • |
Properly position brands with respect to target-market, price, potency and quality, such as our IMC brand in Israel and Germany.
|
| • |
Strong focus on efficiencies and synergies with domestic expertise in Israel and Germany.
|
| • |
High-quality, reliable supply to our customers and patients, leading to recurring sales.
|
| • |
Ongoing introduction of new Stock Keeping Units (“SKU”) to keep consumers and patients engaged.
|
| • |
Anticipate potential limits on telemedicine and mail-order by broadening local-pharmacy coverage, using pharmacy couriers where allowed, and supporting in-person prescribing with key physicians.
|
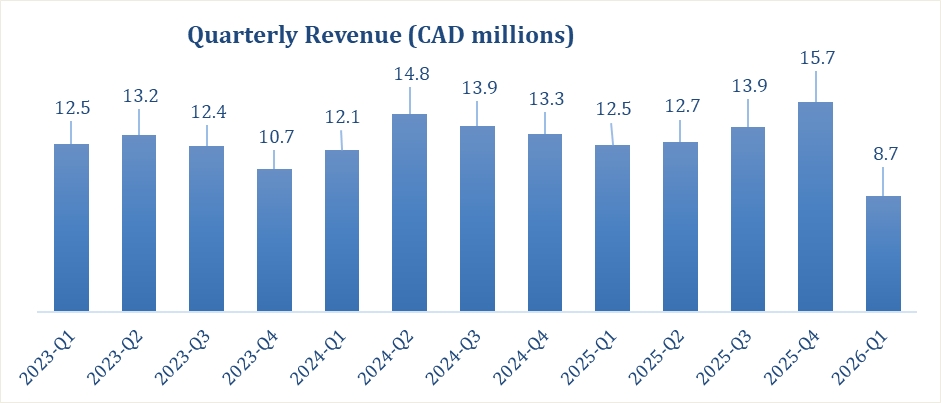
As reflected above, revenue fluctuated during the periods presented. In 2026, the quarterly revenues reflects a decrease compared to 2025, primarily driven by delays in shipments, variations in
product mix, pricing and volume, as well as reduced imports driven in part by cash flow constraints, which negatively impacted sales volumes.
7
|
Management’s Discussion and Analysis
|
|
GEOGRAPHIES AND NEW MARKETS
The Company is a medical cannabis company operating in Germany and Israel, two high-value markets, but highly challenging and rapidly evolving markets. In recent periods, the Company has faced
significant regulatory changes and uncertainty, increased competition, supply constraints, pricing pressures and adverse macro-economic conditions affecting the entire cannabis industry. In addition, geopolitical events and security-related
disruptions in Israel, including recent military operations, have adversely affected logistics, availability of products, patient behavior and the stability of the Israeli market. As a result, the Company's financial position and operating
performance have been negatively impacted, and substantial efforts, restructuring measures and strategic focus are required in order to stabilize and gradually rebuild its business.
With the April 2024 legalization in Germany, we pivoted our focus and resources to this market, leveraging our experience in product sourcing, quality assurance and regulatory compliance to capture
new growth opportunities. However, the German market is still in a formative stage, subject to ongoing regulatory developments and operational uncertainties, including licensing timelines, evolving distribution models and margin pressure. While we
view Germany as a key long-term growth driver, there can be no assurance that our efforts will offset the challenges faced in our legacy markets in the near term.
Israel
As one of the original eight Israeli cannabis pioneers, IMC has built a strong sourcing infrastructure in Israel. We have advanced product knowledge, regulatory expertise, and strong commercial
partnerships. Our extensive experience has made IMC a leading brand within the premium market segment.
We supply the Israeli medical cannabis market with our own IMC-branded products and exclusive ultra-premium Canadian cannabis brands, with which we have signed strategic licensing agreements.
The company also operates in the retail segment. The Company, through IMC Holdings, holds two licensed pharmacies, each selling medical cannabis products to patients: (i) Vironna, a leading pharmacy
in the Arab sector, and (ii) Pharm Yarok, the largest pharmacy in the Sharon Plain area and a big call center in the country (Vironna and Pharm Yarok collectively, the “Israeli Pharmacies”).
In addition, IMC, through IMC Holdings, operates a home delivery service, through a call center, effectively covering the entire country.
Germany
IMC has been operating through Adjupharm, its German subsidiary, since 2019, building the foundation needed to drive growth. We believe that our strong sourcing infrastructure in Israel, powered by
advanced product knowledge and regulatory expertise, gives the Company a competitive advantage in the growing German market. This is based on the premise that the German and Israeli markets share several common attributes such as robust commercial
infrastructure, highly developed digital capabilities, favourable demographics and customer preferences.
8
|
Management’s Discussion and Analysis
|
|
The Company’s focus in Germany is to import cannabis from its supply partners, which are then sold through our own IMC branded products, as well as exclusive ultra-premium Canadian cannabis brands,
with which we have signed strategic licensing agreements.
Our German operations are underpinned by a state-of-the-art warehouse and EU-GMP production facility in Germany (the “German Logistics Center”) with all the
necessary licenses to engage in additional production, cannabis testing, and release activities. Adjupharm can repackage bulk, perform stability studies, and offer such services to third parties.
Following the April 2024 implementation of Germany’s cannabis reform (CanG, comprising KCanG for consumer use and MedCanG for medical use), we have re-allocated resources to the German medical
channel while leveraging operating know-how from Israel. Medical cannabis now falls under MedCanG (with BfArM as the competent authority) rather than the BtMG. We continue to align product specifications with the European Pharmacopoeia 11.5 ‘Cannabis
flos’ monograph effective April 1, 20251.
In September 2025, BfArM temporarily paused new import approvals after Germany’s annual estimate of 122 tonnes for medical cannabis was reached; in late October 2025 authorities increased the
permissible volume (industry reports indicate a ~70-tonne uplift) and resumed approvals. Future adjustments to annual estimates could impact the timing of our import licences and inventory availability. In October 2025, the Federal Cabinet approved a
draft amendment to MedCanG proposing, among other items, an in-person physician visit requirement for prescribing cannabis flowers and a ban on mail-order pharmacy shipment. These proposals are not yet law; if enacted, they could affect patient
access patterns and our distribution mix. We will update our operating plan as the legislative process advances2.
We are updating QA/QC and labelling to Ph. Eur. 11.5, and reviewing licensing needs under MedCanG (import/trade authorisations, and, if pursued, domestic activities under the new cultivation
licensing framework). We are also stress-testing our pharmacy channel strategy to mitigate potential restrictions on telemedicine-origin prescriptions and mail-order fulfilment.
BRANDS
The IMC brand is well-known in the Israeli medical cannabis market, with reputable brands highly popular among Israeli consumers.
Israeli Medical Cannabis Business
The IMC brand has established its reputation in Israel for quality and consistency over the past 15 years and, more recently, with new high-end, ultra-premium strains that have made it to the
top-sellers list in pharmacies nationwide.
The Group maintains a portfolio of strains sold under the IMC umbrella from which popular medical cannabis dried flowers and full-spectrum cannabis extracts are produced.
1
https://www.skwschwarz.de/en/news/the-medical-cannabis-act-at-a-glance-legalization-with-conditions?utm_source=chatgpt.com
2
https://stratcann.com/news/germany-no-new-import-permits-for-dried-cannabis-in-2025/?utm_source=chatgpt.com
9
|
Management’s Discussion and Analysis
|
|
The IMC brand offers different products, leading with the highest-quality Canadian craft cannabis flower, which has established IMC as the leader of the super-premium segment in Israel.
Brands under the IMC Cannabis Portfolio:
The Craft Collection – IMC brand’s premium product line with indoor-grown, hand-dried and hand-trimmed high-THC cannabis flowers. The Craft Collection includes
exotic and unique cannabis strains such as Sup.S and PI.WI.
The Full Spectrum Extracts –IMC brand’s full spectrum, strain-specific cannabis extracts, includes high-THC Roma®T20 oil and OIL GLTO 33.
Roma® Product Portfolio – IMC's Roma® portfolio also includes oils. IMC’s Roma® strain is a high-THC medical cannabis
flower that offers a therapeutic continuum and is known for its strength and longevity of effect.
|
BLKMKT™, the Company’s second Canadian brand. It is a super-premium product line with indoor-grown, hand-dried and hand-trimmed high-THC cannabis
flowers. The BLKMKT™ includes BLK MLK, YA HEMI, PURPLE RAIN, JEALOUSY, Hemi GLTO, RAINBOW P, GUVA BOBA, Sunsets.rudel, Park fire OG, Up side down C and BACLTO.
|
|
LOT420 – this brand launched in Israel in Q2 2023, with super-premium
indoor-grown cannabis imported from Canada with high-THC. The LOT420 brand includes GLTO 33, Apps and Bans, O.C., and GLTO 33 oil. The Company ceased selling Atomic
APP.

The PICO collection (minis)- Under the BLKMKT™ and LOT420 brands, the Company launched in 2023 a new type of product (small flowers), which is a super-premium
indoor-grown cannabis imported from Canada with high-THC. The PICO collection includes the following products: PICO PURPLE RAIN, PICO YA HEMI, PICO JEALOUSY, Pico upside Down, PICO RAIN BOW, Pico California love, PICO BLK MLK and PICO Bacio Glto.
Flower – In Q2 2024, the Company launched a super-premium indoor-grown cannabis imported from Canada with high-THC. The Flower brand includes cannabis strains
called California love and Face Sherb.
Token - New Affordable Lifestyle Line. Launched in Israel and Germany in Q2 2025, TOKEN by IMC introduces a new lifestyle edition offering high-quality,
everyday cannabis at accessible prices such as KEYNAPZ and SIPER.

For more information, see “Strategy in Detail – Brands – New Product Offerings” section of the MD&A.
10
|
Management’s Discussion and Analysis
|
|
German Medical Cannabis Business
In Germany, IMC is positioned among the top cannabis companies. The Group’s competitive advantage in Germany lies in its track record, experience and brand reputation as a reliable partner for
medical cannabis for both pharmacies and patients.
In Germany, IMC initially focused on selling only IMC-branded products, both flowers and full-spectrum extracts, to increase brand awareness and build brand heritage among German healthcare
professionals.
In 2025, IMC expanded its portfolio to include a new mid-market brand “Token” by IMC as well as a new value brand “Mids” by IMC. These two brands join “Selected” by IMC, as well as BLKMKT™, an
ultra-premium Canadian brand.
The Company maintains a portfolio of strains sold under the IMC umbrella from which popular medical cannabis dried flowers and full-spectrum cannabis extracts
are produced.
HIGH-QUALITY,
RELIABLE SUPPLY:
Israel
The Company is concentrating on leveraging its skilled sourcing team and strategic alliances with Canadian suppliers as well as the import of medical cannabis from its Canadian Facilities. The
Company continues to import cannabis products and supply medical cannabis to patients through licensed pharmacies. To supplement growing demand, the Company continues its relationships with third-party cultivation facilities in Israel for the
propagation and cultivation of the Company’s existing proprietary genetics and for the development of new products.
In addition, the Company is operating through its subsidiaries who obtained a license from the IMCA to, among others, import cannabis products and supply medical cannabis to patients.
Pursuant to the applicable Israeli cannabis regulations, following the import of medical cannabis, medical cannabis products are then packaged by contracted GMP-licensed producers of medical
cannabis. The packaged medical cannabis products are then sold by the Group under the Company’s brands to local Israeli pharmacies directly or through contracted distributors.
Germany
The Company continues to expand its presence in the German market by forging partnerships with pharmacies and distributors across the country and developing Adjupharm and its German Logistics Center
as the Company’s European hub. Adjupharm sources its supply of medical cannabis for the German market and from various EU-GMP certified European and Canadian suppliers. The German Logistics Center is EU-GMP certified, upgrading Adjupharm production
technology and increasing its storage capacity to accommodate its anticipated growth. Adjupharm has a certification for primary repackaging, making it one of a handful of companies in Germany fully licenced to repack bulk.
Adjupharm currently holds wholesale, narcotics handling, manufacturing, procurement, storage, distribution, and import/export licenses granted to it by the applicable German regulatory authorities
(the “Adjupharm Licenses”).
11
|
Management’s Discussion and Analysis
|
|
KEY HIGHLIGHTS FOR THE
FIRST QUARTER OF 2026
In the first quarter of 2026, the Company continued to focus on efficiency operations, increasing sales and presence in German cannabis market resulting in accelerated growth in the region while
continuing its sales efforts in the Israeli market. The Company continues with its efforts to establish new supply chain processes and to improve its supplier base for the German market to support the year 2026 goal of profitability. The Company’s
key highlights and events for the first quarter ended March 31, 2026, include:
SUBSEQUENT EVENTS
April Capital Note
On April 6, 2026, the Company entered into a Note Purchase Agreement (the “April 2026 Purchase Agreement”) with the Investor, pursuant to which the Company issued to the Investor (A) a convertible
note (the “April 2026 Note”) in the principal amount of approximately US$250 thousand (approximately $340) (the “April 2026 Note’s Subscription Amount”), which is convertible into the Common Shares, no par value per share, at a purchase price equal
to 90% of the April 2026 Note’s Subscription Amount and (B) a warrant to purchase up to 272,861 Common Shares (the “April 2026 Warrant”), which is the number equal to thirty-three and one-third percent (331/3%) of the April 2026 Note’s Subscription Amount divided by an exercise price of $0.47 per Common Share. Such transaction
closed on April 6, 2026.
The April 2026 Note bears an interest rate of eight percent (8.0%) per annum accruing from its closing date (which shall increase to fourteen percent (14.0%) upon the occurrence of an Event of
Default (as defined in the April 2026 Note). The April 2026 Note is not repayable in cash and the Company’s obligations thereunder will be satisfied solely through the issuance of Common Shares upon conversion of the Note in accordance with its
terms.
The number of Common Shares issuable upon any conversion of the principal amount under the April 2026 Note is determined by dividing the applicable conversion amount by its conversion price. The
conversion price of the April 2026 Note is equal to the lower of (i) the fixed price, as defined in the April 2026 Note, or (ii) its variable price, which equals to ninety percent (90%) of the lowest daily volume-weighted average price of the Common
Shares during the twenty (20) consecutive trading days immediately preceding the conversion date, provided, however, that such variable price will not be lower than the floor price, as defined in the April 2026 Note. The fixed price set in the April
2026 Note is $0.339. The floor price set in the April 2026 Note is $0.07. No fractional Common Shares will be issued upon conversion, and any fractional amount will be rounded to the nearest $0.0001. Any fractional Common Shares will be rounded down
to the nearest whole share.
The April 2026 Warrant entitles its holder to purchase one Common Share (each, a “warrant share”) at an exercise price of $0.47 per such warrant share. The April 2026 Warrant became exercisable
immediately upon its issuance date, April 6, 2026, and will be exercisable for a period of five (5) years, until April 6, 2031. If the Warrant is not exercised by its termination date, April 6, 2031, the April 2026 Warrant will expire and be of no
further force or effect. The April 2026 Warrant and the warrant shares may not be traded for a period of four (4) months, unless permitted under applicable securities legislation.
The April 2026 Note includes customary limitations on conversion, including a beneficial ownership cap of 4.99% of the outstanding Common Shares after giving effect to such conversion.
12
|
Management’s Discussion and Analysis
|
|
The April 2026 Purchase Agreement include customary representations, warranties and covenants of the Company and the Investor, including the Company’s obligation to reserve sufficient Common Shares
for issuance upon conversion of the Notes and to file a resale Registration Statement with the SEC providing for the resale by the Investor of the Common Shares and the warrant shares issuable upon conversion of the April 2026 Note within thirty (30)
trading days after the applicable closing date. The Company has also agreed to use commercially reasonable efforts to cause the Registration Statement to become effective as soon as possible, but in no event later than the date which shall be the
earlier of: (x) in the event that the Registration Statement is not subject to a full review by the SEC, sixty (60) calendar days after the applicable closing date, or in the event that the Registration Statement is subject to a full review by the
SEC, ninety (90) calendar days after the applicable closing date, and (y) the fifth (5th) business day after the date on which the Company is notified (orally or in writing, whichever is earlier) by the SEC that such Registration Statement will not
be reviewed or will not be subject to further review.
May Capital Note
On May 7, 2026, entered into a Note Purchase Agreement (the “May 2026 Purchase Agreement”) with the Investor, pursuant to which the Company issued to the Investor (A) a convertible note (the “May
2026 Note”) in the principal amount of US$300,000 (the “May 2026 Subscription Amount”), which is convertible into the Common Shares, no par value per share, at a purchase price equal to ninety percent (90%) of the May 2026 Subscription Amount; and
(B) a warrant to purchase up to 1,127,820 Common Shares (the “May 2026 Warrant”), equal to a number of Common Shares determined by one hundred percent (100%) of the May 2026 Subscription Amount divided by an exercise price of $0.36 per Common Share.
Such offering closed on May 7, 2026. The Company intends to use the net proceeds of US$270,000 received from such offering for general corporate purposes.
The May 2026 Note bears an interest rate of eight percent (8.0%) per annum accruing from its closing date (which shall increase to fourteen percent (14.0%) upon the occurrence of an Event of Default
(as defined in the May 2026 Note). The May 2026 Note is not repayable in cash and the Company’s obligations thereunder will be satisfied solely through the issuance of Common Shares upon conversion of the May 2026 Note in accordance with its terms.
The number of Common Shares issuable upon any conversion of the principal amount under the May 2026 Note is determined by dividing the applicable conversion amount by its conversion price. The
conversion price is equal to the lower of (i) the fixed price, as defined in the May 2026 Note, or (ii) its variable price which equals to ninety percent (90%) of the lowest daily volume-weighted average price of the Common Shares during the twenty
(20) consecutive trading days immediately preceding the conversion date, provided, however, that such variable price will not be lower than the floor price, as defined in the May 2026 Note. The fixed price set in the May 2026 Note is $0.266. The
floor price set in the Note $0.05. No fractional Common Shares will be issued upon conversion, and any fractional amount will be rounded to the nearest $0.0001. Any fractional Common Shares will be rounded down to the nearest whole share.
The May 2026 Warrant entitles its holder to purchase one Common Share (each, a warrant share) at an exercise price of $0.36 per warrant share. The May 2026 Warrant became exercisable immediately upon
its issuance date, May 7, 2026, and will be exercisable for a period of five (5) years, until May 7, 2031. If the Warrant is not exercised by its termination date, May 7, 2031, the May 2026 Warrant will expire and be of no further force or effect.
The May 2026 Warrant and the warrant shares may not be traded for a period of four (4) months, unless permitted under applicable securities legislation.
The May 2026 Note includes customary limitations on conversion, including a beneficial ownership cap of 4.99% of the outstanding Common Shares after giving effect to such conversion.
13
|
Management’s Discussion and Analysis
|
|
The May 2026 Purchase Agreement include customary representations, warranties and covenants of the Company and the Investor, including the Company’s obligation to reserve sufficient Common Shares for issuance upon
conversion of the May 2026 Notes and to file a resale Registration Statement with the SEC providing for the resale by the Investor of the Common Shares and the warrant shares issuable upon conversion of the May 2026 Note within thirty (30)
trading days after the applicable closing date. The Company has also agreed to use commercially reasonable efforts to cause the Registration Statement to become effective as soon as possible, but in no event later than the date which shall be
the earlier of: (x) in the event that the Registration Statement is not subject to a full review by the SEC, sixty (60) calendar days after the applicable closing date , or in the event that the Registration Statement is subject to a full
review by the SEC, ninety (90) calendar days after the applicable closing date, and (y) the fifth (5th) business day after the date on which the Company is notified (orally or in writing, whichever is earlier) by the SEC that such Registration
Statement will not be reviewed or will not be subject to further review.
Other Loan Agreements
On April, 2026, IMC Holdings entered into a loan agreement with the Company's Chief Executive Officer and main shareholder, in the amount of NIS 725 thousand (approximately $333) which bears fixed
annual interest at the rate prescribed by the Income Tax Regulations for determining the interest rate under Section 3(i) of the Income Tax Ordinance and shall be repaid by August, 2026.
On April, 2026, IMC Holdings entered into loan agreements with a relative of one of the Company's main shareholders, in total amount of NIS 1,250 thousand (approximately $573). As of the date of this
report the loan terms have not yet been set.
Share Issuances
During the period between April 1 to May 13, 2026, the Company issued 2,793,216 Common Shares in respect of conversion of convertible notes in the amount of US$839 (approximately $1,141) at an
average exercise price of US$0.3004 per share.
14
|
Management’s Discussion and Analysis
|
|
REVIEW
OF FINANCIAL PERFORMANCE
FINANCIAL HIGHLIGHTS
Below is the analysis of the changes that occurred for the three months ended March 31, 2025, with further commentary provided below.
|
For the three months ended
March 31
|
For the year ended
December 31
|
|||||||||||
|
2026
|
2025
|
2025
|
||||||||||
|
Net Revenues
|
$
|
8,679
|
$
|
12,500
|
$
|
54,731
|
||||||
|
Gross profit before fair value impacts in cost of sales
|
$
|
1,419
|
$
|
3,448
|
$
|
9,686
|
||||||
|
Gross margin before fair value impacts in cost of sales (%)
|
16
|
%
|
28
|
%
|
18
|
%
|
||||||
|
Operating Profit (Loss)
|
$
|
(1,676
|
)
|
$
|
158
|
$
|
(11,587
|
)
|
||||
|
Net Profit (Loss)
|
$
|
(2,467
|
)
|
$
|
175
|
$
|
(11,750
|
)
|
||||
|
Loss per share attributable to equity holders of the Company – Basic (in CAD)
|
$
|
(0.38
|
)
|
$
|
0.09
|
$
|
(2.67
|
)
|
||||
|
Loss per share attributable to equity holders of the Company - Diluted (in CAD)
|
$
|
(0.38
|
)
|
$
|
0.09
|
$
|
(2.67
|
)
|
||||
The Overview of Financial Performance includes reference to “Gross Margin”, which is a non-IFRS financial measure that the Company defines as the difference between revenue and cost of revenues
divided by revenue (expressed as a percentage), prior to the effect of a fair value adjustment for inventory and biological assets. For more information on non-IFRS financial measures, see the “Non-IFRS Financial
Measures” and “Metrics and Non-IFRS Financial Measures” sections of the MD&A.
OPERATIONAL RESULTS
In each of the markets in which the Company operates, it must navigate evolving customer and patient trends to remain competitive with other suppliers of medical cannabis products.
The Company believes several key factors create tailwinds to facilitate further industry growth. In Israel, the number of medical patients currently stands at 136,900 out of which – 105,100 with
licenses and 31,800 with prescriptions as of March 2026. This figure is expected to grow in the coming years and may further benefit from regulatory changes liberalizing the cannabis market in Israel. IM Cannabis is a large distributor of medical
cannabis in Israel.
|
Germany Region Revenue for the three months ended
|
||||||||||||||||||||
|
2026
|
2025
|
|||||||||||||||||||
|
March 31,
|
December 31,
|
September 30
|
June 30,
|
March 31,
|
||||||||||||||||
|
Revenue for the period
|
$
|
4,278
|
$
|
13,073
|
$
|
8,768
|
$
|
6,802
|
$
|
7,705
|
||||||||||
|
Q vs Q change%
|
(67
|
)%
|
49
|
%
|
29
|
%
|
(12
|
)%
|
-
|
|||||||||||
The Company's products are in high demand in the German market, and it is investing efforts in building a strong, high-volume supply chain to support its current operation and future growth in the
country. The decreased revenue in the three months ended March 31, 2026, compared to the three months ended December 31, 2025, is due to delays in shipments, variations in product mix, pricing and volume, as well as reduced imports driven in part by
cash flow constraints, which negatively impacted sales volumes.
15
|
Management’s Discussion and Analysis
|
|
REVENUES
The following table summarizes the Company’s revenues for the periods presented. The period-to-period comparison of results is not necessarily indicative of results for future periods.
For the three months ended March 31:
|
CAD in thousands
|
Israel
|
Germany
|
Adjustments
|
Total
|
||||||||||||||||||||||||||||
|
For the Three Months Ended
March 31,
|
For the Three Months Ended
March 31,
|
For the Three Months Ended
March 31,
|
For the Three Months Ended
March 31,
|
|||||||||||||||||||||||||||||
|
2026
|
2025
|
2026
|
2025
|
2026
|
2025
|
2026
|
2025
|
|||||||||||||||||||||||||
|
Revenue
|
$
|
4,401
|
$
|
4,795
|
$
|
4,278
|
$
|
7,705
|
$
|
-
|
$
|
-
|
$
|
8,679
|
$
|
12,500
|
||||||||||||||||
|
Segment income (loss)
|
$
|
(575
|
)
|
$
|
111
|
$
|
(720
|
)
|
$
|
577
|
$
|
-
|
$
|
-
|
$
|
(1,295
|
)
|
$
|
688
|
|||||||||||||
|
Unallocated corporate expenses
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
(381
|
)
|
$
|
(530
|
)
|
$
|
(381
|
)
|
$
|
(530
|
)
|
||||||||||||
|
Total operating income (loss)
|
$
|
(575
|
)
|
$
|
111
|
$
|
(720
|
)
|
$
|
577
|
$
|
(381
|
)
|
$
|
(530
|
)
|
$
|
(1,676
|
)
|
$
|
158
|
|||||||||||
|
Depreciation, amortization and impairment
|
$
|
442
|
$
|
439
|
$
|
55
|
$
|
24
|
$
|
-
|
$
|
-
|
$
|
497
|
$
|
463
|
||||||||||||||||
The Company’s consolidated revenues for the three months ended March 31, 2026, were attributed mostly to the sale of medical cannabis products in Israel and Germany. Revenue in Germany reflects
demand trends, product availability and import volumes during the periods presented.
Revenues for the three months ended March 31, 2026, and 2025 were $8,679 and $12,500, respectively, representing a decrease of $3,821, or 31%. The decrease was primarily attributable to a decrease in
revenue in Germany of $3,427 due to delays in shipments, and a decrease of $394 in revenue in Israel, reflecting variations in product mix, pricing and volume as well as reduced imports driven in part by cash flow constraints, which negatively
impacted sales volumes.
16
|
Management’s Discussion and Analysis
|
|
COST
OF REVENUES
The cost of revenues is comprised of the purchase of raw materials and finished goods, import costs, production costs, product laboratory testing, shipping, and salary expenses. When sold, inventory
is later expensed to the cost of sales. Direct production costs are also expensed through the cost of sales.
The cost of revenues for the three months ended March 31, 2026, and 2025 were $7,260 and $9,052, respectively, representing a decrease of $1,792, or 20%. The decrease was primarily attributable to
the decrease in revenues.
GROSS PROFIT
Gross profit for the three months ended March 31, 2026, and 2025 was $1,419 and $3,448, respectively, representing a decrease of $2,029, or 59%.
GENERAL AND ADMINISTRATIVE
General and administrative expenses for the three months ended March 31, 2026, and 2025 were $1,569 and $2,009, respectively, representing a decrease of $440 or 22%.
The decrease in general and administrative expenses for the three months ended March 31, 2026 was primarily attributable to lower insurance costs, reduced salaries and related expenses, and lower
other administrative expenses, partially offset by increased professional fees associated with financing activities, legal matters and corporate initiatives.
Insurance costs decreased by $423 to $(120) for the three months ended March 31, 2026, compared to $303 for the comparable prior-year period, due to a refund received. Salaries and related expenses
decreased by $165 to $411 for the three months ended March 31, 2026, compared to $576 for the comparable prior-year period. Other administrative expenses decreased by $88 to $467 for the three months ended March 31, 2026, compared to $555 for the
comparable prior-year period. These decreases were partially offset by an increase in professional fees of $233 to $696 for the three months ended March 31, 2026, compared to $463 for the comparable prior-year period.
SELLING AND MARKETING
Selling and marketing expenses for the three months ended March 31, 2026, and 2025 were $1,526 and $1,273, respectively, representing an increase of $253 or 20%.
The increase in sales and marketing expenses for the three months ended March 31, 2026 was primarily attributable to (i) an increase in selling and marketing expenses of $125 ($190 in 2026 compared
to $65 in 2025), and (ii) an increase in professional fees of $74 ($77 in 2026 compared to $3 in 2025).
OTHER
OPERATING EXPENSES
Other operating expenses for the three months ended March 31, 2026, and 2025 were $nil and $nil, respectively.
17
|
Management’s Discussion and Analysis
|
|
SHARE-BASED COMPENSATION
Share-based compensation expenses for the three months ended March 31, 2026, and 2025 were $nil and $8, respectively, representing a decrease of $8 or 100%.
OPERATING EFFICIENCY AND
OPERATING RATIO
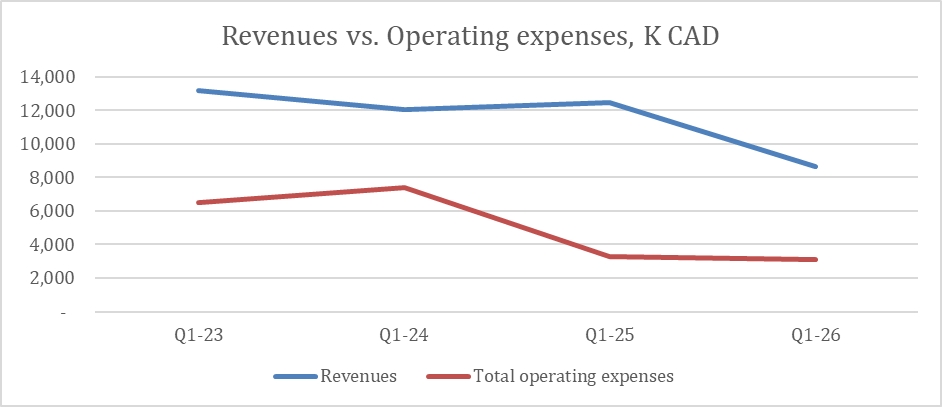
The operating expense ratio for the three months ended March 31, 2026, and 2025, were 36% and 26% respectively, representing a decrease in efficiency of approximately 35%. The efficiency ratio
decline resulted from decreased revenues.
FINANCING
INCOME (EXPENSE), NET
Financing (expense) net for the three months ended March 31, 2026 and 2025 was $(832) and $(12) respectively, representing an increase in financing expense of $820.
NET
INCOME (LOSS) PER SHARE BASIC AND DILUTED
Net loss for the three months ended March 31, 2026 was $2,467 compared to a net income of $175 for the three months ended March 31, 2025, representing an increase in net loss of $2,642. The increase
in net loss primarily reflected lower revenues, reduced gross profit and increased financing expenses during the quarter.
Basic income or loss per share is calculated by dividing the net income attributable to holders of Common Shares by the weighted average number of Common Shares outstanding during the period. Diluted
income or loss per Common Share is calculated by adjusting the earnings and number of Common Shares for the effects of dilutive warrants and other potentially dilutive securities. The weighted average number of Common Shares used as the denominator
in calculating diluted income or loss per Common Share excludes unissued Common Shares related to Options as they are anti-dilutive.
18
|
Management’s Discussion and Analysis
|
|
Basic Income or (Loss) per Common Share for the three months ended March 31, 2026, and 2025 were $(0.38) and $0.09 per Common Share, respectively.
Diluted net Income or (Loss) per share for the three months ended March 31, 2026, and 2025 were $(0.38) and $0.09, respectively.
TOTAL ASSETS
Total assets as of March 31, 2026, and December 31, 2025 were $23,701 and $31,736, respectively, representing a decrease of $8,035 or 25%. The decrease is mainly attributed to a decrease in trade
receivables of $3,681, and to a decrease of $2,267 in inventory.
Investment in Xinetza
On December 26, 2019, IMC Holdings entered into a Share Purchase Agreement with Xinteza API Ltd. (“Xinteza”), under which IMC Holdings invested an aggregate amount of US$1,700 (approximately $2,468)
in exchange for the issuance of 38,082 preferred shares of Xinteza.
On February 24, 2022, IMC Holdings entered into a Simple Agreement for Future Equity with Xinteza, under which IMC Holdings invested US$100 (approximately $125), in exchange for additional future
shares of Xinteza.
As of December 31, 2025, IMC Holdings holds 25.32% of the voting rights of Xinteza and has the right for two members of the Board of Directors out of five. However, it was determined that the
economic interests of the preferred shares are not substantially identical to those of ordinary shares (due to such features as liquidation preference and redemption feature). Accordingly, since the preferred shares do not meet the ordinary equity
ownership interest criteria, the equity method is not applicable, and the investment in Xinteza is subject to the provisions of IFRS 9 and is accounted for as a financial asset measured at fair value through profit or loss categorized within Level 3
of the fair value hierarchy.
As of March 31, 2026, and December 31, 2025, the investment in an affiliate amounted to $1,819 and $1,776, respectively, due to an effect of foreign currency translation of $43.
TOTAL LIABILITIES
As of March 31, 2026, total liabilities were $28,825, compared to $35,351 as of December 31, 2025. The change was primarily attributable to changes in trade payables, bank borrowings, convertible
instruments and other financial liabilities.
As of March 31, 2026, total borrowings and credit from financial and non-financial institutions amounted to $11,495, compared to $15,269 as of December 31, 2025.
19
|
Management’s Discussion and Analysis
|
|
As of March 31, 2026 and December 31, 2025, the Company’s borrowings consisted of:
|
2026
|
2025
|
|||||||
|
Credit from bank institutions
|
$
|
1,109
|
$
|
1,067
|
||||
|
Credit from non-financial institutions
|
8,952
|
9,696
|
||||||
|
Check receivables
|
1,434
|
4,506
|
||||||
|
Total borrowings
|
$
|
11,495
|
$
|
15,269
|
||||
Since the Company’s inception through March 31, 2026, the Company has funded its operations through raising capital, inter alia, through public offering, non-broker private placement transactions and
credits from bank institutions and others.
The Company’s liquidity is affected by the timing of customer collections, inventory turnover and payment terms with suppliers.
As of March 31, 2026, the Company’s cash and restricted cash totaled $1,152 and the Company’s working capital deficit (current assets minus current liabilities) amounted to $12,602. For the three
months ended March 31, 2026, the Company had an operating loss of ($1,676) and cash flows used in operating activities of $186.
As of March 31, 2026, the Company’s financial liabilities were $28,825, which included trade payables, other payables and accrued expenses, borrowings and other financial liabilities, a substantial
portion of which had contractual maturities of less than one year.
The Company manages its liquidity risk by reviewing its capital requirements on an ongoing basis. Based on the Company’s working capital position on March 31, 2026, management considers liquidity
risk to be high. As of March 31, 2026, the Company has identified the following liquidity risks related to financial liabilities:
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
> 10 years
|
|||||||||||||
|
Contractual Obligations
|
$
|
10,949
|
$
|
860
|
$
|
-
|
$
|
-
|
||||||||
The maturity profile of the Company’s other financial liabilities (trade payables, other account payable and accrued expenses, and warrants) as of March 31, 2026, are less than one year.
|
Payments Due by Period
|
||||||||||||||||||||
|
Contractual Obligations
|
Total
|
Less than one year
|
1 to 3 years
|
4 to 5 years
|
After 5 years
|
|||||||||||||||
|
Debt
|
$
|
11,495
|
$
|
10,710
|
$
|
785
|
$
|
-
|
$
|
-
|
||||||||||
|
Finance Lease Obligations
|
$
|
314
|
$
|
239
|
$
|
75
|
$
|
-
|
$
|
-
|
||||||||||
|
Total Contractual Obligations
|
$
|
11,809
|
$
|
10,949
|
$
|
860
|
$
|
-
|
$
|
-
|
||||||||||
20
|
Management’s Discussion and Analysis
|
|
As of December 31, 2026 and 2025, the Company’s financial liabilities were $28,825 and $35,351, respectively.
As of March 31, 2026 and December 31, 2025, the Company did not have any distributions or cash dividends declared per-share for the outstanding Common Shares.
The Company’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outflows for operating expenses and capital expenditures,
including cost saving plans. In 2023, the Board approved a cost saving plan, to allow the Company to continue its operations and meet its cash obligations. The cost saving plan entailed reducing costs through efficiencies and synergies primarily
involving the following measures: discontinuing loss-making activities, reducing payroll and headcount, reduction in compensation paid to key management personnel (including layoffs of key executives), operational efficiencies and reduced capital
expenditures. These actions resulted in cost savings during 2024 and 2025, and the Company will continue its efforts for efficiency operations also during 2026.
The projected cash flow for the rest of 2026 indicates that there is uncertainty regarding whether the Company will generate sufficient funds to continue its operations and meet its obligations as
they become due. The Company continues to evaluate additional sources of capital and financing. However, there is no assurance that additional capital and or financing will be available to the Group, and even if available, whether it will be on terms
acceptable to the Group or in amounts required.
These conditions raise substantial doubt about the Company’s ability to continue as a going concern. The interim consolidated financial statements included do not include any adjustments relating to
the recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
The interim consolidated financial statements included have been prepared on the basis of accounting principles applicable to a going concern, which assumes that the Company will continue in
operation for the foreseeable future and will be able to realize its assets and discharge its liabilities in the normal course of operations. The interim consolidated financial statements do not include any adjustments to the amounts and
classification of assets and liabilities that would be necessary should the Company be unable to continue as a going concern. Such adjustments could be material.
SOURCES
OF LIQUIDITY AND FINANCING ARRANGEMENTS
The Company’s primary sources of liquidity consist of cash generated from operations, borrowings under credit facilities, private placements of equity securities and convertible instruments, and
short-term financing arrangements with financial and non-financial institutions.
As of March 31, 2026, the Company had cash and restricted cash of $1,152. The Company continues to rely on external financing arrangements to support working capital needs and to fund operations.
and capital resources.
21
|
Management’s Discussion and Analysis
|
|
Revolving Credit Facility with Bank Mizrahi
On March 23, 2022, Focus entered into a revolving credit facility with an Israeli bank, Bank Mizrahi (the “Mizrahi Facility”). The Mizrahi Facility is
guaranteed by Focus assets. Advances from the Mizrahi Facility were used for working capital needs. The Mizrahi Facility had a total commitment of up to NIS 15,000 (approximately $6,000) and had a one-year term for on-going needs and 6 months term
for imports and purchases needs. The Mizrahi Facility is renewable upon mutual agreement by the parties. The borrowing base available for draw at any time throughout the Mizrahi Facility and is subject to several covenants to be measured on a
quarterly basis. The Mizrahi Facility bears interest at the Israeli Prime interest rate plus 1.5%.
On May 17, 2023, the Company and Bank Mizrahi entered into a new credit facility with total commitment of up to NIS 10,000 (approximately $3,600) (the “New Mizrahi
Facility”). The New Mizrahi Facility consists of NIS 5,000 credit line and NIS 5,000 loan to be settled with twenty-four (24) monthly installments from May 17, 2023. This loan bears interest at the Israeli Prime interest rate plus 2.9%.
On August 1, 2024, the credit line of approximately NIS 1,825 related to the New Mizrahi Facility was converted into a six-month short-term loan, bearing an annual variable interest rate of P+1.9%
(with the Israel Prime interest rate as of the submission date being 6%).
As of December 31, 2024, Focus had a short-term loan of $2,586 in respect of the new Mizrahi facility. The New Credit facility is also subject to several covenants to be measured on a quarterly basis
which were not met as of December 31, 2024.
As of March 20, 2025, Mizrahi Bank has been extending the short-term loan on a weekly basis; however, on March 20, 2025, the bank and the company signed an agreement modifying the New Mizrahi
Facility terms as follows:
| • |
$1,560 (NIS 4 million) was extended as a loan with a six-month grace period, after which repayment will be made in 31 monthly installments commencing on September 10, 2025. The principal loan will not require a personal guarantee and will
bear an interest at a rate of P+2.9% to be paid monthly, commencing on April 20, 2025.
|
| • |
The remaining $390 (NIS 1 million) was extended as a credit line from March 19, 2025, to March 12, 2026. As of the date of this report, the credit line has been extended to September 25, 2026.
|
Mr. Oren Shuster, the Company’s Chief Executive Officer and director provided the bank with a personal guarantee for the outstanding borrowed amount, allowing the New Mizrahi Facility to remain
effective.
On June 29, 2025, the Mizrahi Facility approved to postpone by one month the first loan installment of the principal amount (only and not the interest) from September 21, 2025, to October 21, 2025,
which was paid in full on time.
On April 29, 2025, IMC Holdings entered into a short-term loan agreement with a non-financial institution in the amount of NIS 1,000 (approximately $375). This loan bears interest at an annual rate
of 17% and matures 12 months from the date of signing the loan agreement.
22
|
Management’s Discussion and Analysis
|
|
Loan and Repayment to ADI
On October 11, 2022, IMC Holdings entered into a loan agreement with A.D.I. Car Alarms Stereo Systems Ltd (“ADI” and the “ADI
Agreement”), to borrow a principal amount of NIS 10,500 (approximately $4 million) at an annual interest of 15% (the “ADI Loan”), which was to be repaid within 12 months of the date of the ADI
Agreement. The ADI Loan was secured by a second rank land charge on the German Logistics Center. In addition, Mr. Oren Shuster, the Company’s Chief Executive Officer and director, provided a personal guarantee to ADI should the security not be
sufficient to cover the repayment of the ADI Loan.
On October 25, 2023, IMC Holdings and ADI signed an amendment to the ADI Agreement, extending the loan period by an additional 3 months. During this extended period, the interest rate was 15%, with
associated fees and commissions of 3% per annum for the application fee and an origination fee of 3% per annum. On February 26, 2024, IMC Holdings and ADI signed an additional amendment to the ADI Agreement, extending the loan period until April 15,
2024, with the same terms as the first amendment, as specified above.
On March 5, 2025, IMC Holdings and ADI signed an amendment postponing the repayment of the remaining ADI Loan to June 30, 2025. The Company repaid NIS 6 million (approximately $2,575) of the
outstanding balance of the ADI Loan by using the proceeds from the November 2024 Offering (defined below). The parties are currently in discussions about the repayment of the outstanding balance of the ADI Loan.
April 2024 Loan Agreement
On April 17, 2024, Pharm Yarok entered into a loan agreement with a non-financial institution in the amount of NIS 3,000 (approximately $1,082) (the “April 2024 Loan”).
The April 2024 Loan bore an annual interest rate of 15% and matured 12 months from the date of issuance. The April 2024 Loan was secured by the following collaterals and guarantees: (a) a first-ranking floating charge over the assets of Pharm Yarok
(b) a first-ranking fixed charge over the holdings (23.3%) of its subsidiary, IMC Holdings, of Xinteza; (c) a personal guarantee by Mr. Oren Shuster, the Company’s Chief Executive Officer and director; and (D) a guarantee by the Company.
On January 30, 2025, Pharm Yarok and the lender signed an amendment to the April 2024 Loan pursuant to which Pharm Yarok paid NIS 1,000 (approximately $393) on January 31, 2025, and the remaining
loan principal amount of NIS 2,000 (approximately $844) was extended until June 30, 2026.
May 2024 Convertible Debenture Offering
On May 26, 2024, the Company closed a non-brokered private placement (the “May 2024 Private Placement”) of secured convertible debentures (each, a “May 2024 Debentures”) for aggregate proceeds of $2,092. The May 2024 Debentures were issued to holders of short-term loans and obligations owed by the Company or its wholly owned subsidiaries and were inclusive of
a 10% extension fee in full settlement of such debt to the holders. The May 2024 Debentures matured on May 26, 2025 and have not incurred interest. The May 2024 Debentures were convertible into Common Shares at a conversion price of $5.1 per Common
Share (following the July 2024 Consolidation). The Company was entitled through the term of the May 2024 Debentures to early repayment of the May 2024 Debentures for cash amount of $2,092. Mr. Oren Shuster, the Company’s Chief Executive Officer and
director, subscribed for an aggregate of $237 of May 2024 Debentures in the May 2024 Private Placement.
Effective May 26, 2025, following the shareholders' approval, the Company and the creditors agreed to extend the term of the May 2024 Debentures until May 25, 2026, subject to extension fee of
additional 10%, such that upon maturity of the May 2024 Debentures, the principal to be paid will be $2,301. The conversion price was determined as $2.61 per Common Share and the Company was entitled to through the term of the May 2024 Debentures to
early repayment of the May 2024 Debentures for cash amount of $2,301.
23
|
Management’s Discussion and Analysis
|
|
July 2024 Short-term Loan Agreement
On July 1, 2024, IMC Holdings entered into a short-term loan agreement with a non-financial institution in the amount of NIS 3,000 (approximately $1,113). Such loan bore interest at an annual rate of
12% and originally matured 62 days from the date of signing the loan agreement. IMC Holdings and the lender executed amendments to the loan agreement, each extending the maturity date, thereby postponing the
maturity date to February 28, 2026, under the same terms and conditions. The loan, including the accrued interest, was fully paid by February 28, 2026.
Payment schedule with third party
On July 30, 2024, the Company entered into an acknowledgment and payment schedule agreement with a third party regarding unpaid fees, charges, and disbursements for services rendered to us. According
to the terms of the agreement, we shall pay $54,000 on the first business day of each month for twenty-four (24) months, with the first payment due on November 1, 2024.
April 2025 Short-Term Loan Agreement
On April 29, 2025, IMC Holdings entered into a short-term loan agreement with a non-financial institution in the amount of NIS 1,000 (approximately $375). The loan bears interest at an annual rate of
17% and matures 12 months from the date of signing the loan agreement.
May 2025 Short-Term Loan Agreement
On May 25, 2025, IMC Holdings entered into a short-term loan agreement with a non-financial institution in the amount of NIS 350 (approximately $131). This loan bears interest at an annual rate of
17% and matures on June 25, 2025. The parties extended the maturity date to June 25, 2026.
July 2025 Loan Agreement
On July 6, 2025, the Company entered into a loan agreement with L.I.A. Pure Capital Ltd. (the “Lender”) for an aggregate amount of US$2 million. Pursuant to
the Loan Agreement, the Company received an amount of US$1 million (the “First Loan Tranche”) and may receive an additional amount of US$1 million (the “Second Loan Tranche”)
no later than 60 days from signing the Loan Agreement, subject to satisfying certain conditions. Pursuant to the Loan Agreement, the Lender has a right to recommend a director to be appointed to the Company’s Board.
The loan bears annual interest at a rate of 8% (plus VAT) and is repayable in full, including accrued interest, by June 30, 2026. In the event of non-repayment by that date, default interest at a
rate of 15% per annum (plus VAT) will apply. The loan is secured by a pledge over 100% of the shares of IMC Holdings Ltd., the Company’s wholly owned subsidiary, with the pledged shares held directly by us.
The Company has also committed to raise at least US$ 3 million in capital within 60 days of signing the Loan Agreement, through a public offering underwritten or distributed by Aegis Capital Corp. If
the Company raises US$4 million or more, the Lender will not be obligated to provide the second loan tranche. In the event that the Company raises US$5 million or more, the Lender may exercise an acceleration right, requiring the Company to repay the
outstanding loan within 45 business days of written notice.
24
|
Management’s Discussion and Analysis
|
|
July 2025 Short-Term Loan Agreement
On July 16, 2025, Rosen High Way entered into a short-term loan agreement, with a non-financial institution in the amount of NIS 500 (approximately $202). This loan bears interest at an annual rate
of 17% and matures on July 16, 2026.
October 2025 Short-Term Loan Agreement
On October 5, 2025, IMC Holdings entered into a short-term loan agreement with a non-financial institution in the amount of NIS 500 (approximately $211). This loan bears interest at an annual rate of
17% and matured on November 13, 2025. The Company is currently in discussions with the lender regarding a potential extension of the loan.
January 2026 Note Purchase Agreements, Convertible Notes and Warrants
On January 7, 2026, the Company entered into a Note Purchase Agreement (the “Purchase Agreement”) with an institutional investor (the “Investor”), pursuant to which the Company issued to the Investor: (A) a convertible note (the “Note”) in the principal amount of US$1,710 (the “Subscription Amount”), which is convertible into the Company’s Common Shares at a price equal to ninety percent (90%) of the Subscription Amount and (B) a warrant to purchase up to 228,150 Common Shares, which is the number equal to
thirty-three and one-third percent (33⅓%) of the Subscription Amount divided by an exercise price of $3.45 per Common Share (the “First Transaction”). The First Transaction closed on January 20, 2026.
In addition, on January 20, 2026, the Company entered into an additional Note Purchase Agreement (the “Additional Purchase Agreement” and, together with the
Purchase Agreement, the “Purchase Agreements”) with the Investor, pursuant to which the Company issued to the Investor: (A) a convertible note (the “Second Note” and,
together with the Note, the “Notes”) in the principal amount of US$703 (the “Additional Subscription Amount” and, together with the Subscription Amount, the “Subscription Amounts”) which is convertible into Common Shares at a price equal to 90% of the Additional Subscription Amount and (B) a warrant to purchase up to 93,671 Common Shares, which is the number equal to
thirty-three and one-third percent (33⅓%) of the Additional Subscription Amount divided by an exercise price of $3.45 per Common Share (the “Second Transaction” and, together with the First Transaction, the “Offerings”). The Second Transaction closed on January 21, 2026.
The Company intends to use the net proceeds of $2,172 received from the Offerings for debt repayment and general corporate purposes.
25
|
Management’s Discussion and Analysis
|
|
Each Note bears an interest rate of 8.0% per annum accruing from the closing date of the First Transaction and the Second Transaction, as applicable, (which shall increase to 14.0% upon the
occurrence of an Event of Default, as defined in the Notes). The Notes are not repayable in cash and the Company’s obligations thereunder will be satisfied solely through the issuance of the Company’s Common Shares upon conversion of the Notes in
accordance with their terms.
The number of Common Shares issuable upon any conversion of principal amount under the Notes is determined by dividing the applicable conversion amount by the conversion price (the “January 2026 Notes Conversion Price”). The January 2026 Notes Conversion Price is equal to the lower of (i) the Fixed Price, as
defined in each of the Notes, or (ii) 90% of the lowest daily volume-weighted average price of the Common Shares during the 20 consecutive trading days immediately preceding the conversion date, (the “January 2026
Notes Variable Price”), provided, however, that the January 2026 Notes Variable Price will not be lower than the Floor Price, as defined in each of the Notes.
The Fixed Price in the Note and in the Second Note is $0.29 and $1.38, respectively. The Floor Price in the Note and in the Second Note is $0.29 and $0.275, respectively.
The warrants entitle their holder to purchase one Common Share at an exercise price of $3.45 per warrant share. The warrants are exercisable immediately upon their issuance date, January 21, 2026,
for a period of 5 years, until January 21, 2031. If the warrants are not exercised by their applicable expiry date, they will expire and be of no further force or effect. The warrants and the warrant shares may not be traded for a period of four
months, unless permitted under applicable securities legislation.
The Notes include customary limitations on conversion, including a beneficial ownership cap of 4.99% of the outstanding Common Shares following the conversion.
The Company’s authorized share capital as of March 31, 2026, consists of an unlimited number of Common shares without a par value of 6,223,323. The Common Shares confer upon their holders the right
to participate in the general meeting, with each Common Share carrying the right to one vote on all matters. The Common Shares also allow holders to receive dividends if declared and to participate in the distribution of surplus assets in the case of
liquidation of the Company.
OTHER SECURITIES
As of March 31, 2026, the Company also has the following outstanding securities that are convertible into, exercisable or exchangeable for, voting or equity securities of the Company: 28,269 Options,
791,571 unlisted Warrants, 2,511,821 Listed Warrants, 200,000 Pre-Funded Warrants and 256,215 Debentures.
26
|
Management’s Discussion and Analysis
|
|
The following table highlights the Company’s cash flow activities for the three months ended March 31, 2026, and 2025:
|
For the Three Months Ended
March 31,
|
||||||||
|
2026
|
2025
|
|||||||
|
Net cash provided by (used in):
|
||||||||
|
Operating activities
|
$
|
(186
|
)
|
$
|
4,463
|
|||
|
Investing activities
|
$
|
429
|
$
|
-
|
||||
|
Financing activities
|
$
|
(933
|
)
|
$
|
(2,872
|
)
|
||
|
Effect of foreign exchange
|
$
|
(1,033
|
)
|
$
|
(1,065
|
)
|
||
|
Increase (decrease) in cash
|
$
|
(1,723
|
)
|
$
|
526
|
|||
Operating activities used cash of $186 and provided cash of $4,463 for the three months ended March 31, 2026 and 2025, respectively. Net cash from operating activities primarily reflects the
Company’s operating loss, changes in working capital and non-cash items. The change during the quarter was primarily driven by changes in trade receivables, inventory and trade payables.
Investing activities provided cash of $429 and $nil for the three months ended March 31, 2026, and 2025, respectively, primarily driven by changes in restricted cash.
Financing activities used cash of $933 and $2,872 for the three months ended March 31, 2026 and 2025, respectively. Financing cash flows during 2026 primarily reflected proceeds from the issuance of
convertible notes, borrowings and repayments under credit facilities, offset by proceeds from discounted checks and cash paid for interest.
SELECTED ANNUAL INFORMATION
– CONTINUING OPERATIONS
|
For the year ended
|
December 31,
2025 |
December 31,
2024 |
December 31, 2023
|
|||||||||
|
Revenues
|
$
|
54,731
|
$
|
54,031
|
$
|
48,804
|
||||||
|
Net Loss
|
$
|
(11,750
|
)
|
$
|
(11,771
|
)
|
$
|
(10,228
|
)
|
|||
|
Basic net income (Loss) per share:
|
$
|
(2.67
|
)
|
$
|
(4.51
|
)
|
$
|
(4.45
|
)
|
|||
|
Diluted net income (Loss) per share:
|
$
|
(2.67
|
)
|
$
|
(4.51
|
)
|
$
|
(4.45
|
)
|
|||
|
Total assets
|
$
|
31,736
|
$
|
39,188
|
$
|
48,813
|
||||||
|
Total non-current liabilities
|
$
|
1,345
|
$
|
1,124
|
$
|
2,267
|
||||||
27
|
Management’s Discussion and Analysis
|
|
The following tables set out certain financial information for each of the Company’s prior quarterly reporting periods:
|
For the quarters ended
|
March 31,
2026 |
December 31,
2025 |
September 30,
2025 |
June 30,
2025 |
||||||||||||
|
Revenues
|
$
|
8,679
|
$
|
15,684
|
$
|
13,851
|
$
|
12,696
|
||||||||
|
Net Profit (loss)
|
$
|
(2,467
|
)
|
$
|
(7,866
|
)
|
$
|
(3,865
|
)
|
$
|
(194
|
)
|
||||
|
Basic net income (loss) per share:
|
$
|
(0.38
|
)
|
$
|
(1.20
|
)
|
$
|
(0.75
|
)
|
$
|
(0.09
|
)
|
||||
|
Diluted net income (loss) per share:
|
$
|
(0.38
|
)
|
$
|
(1.20
|
)
|
$
|
(0.75
|
)
|
$
|
(0.09
|
)
|
||||
|
For the quarters ended
|
March 31,
2025 |
December 31,
2024 |
September 30,
2024 |
June 30,
2024 |
||||||||||||
|
Revenues
|
$
|
12,500
|
$
|
13,335
|
$
|
13,883
|
$
|
14,750
|
||||||||
|
Net income (loss)
|
$
|
175
|
$
|
(1,213
|
)
|
$
|
(1,082
|
)
|
$
|
(3,456
|
)
|
|||||
|
Basic net income (loss) per share:
|
$
|
0.09
|
$
|
(0.32
|
)
|
$
|
(0.41
|
)
|
$
|
(1.36
|
)
|
|||||
|
Diluted net income (loss) per share:
|
$
|
0.09
|
$
|
(0.32
|
)
|
$
|
(0.41
|
)
|
$
|
(1.36
|
)
|
|||||
* Shares Consolidation - On July 12, 2024, the Company consolidated its issued and outstanding Common Shares based on one post-consolidated Common Share for every six pre-consolidated Common Shares.
Post Consolidation, total Common Shares were reduced from 13,394,136 to 2,232,359 Common Shares (after rounding fractional Common Shares).
This MD&A refers to “Gross Margin”, “EBITDA”, and “Adjusted EBITDA”. These financial measures are not recognized measures under IFRS and do not have a standardized meaning prescribed by IFRS and
are, therefore, unlikely to be comparable to similar measures presented by other companies. Rather, these measures are provided as additional information to complement those IFRS measures by providing a further understanding of our results of
operations from management’s perspective. Accordingly, these measures should neither be considered in isolation nor as a substitute for analysis of our financial information reported under IFRS.
Management defines Gross Margin as the difference between revenue and cost of goods sold divided by revenue (expressed as a percentage), prior to the effect of a fair value adjustment for inventory
and biological assets. Management defines EBITDA as income earned or lost from operations, as reported, before interest, tax, depreciation, and amortization.
28
|
Management’s Discussion and Analysis
|
|
Adjusted EBITDA is defined as EBITDA adjusted by removing other non-recurring or non-cash items, including the unrealized change in fair value of biological assets, realized fair value adjustments on
inventory sold in the period, share-based compensation expenses, and revaluation adjustments of financial assets and liabilities measured on a fair value basis. Management believes Adjusted EBITDA is a valuable financial metric to assess its
operating performance on a cash-adjusted basis before the impact of non-recurring or non-cash items. The closest IFRS metric to EBITDA and Adjusted EBITDA is “operating loss”.
The non-IFRS financial measures can provide investors with supplemental measures of our operating performance and thus highlight trends in our core business that may not otherwise be apparent when
relying solely on IFRS measures. We also believe that securities analysts, investors, and other interested parties frequently use non-IFRS financial measures to evaluate issuers. These financial measures are metrics that have been adjusted from the
IFRS statements in an effort to provide readers with a normalized metric in making comparisons more meaningful across the cannabis industry. However, other companies in our industry may calculate this measure differently, limiting their usefulness as
comparative measures.
Our management also uses these non-IFRS financial measures to facilitate operating performance comparisons from period to period and prepare annual operating budgets and forecasts. As Canadian
securities laws require, we reconcile these non-IFRS financial measures to the most comparable IFRS measures.
GROSS MARGIN
|
For the Three Months Ended
March 31,
|
||||||||
|
2026
|
2025
|
|||||||
|
Net revenue
|
$
|
8,679
|
$
|
12,500
|
||||
|
Cost of sales
|
$
|
(7,260
|
)
|
$
|
(9,052
|
)
|
||
|
Gross profit
|
$
|
1,419
|
$
|
3,448
|
||||
|
Gross margin
|
16
|
%
|
28
|
%
|
||||
EBITDA AND ADJUSTED EBITDA
|
For the three Months
Ended March 31,
|
||||||||
|
2026
|
2025
|
|||||||
|
Operating loss
|
$
|
(1,676
|
)
|
$
|
158
|
|||
|
Depreciation & amortization
|
$
|
497
|
$
|
463
|
||||
|
EBITDA
|
$
|
(1,179
|
)
|
$
|
621
|
|||
|
Share-based payments
|
$
|
-
|
$
|
8
|
||||
|
Adjusted EBITDA (non-IFRS)
|
$
|
(1,179
|
)
|
$
|
629
|
|||
29
|
Management’s Discussion and Analysis
|
|
RENTAL LIABILITIES
The table below summarizes the maturity profile of the Group’s lease liabilities based on contractual undiscounted payments (including interest payments):
March 31, 2026:
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
>10 years
|
|||||||||||||
|
Lease liabilities
|
$
|
239
|
$
|
75
|
-
|
-
|
||||||||||
March 31, 2025:
|
Less than one year
|
1 to 5 years
|
6 to 10 years
|
>10 years
|
|||||||||||||
|
Lease liabilities
|
$
|
322
|
$
|
209
|
-
|
-
|
||||||||||
LITIGATION
AND REGULATORY PROCEEDINGS
COVID-19 TEST KITS CLAIM,
DISTRICT COURT OF STUTTGART
On November 19, 2021, Adjupharm filed a statement of claim (the “Claim”) to the District Court of Stuttgart (the “Stuttgart Court”) against Stroakmont & Atton Trading GmbH (“Stroakmont &
Atton”), its shareholders and managing directors regarding a debt owed by Stroakmont & Atton to Adjupharm of approximately EUR 948 thousand for COVID-19 test kits purchased by Stroakmont & Atton from Adjupharm in May 2021. The Claim was
served on December 2, 2021. In January 2022, Stroakmont & Atton filed its statement of defense to the Stuttgart Court in which it essentially stated two main arguments for defense:
| • |
The contractual party of the company was not Stroakmont. The contract with Stroakmont was only concluded as a sham transaction to cover up a contract with a company named Uniclaro GmbH (“Uniclaro”).
Therefore, Stroakmont is not the real purchaser rather than Uniclaro.
|
| • |
The Company allegedly placed an order with Uniclaro for a total of 4.3 million Clongene COVID-19 tests, of which Uniclaro claims to have a payment claim against the Company for a partial delivery of 380,400 Clongene COVID-19 tests in a
total amount of EUR 941,897. Uniclaro has assigned this alleged claim against the Company to Stroakmont & Atton Trading GmbH, and Stroakmont & Atton Trading GmbH has precautionary declared a set-off against the Company’s claim.
|
30
|
Management’s Discussion and Analysis
|
|
On March 22, 2022, Adjupharm filed a response to Stroakmont & Atton’s statement of defense and rejected both allegations with a variety of legal arguments and facts and also offered evidence to the contrary in the
form of testimony from the witnesses in question.
The burden of proof for both allegations lies with the opponents and they offered evidences to the court in the form of testimony from certain witnesses.
A court hearing with witnesses was held on January 11, 2023 and February 22, 2023, where witnesses testified. According to the court the witnesses were not able to provide the required evidence for
the allegation regarding the sham transaction with Stroakmont. On April 5, 2023, Stuttgart Court announced its decision (the “Test Kits Judgment”) and sentenced Stroakmont to pay to Adjupharm EUR 948 thousand plus interest of 5% above the German
basis rate since May 8, 2021. In addition, Stroakmont was sentences to pay Adjupharm EUR 7 thousand plus interest at 5% above the German basis rate since December 14, 2021.
The directors of Stroakmont, Mr. Simic and Mr. Lapeschi, were not sentenced and in this respect, the COVID-19 Test Kit Claim was dismissed against them with regard to their personal liability.
Adjupharm shall pay 2/3 of the Stuttgart Court expenses and the out-of-court expenses of Mr. Simic and Mr. Lapeschi. Stroakmont shall bear 1/3 of the Stuttgart Court expenses and 1/3 of the out-of-court expenses of Adjupharm. The remaining
out-of-court expenses shall be borne by each party.
Furthermore, the court did not decide on the counterclaims from an alleged order by Adjupharm for 4.3 million Clongene tests due to a set-off prohibition. This set-off prohibition follows from a
jurisdiction agreement concluded between Adjupharm and Uniclaro, which determined the courts in Hamburg to be the competent court to decide about such allegations.
The judgment is not final. An Appeal by Stroakmont & Atton was not successful. Adjupharm initiated enforcement measurements and pledged the bank account of Stroakmont & Atton, however there
is no information about the possible amount in the bank account.
In the meantime, after the pledge of the bank account, Stroakmont & Atton filed for insolvency proceedings and the court ordered not to transfer any pledged amount to Adjupharm until an
insolvency administrator is appointed and will decide about the further steps in the insolvency proceedings. According to the information available to the Company the insolvency proceedings were not commenced yet and an insolvency administrator was
not yet appointed.
UNICLARO
GMBH VS. ADJUPHARM
On December 22, 2022, Uniclaro filed a statement of claim against Adjupharm with the district court in Hamburg, pursuant to which Uniclaro is claiming the purchase price for 300,000 COVID-19 rapid
tests in the total amount of approximately EUR 1,046 thousand (approximately $1,540), including VAT, which Uniclaro has in its storage.
Uniclaro alleged in this lawsuit that Adjupharm placed an order for 4.3 million Covid-19 rapid tests of the brand “Clongene”. Furthermore, Uniclaro claims that the order was placed verbally on March
23, 2021 and that Adjupharm has already paid for a portion of these tests and received them, but not yet the entire 4.3 million tests. They reserve the right to extend the lawsuit for the remaining amount (which they did not specify).
31
|
Management’s Discussion and Analysis
|
|
On February 23, 2023, the Company provided its statement of defense to the court. The statement of defense contains similar arguments to reject the allegations in this respect as in the court
proceedings in Stuttgart about the counterclaims.
On February 14, 2024, a court hearing took place before the district court of Hamburg, at which the court first heard the managing directors of Uniclaro and Adjupharm.
On April 24, 2024, the Regional Court of Hamburg announced its decision under which (i) Adjupharm was not sentenced. Uniclaro's lawsuit for payment of approximately EUR 1,046 thousand in exchange for
delivery of 300,000 Clungene tests was dismissed and (ii) Uniclaro is sentenced to pay Adjupharm approximately EUR 54 thousand plus interest at a rate of 5% above the German basis rate since January 17, 2023.
The judgement is not yet final. Uniclaro has appealed the judgment and applied for the judgment to be overturned and to sentence Adjupharm in accordance with Uncilaro's original application to pay
the amount of approximately EUR 1,046 thousand (including VAT) in exchange for 300,000 Covid-19 rapid. Furthermore, Uniclaro has requested in its appeal to dismiss Adjupharm's counterclaim.
On January 15, 2026, a court hearing took place before the High Court of Justice of Hamburg. The attorney of Uniclaro did not arrive to the court hearing, hence the court dismissed the appeal and
issued a default judgment against Uniclaro. According to this judgment the initial judgment of the Regional Court of Hamburg on behalf of Adjupharm was confirmed and the appeal was dismissed.
On January 29, 2026, Uniclaro objected against the default judgment and requested to continue the proceedings.
The High Court of Justice of Hamburg will have now to appoint a new court hearing to decide about the objection and whether the default judgment will remain in place.
At this stage, management cannot assess the chances of the claim advancing or the potential outcome of these proceedings. Thus, no provision has been recorded.
FOCUS
FACILITY PLANNING AND CONSTRUCTION LEGAL PROCEEDINGS
On July 11, 2021, we were informed that on June 30, 2021, a claim (the “Claim”) was filed in the Beer Sheva Magistrate Court (the “BSMC”) by the municipal planning and construction committee for the southern Israel against Focus, certain of its directors and officers, including Oren Shuster and Rafael Gabay, and certain landowners, alleging deficiencies in
permitting construction of Focus’s cultivation facility in Sde Avraham, Israel (the “Focus Facility”).
On December 6, 2021, the defendants filed a motion request for dismissal the indictment on the ground of “defense of justice”. The municipal committee filed its response and the defendants
subsequently filed a reply. As of that time, no decision has been rendered on the motion.
A hearing initially scheduled for December 1, 2021 was postponed several times to allow the parties to engage in settlement discussions. The hearing was ultimately held on June 22, 2023 before the
Honorable Judge Orit Kertz, during which the parties were directed to continue negotiations and make diligent efforts to reach a settlement by August 14, 2023, with the municipal committee’s counsel responsible for updating the court on any progress.
Subsequent proceedings were postponed, and a further hearing was scheduled for December 28, 2023.
On January 2, 2024, we announced that the proceedings had concluded on December 28, 2023. Focus was found liable, and a fine of $129 was imposed and fully paid in 2024. The Focus Facility, which was
the subject of the proceedings, had been closed in June 2022 as part of our strategic shift toward import and sales operations.
32
|
Management’s Discussion and Analysis
|
|
CANADIAN
RESTRUCTURING
There were no material developments with respect to the Company’s Canadian restructuring during the three months ended March 31, 2026. For background and a description of the Canadian restructuring
(including the Company’s exit from Canadian operations and related proceedings), please refer to the Company’s Annual Report on Form 20‑F for the fiscal year ended December 31, 2025 (the “Annual Report”),
available on the Company’s profile on SEDAR+ at www.sedarplus.ca and on EDGAR at www.sec.gov/edgar.
THE
REGIONAL LABOR COURT - TEL AVIV (BAT YAM) 17419-04-23
On May 10, 2023, we received notice that a former employee filed a claim with the Regional Labor Court – Tel Aviv (Bat Yam) against three entities, including us.
On April 4, 2024, we filed our statement of defense. A preliminary hearing was held on May 6, 2024. Following the hearing, the plaintiff filed an amended claim, which reduced the claimed amount,
added an additional defendant and included requests for certain documents. On January 21, 2025, the court granted the plaintiff’s document requests, and we have since provided the requested materials. A subsequent hearing was held on June 19, 2025.
The claim remains at an early stage, and we are continuing to evaluate the allegations. At this time, we are unable to assess the likelihood of an adverse outcome or to reasonably estimate any
potential loss. Accordingly, no provision has been recorded in our financial statements.
35 OAK
HOLDINGS LTD – STATEMENT OF CLAIM
On November 17, 2023, we received a copy of the 35 Oak Statement of Claim that was filed in the ONSC by 35 Oak Holdings Ltd., MW Investments Ltd., 35 Oak Street Developments Ltd., Michael Wiener,
Kevin Weiner, William Weiner, Lily Ann Goldstein-Weiner, in their capacity as trustees of the Weiner Family Foundation (collectively the "MYM Shareholder Plaintiffs") against the Company and the Company's Board and officers, (collectively, the "MYM
Defendants").
MYM Shareholder Plaintiffs claims that the MYM Defendants made misrepresentations in its disclosures prior to our transaction with MYM in 2021, as well as oral misrepresentations. The MYM Shareholder
Plaintiffs are claiming damages that amount to approximately $15,000 and aggravated, exemplary and punitive damages in the amount of $1,000.
IM Cannabis has reviewed the complaint and believes that the allegations are without merit.
IM Cannabis, together with some of the Defendants brought, on February 22, 2024, a preliminary motion to strike out several significant parts of the claim. Following prolonged discussions and
negotiations with the plaintiffs, the MYM Shareholder Plaintiffs narrowed their claim, provided further particulars of it, and agreed to resolve the motion on consent. The Plaintiffs have agreed to discontinue or dismiss their claims against a number
of the individual defendants. Their claim will only continue against IM Cannabis, MYM Nutraceuticals Inc., Michael Ruscetta, and Howard Stienberg.
33
|
Management’s Discussion and Analysis
|
|
IM Cannabis delivered a Statement of Defence by March 23, 2026, as did Messrs. Steinberg and Ruscetta.
Given the preliminary stage of this action, it is too early to opine on the merits of the claim or whether it is more likely than not to result in an outflow of funds to the Company and if so, how
much.
We plan to vigorously defend itself against the allegations. At this stage, our management cannot assess the chances of the claim advancing or the potential outcome of this these proceedings.
IM Cannabis had no off-balance sheet arrangements as of March 31, 2026.
TRANSACTIONS
WITH RELATED PARTIES
Approval of Related Party Transactions
All related party transactions are reviewed and approved by our Board of Directors or an independent committee thereof in accordance with applicable corporate governance requirements and, where
applicable, MI 61-101. Unless applicable exemptions from the requirements of MI 61-101 apply, related party transactions are subject to formal valuation and minority shareholder approval. Minority shareholder approval excludes the votes of any
interested party or related parties of an interest party from the votes counted in the shareholder approval.
Employment and Management Agreements
We have entered into written employment or service agreements with certain of our executive officers. These agreements contain customary provisions regarding confidentiality, assignment of inventions
and, where applicable, non-competition, subject to applicable law. We have also entered into indemnification agreements with our directors and officers and maintain directors’ and officers’ liability insurance.
Effective January 15, 2018, we and Ewave entered into a management services agreement (the “Shuster Agreement”) pursuant to which Oren Shuster provides
services as our Chief Executive Officer. Mr. Shuster is employed and compensated by Ewave. Pursuant to the Shuster Agreement, Ewave charges a monthly fee of NIS 108,350 plus VAT (approximately $43,900 plus tax per month). Either party may terminate
the agreement upon three months’ prior notice, during which payments continue. We may terminate the agreement immediately for cause without notice. Ewave, which is jointly owned by Mr. Shuster and Rafael Gabay, is a related party to us.
Equity Compensation
See Item 6B in the Company’s Annual Report– “Compensation” for a description of our Securities-Based Compensation Arrangements.
34
|
Management’s Discussion and Analysis
|
|
Transactions Since January 1, 2023
For purposes of this section, related parties include (i) our directors and executive officers; (ii) beneficial owners of 10% or more of our voting power; (iii) entities controlled by such persons;
and (iv) other persons or entities meeting the definition of a related party under applicable securities laws and IFRS.
IMC Holdings leases a 358 square-meter facility in Kibutz Glil Yam for administrative activities. Since August 2024, IMC Holdings sub leases the facility to Ewave Nadlan International Investments
Ltd, a subsidiary of Ewave Group owned by Mr. Oren Shuater and Refael Gabay, pursuant to a sublease arrangement.
On April 2, 2019, IMC Holdings and Focus entered into t the Focus Agreement, pursuant to which IMC Holdings obtained an option to acquire all ordinary shares of Focus held by Messrs. Shuster and
Gabay. Following IMCA approval on February 26, 2024, IMC Holdings acquired 74% of Focus. On September 2024, the Board engaged an independent third-party valuator to determine the purchase price of the remaining 26% interest. The purchase price was
determined to be NIS 818,740 (the “Focus Purchase Price”). To preserve cash, we agreed to settle the Focus Purchase Price through the issuance of 128,818 Common Shares at a deemed price of C$2.44 per share,
equal to the ten-day VWAP on the CSE prior to shareholder approval. The shares were subject to a four-month and one-day hold period and applicable U.S. Securities Act legends.
Certain insiders participated in tranches of the LIFE Concurrent Offering and LIFE Offering in 2023. These transactions constituted related party transactions under MI 61-101, and we relied on
applicable exemptions based on the relative size of insider participation.
On October 12, 2023, Mr. Shuster loaned NIS 500 (approximately $170) to IMC Holdings. The transaction constituted a related party transaction under MI 61-101. We relied on available exemptions based
on the size of the transaction relative to our market capitalization.
On November 12, 2024, we completed a debt settlement in the amount of US$560,000 with Mr. Shuster in connection with personal guarantees he had provided for certain of our loan obligations. The
independent members of our Board obtained an independent valuation of the benefit derived from such guarantees. To settle this amount and preserve cash, we issued 110,576 Common Shares and 152,701 pre-funded warrants at a deemed price of C$2.88. The
warrants were subsequently exercised following disinterested shareholder approval for Mr. Shuster to become a Control Person.
On May 27, 2025, we completed a non-brokered private placement of secured convertible debentures for aggregate proceeds of C$2,301,174.70. The debentures mature on May 26, 2026 and are convertible at
C$2.61 per Common Share.
Mr. Shuster and Rafael Gabay participated in the offering, subscribing for C$260,935.40 and C$260,278.70, respectively.
We are party to indemnification arrangements with certain directors and officers in connection with prior acquisitions.
Other than the transactions described above and compensation paid in the ordinary course to key management personnel, we had no other material related party transactions during the period covered by
the Company’s Annual Report.
35
|
Management’s Discussion and Analysis
|
|
On March 17, 2026, the Company announced that it entered into a non-binding letter of intent (“LOI”) to acquire 51% of Blackaxe Technologies Spolka z
Organiczona (“Blackaxe”), a Polish technology company specializing in advanced defense and intelligence solutions. The Company indicated that it entered into the LOI as part of its efforts to explore the
introduction of additional business activities and to evaluate opportunities to expand into additional technology-driven sectors beyond its existing medical cannabis operations. Blackaxe’s business is described as including satellite intelligence,
imagery and AI analytics, and counter‑UAS (anti‑drone) solutions based on cyber interception technologies. The proposed transaction is subject to completion of due diligence, negotiation and execution of definitive agreements, satisfaction of
customary closing conditions, and receipt of required regulatory approvals, and there can be no assurance that it will be completed.
MATERIAL
ACCOUNTING POLICIES
The Company’s consolidated financial statements have been prepared in accordance with IFRS, as issued by the IASB, and interpretations of the IFRS Interpretations Committee (“IFRIC”).
Basis of Measurement
These consolidated financial statements have been prepared on a historical cost basis, except for financial instruments presented at fair value through profit or loss. In addition, these audited
financial statements have been prepared using the accrual basis of accounting. The material accounting policies set out below have been applied consistently to the period presented in these audited financial statements.
The Group has elected to present the profit or loss items using the function of expense method.
In the process of applying the significant accounting policies, the Group has made the following judgments which have the most significant effect on the amounts recognized in the financial
statements:
The Functional Currency and the Presentation Currency
The Company prepares its financial statements in accordance with the currency of the country and principal economic environment in which it operates, which constitutes the functional currency from
which it is primarily affected (the “Functional Currency”). Management has determined that the Functional Currency of the Group is the Canadian dollar.
The Group's financial statements are presented in Canadian dollars. Consequently, in accordance with IAS 21, “Accounting for Foreign Exchange Rates”, results of operations of each Group entity were
translated into CAD using the actual action date currency rate and assets and liabilities were translated into CAD using currency rates at period end. Foreign currency translation adjustments are recorded as a component of accumulated other
comprehensive income (loss) within shareholders’ equity.
Upon full or partial disposal of a foreign operation resulting in loss of control in the foreign operation, the cumulative gain (loss) from the foreign operation which had been recognized in other
comprehensive income (loss) is transferred to profit or loss. Upon the partial disposal of a foreign operation that results in the retention of control in the subsidiary, the relative portion of the amount recognized in other comprehensive income
(loss) is reattributed to non-controlling interests.
36
|
Management’s Discussion and Analysis
|
|
JUDGMENTS
Determining the fair value of share-based payment transactions
The fair value of share-based payment transactions is determined upon initial recognition by an acceptable option pricing model. The inputs to the model include share price, exercise price and
assumptions regarding expected volatility, expected life of share option and expected dividend yield.
Discount rate for lease liability:
When the Group is unable to readily determine the discount rate implicit in a lease in order to measure the lease liability, the Group uses an incremental borrowing rate. That rate represents the
rate of interest that the Group would have to pay to borrow over a similar term and with similar security, the funds necessary to obtain an asset of similar value to the right-of-use asset in a similar economic environment. When there are no
financing transactions that can serve as a basis, the Group determines the incremental borrowing rate based on its credit risk, the lease term and other economic variables deriving from the lease contract's conditions and restrictions. In certain
situations, the Group is assisted by an external valuation expert in determining the incremental borrowing rate.
ESTIMATES
AND ASSUMPTIONS
The preparation of the Company’s interim condensed consolidated financial statements in conformity with IFRS Accounting Standards requires management to make accounting estimates and assessments that
involve the use of judgment and that affect the amounts of assets and liabilities presented in the financial statements, the disclosure of contingent assets and liabilities at the dates of the financial statements, the amounts of revenue and expenses
during the reporting periods and the accounting policies adopted by the Company. Actual results could differ from those estimates.
Estimates and underlying assumptions are reviewed on an ongoing basis for reasonableness and relevancy. Where revisions are required, they are recognized in the period in which the estimate is
revised as well as future periods that are affected.
Determining the fair value of unquoted financial assets
The fair value of unquoted financial assets in Level 3 of the fair value hierarchy is determined using valuation techniques, generally using future cash flows discounted at current rates applicable
for items with similar terms and risk characteristics. Changes in estimated future cash flows and estimated discount rates, after consideration of risks such as liquidity risk, credit risk and volatility, are liable to affect the fair value of these
assets.
Impairment of goodwill
The Group reviews goodwill for impairment at least once a year. This requires management to estimate the projected future cash flows from the continuing use of the cash-generating unit (or a group of
cash-generating units) to which the goodwill is allocated and choose a suitable discount rate for those cash flows.
Legal claims
In estimating the likelihood of legal claims filed against the Group entities, the Group management relies on the opinion of its legal counsel. These estimates are based on the legal counsel's best
professional judgment, taking into account the stage of proceedings and legal precedents in respect of the different issues. Since the outcome of the claims may be determined in courts, the results could differ from these estimates.
37
|
Management’s Discussion and Analysis
|
|
ASSESSMENT OF GOING CONCERN
The use of the going concern basis of preparation of the financial statements. At each reporting period, management assesses the basis of preparation of the financial statements. The going concern
basis of presentation assumes that the Company will continue its operations for the foreseeable future and be able to realize its assets and discharge its liabilities and commitments in the normal course of business.
The Group’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outlays for operating expenses and capital expenditures. In year
2026, the company will focus on raising funds and/or debt and will continue with cost savings efforts as well as increased efficiency.
These conditions raise uncertainties that cast significant doubt as to whether the Company will be able to continue as a going concern. The consolidated financial statements do not include any
adjustments relating to the recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
BUSINESS COMBINATIONS AND
GOODWILL
Business combinations are accounted for by applying the acquisition method. The cost of the acquisition is measured at the fair value of the consideration transferred on the acquisition date with the
addition of non-controlling interests in the acquiree. In each business combination, the Company chooses whether to measure the non-controlling interest in the acquiree based on their fair value on the acquisition date or at their proportionate share
in the fair value of the acquiree's net identifiable assets.
Direct acquisition costs are carried to the statement of profit or loss as incurred.
In a business combination achieved in stages, equity interests in the acquiree that had been held by the acquirer prior to obtaining control are measured at the acquisition date fair value while
recognizing a gain or loss resulting from the revaluation of the prior investment on the date of achieving control.
Contingent consideration is recognized at fair value on the acquisition date and classified as a financial asset or liability in accordance with IFRS 9. Subsequent changes in the fair value of the
contingent consideration are recognized in profit or loss. If the contingent consideration is classified as an equity instrument, it is measured at fair value on the acquisition date without subsequent remeasurement.
Goodwill is initially measured at cost which represents the excess of the acquisition consideration and the amount of non-controlling interests over the net identifiable assets acquired and
liabilities assumed. If the resulting amount is negative, the acquirer recognizes the resulting gain on the acquisition date.
CASH
Cash is considered as highly liquid investments, including unrestricted short-term bank deposits with an original maturity of three months or less from the date of investment or with a maturity of
more than three months, but which are redeemable on demand without penalty and which form part of the Group's cash management
38
|
Management’s Discussion and Analysis
|
|
INVENTORIES
Inventories are measured at the lower of cost and net realizable value. The cost of inventories comprises costs of purchase and costs incurred in bringing the inventories to their present location
and condition. Net realizable value is the estimated selling price in the ordinary course of business less estimated costs of completion and estimated costs necessary to make the sale. The Company periodically evaluates the condition and age of
inventories and makes provisions for slow moving inventories accordingly.
Cost of inventories is determined as follows:
Raw materials - at cost of purchase using the "first-in, first-out" method.
Work in progress and finished goods - on the basis of average costs including materials, labor and other direct and indirect manufacturing costs based on normal capacity.
Purchased merchandise and products - using the weighted average cost method or using the "first-in, first-out" method.
REVENUE
RECOGNITION
Revenue from contracts with customers is recognized when the control over the goods or services is transferred to the customer. The transaction price is the amount of consideration that is expected
to be received based on the contract terms, excluding amounts collected on behalf of third parties (such as taxes).
In determining the amount of revenue from contracts with customers, the Group evaluates whether it is a principal or an agent in the arrangement. The Group is a principal when the Group controls the
promised goods or services before transferring them to the customer. In these circumstances, the Group recognizes revenue for the gross amount of consideration. When the Group is an agent, it recognizes revenue for the net amount of the
consideration, after deducting the amount due to the principal.
Revenue from the sale of goods
Revenue from the sale of cannabis products is generally recognized when control over the goods has been transferred to the customer. Payment is typically due prior to or upon delivery, and revenue is
recognized upon the satisfaction of the performance obligation. The Group satisfies its performance obligation and transfers control upon delivery.
Bill-and-hold arrangements
Due to strict regulations of security, storage and handling large quantities of cannabis products, the Group's customers may request the Group to retain physical possession of a sold product until it
is delivered to the customer at a future point in time. Revenue from bill-and-hold sales is recognized before the product is physically delivered to the customer when all the following criteria are met:
The reason for the bill-and-hold arrangement is substantive (for example, the customer has requested the arrangement).
The product is identified separately as belonging to the customer.
The product currently is ready for physical delivery to the customer.
The Group does not have the ability to use the product by selling it or delivering it to another customer.
39
|
Management’s Discussion and Analysis
|
|
TAXES
ON INCOME
Current or deferred taxes are recognized in profit or loss, except to the extent that they relate to items which are recognized in other comprehensive income or equity.
Current taxes
The current tax liability is measured using the tax rates and tax laws that have been enacted or substantively enacted by the reporting date as well as adjustments required in connection with the tax
liability in respect of previous years.
Deferred taxes
Deferred taxes are computed in respect of temporary differences between the carrying amounts in the financial statements and the amounts attributed for tax purposes.
Deferred taxes are measured at the tax rate that is expected to apply when the asset is realized or the liability is settled, based on tax laws that have been enacted or substantively enacted by the
reporting date.
Deferred tax assets are reviewed at each reporting date and reduced to the extent that it is not probable that they will be utilized. Deductible carry forward losses and temporary differences for
which deferred tax assets had not been recognized are reviewed at each reporting date and a respective deferred tax asset is recognized to the extent that their utilization is probable.
Deferred taxes in respect of investment property that is held to recover substantially all of the economic benefits embedded in the investment property through sale and not through use are measured
in accordance with the expected manner of recovery of the base asset, based on sale rather than use. When the Company owns an investment in a single property company and the manner in which the Company expects to dispose of the investment is by
selling the shares of the property company rather than by selling the property itself, the Company recognizes deferred taxes for both inside temporary differences arising from the difference between the carrying amount of the property and its tax
basis, and for outside temporary differences arising from the difference between the tax basis of the investment and the
Company's carrying amount of the net assets of the investment in the consolidated financial statements.
Taxes that would apply in the event of the disposal of investments in investees have not been considered in computing deferred taxes, as long as the disposal of the investments in investees is not
probable in the foreseeable future. Also, deferred taxes that would apply in the event of distribution of earnings by investees as dividends have not been considered in computing deferred taxes, since the distribution of dividends does not involve an
additional tax liability or since it is the Company's policy not to initiate distribution of dividends from a subsidiary that would trigger an additional tax liability.
Taxes on income that relate to distributions of an equity instrument and to transaction costs of an equity transaction are accounted for pursuant to IAS 12.
Deferred taxes are offset if there is a legally enforceable right to offset a current tax asset against a current tax liability and the deferred taxes relate to the same taxpayer and the same
taxation authority.
40
|
Management’s Discussion and Analysis
|
|
NON-CURRENT
ASSETS OR DISPOSAL GROUP HELD FOR SALE AND DISCONTINUED OPERATIONS
Non-current assets or a disposal group are classified as held for sale if their carrying amount will be recovered principally through a sale transaction rather than through continuing use. For this
to be the case, the assets must be available for immediate sale in their present condition, the Company must be committed to a sale plan, there must be a program to locate a buyer, and it is highly probable that a sale will be completed within one
year from the date of classification.
Before these assets are classified as available for sale, they are measured in accordance with the Group's accounting policy. After classification as held for sale, these assets are measured at the
lower of their carrying amount and fair value less costs to sell and presented separately in the statement of financial position. From the date of their initial classification, these assets are not depreciated.
The Company recognizes an impairment loss in respect of an asset or group of assets in accordance with IAS 36. An impairment loss and subsequent remeasurement gains or losses are recorded in profit
or loss. Gains are recognized up to the cumulative amount of the previously recognized impairment loss.
Other comprehensive income (loss) in respect of an assets or a group of non-current assets that are classified as held for sale is presented separately in equity.
When the Company no longer plans to sell an asset in a sale transaction, it ceases the classification of the asset as held for sale and measures it at the lower of its carrying amount had it not been
classified as held for sale or the recoverable amount of the asset on the date of the decision not to sell the asset.
When the Company is committed to a sale plan that results in loss of control over a subsidiary, the subsidiary's entire assets and liabilities are classified as held for sale, regardless of whether
the Company will retain any non-controlling interests in the subsidiary.
A discontinued operation is a component of the Company that represents a separate major line of business operation or geographical area of operations that either has been disposed of or is classified
as held for sale. The operating results relating to the discontinued operation (including comparative data) are presented separately in the statement of profit or loss, net of the tax effect.
POST-EMPLOYMENT
BENEFITS
According to the labor laws and Severance Pay Law in Israel, the Israeli entities are required to pay compensation to an employee upon dismissal or retirement or to make current contributions in
defined contribution plans pursuant to section 14 to the Severance Pay Law, as specified below. The Israeli entities’ liability is accounted for as a post-employment benefit only for employees not under section 14. The computation of the Israeli
entities’ employee benefit liability is made in accordance with a valid employment contract or a collective employees agreement based on the employee's salary and employment term which establish the entitlement to receive the compensation.
As of March 31, 2026, all employees in Israel are under section 14.
LEASES
The Group accounts for a contract as a lease when the contract terms convey the right to control the use of an identified asset for a period in exchange for consideration.
For leases in which the Group is the lessee, the Group recognizes on the commencement date of the lease a right-of-use asset and a lease liability, excluding leases whose term is up to 12 months and
leases for which the underlying asset is of low value. For these excluded leases, the Group has elected to recognize the lease payments as an expense in profit or loss on a straight-line basis over the lease term. In measuring the lease liability,
the Group has elected to apply the practical expedient in the standard and does not separate the lease components from the non-lease components included in a single contract.
41
|
Management’s Discussion and Analysis
|
|
On the commencement date, the lease liability includes all unpaid lease payments discounted at the interest rate implicit in the lease, if that rate can be readily determined, or otherwise using the
Group's incremental borrowing rate. After the commencement date, the Group measures the lease liability using the effective interest rate method.
On the commencement date, the right-of-use asset is recognized in an amount equal to the lease liability plus lease payments already made on or before the commencement date and initial direct costs
incurred. The right-of-use asset is measured by applying the cost model and amortized over the shorter of its useful life and the lease term. The amortization periods are up to 5.5 years for premises and 3 years for vehicles.
VARIABLE
LEASE PAYMENTS THAT DEPEND ON AN INDEX
On commencement date, the Group uses the index rate prevailing on the commencement date to calculate the future lease payments. For leases in which the Group is the lessee, the aggregate changes in
future lease payments resulting from a change in the index are discounted (without a change in the discount rate applicable to the lease liability) and recorded as an adjustment of the lease liability and the right-of-use asset, only when there is a
change in the cash flows resulting from the change in the index (that is, when the adjustment to the lease payments takes effect).
Lease extension and termination options
A non-cancelable lease term includes both the periods covered by an option to extend the lease when it is reasonably certain that the extension option will be exercised, and the periods covered by a
lease termination option when it is reasonably certain that the termination option will not be exercised.
In the event of any change in the expected exercise of the lease extension option or in the expected non-exercise of the lease termination option, the Group remeasures the lease liability based on
the revised lease term using a revised discount rate as of the change date of expectations. The total change is recognized in the carrying amount of the right-of-use asset until it is reduced to zero, and any further reductions are recognized in
profit or loss.
Lease modifications
If a lease modification does not reduce the scope of the lease and does not result in a separate lease, the Company remeasures the lease liability based on the modified lease terms using a revised
discount rate as of the modification date and records the change in the lease liability as an adjustment to the right-of-use asset.
If lease modification reduces the lease scope, the Company recognizes a gain or loss arising from the partial or full reduction of the carrying amount of the right-of-use asset and the lease
liability. The Company subsequently remeasures the carrying amount of the lease liability according to the revised lease terms, at the revised discount rate at the modification date and records the change in the lease liability as an adjustment to
the right-of-use asset.
42
|
Management’s Discussion and Analysis
|
|
PROPERTY,
PLANT AND EQUIPMENT, NET
Property, plant and equipment are measured at cost, including directly attributable costs, less accumulated depreciation, accumulated impairment losses and excluding day-to-day servicing expenses.
Cost includes spare parts and auxiliary equipment that are used in connection with plant and equipment.
A part of an item of property, plant and equipment with a cost that is significant in relation to the total cost of the item is depreciated separately using the component method.
Depreciation of property, plant, and equipment is dependent upon estimates of useful lives and residual values, which are determined through the exercise of judgment and calculated on a straight-line
basis over the useful lives of the assets at annual rates.
Leasehold improvements are depreciated on a straight-line basis over the shorter of the lease term and the useful life of the improvement.
The useful life, depreciation method and residual value of an asset are reviewed at least each year-end, and any changes are accounted for prospectively as a change in accounting estimate.
Depreciation of an asset ceases at the earlier of the date that the asset is classified as held for sale and the date that the asset is derecognized.
INTANGIBLE
ASSETS
Separately acquired intangible assets are measured on initial recognition at cost including directly attributable costs. Intangible assets acquired in a business combination are measured at fair
value at the acquisition date.
Intangible assets with a finite useful life are amortized over their useful life and reviewed for impairment whenever there is an indication that the asset may be impaired. The amortization period
and the amortization method for an intangible asset are reviewed at least at the end of each year.
Amortization is calculated on a straight-line basis over the useful life of the assets.
IMPAIRMENT
OF NON-FINANCIAL ASSETS
The Group evaluates the need to record an impairment of non-financial assets whenever events or changes in circumstances indicate that the carrying amount is not recoverable. If the carrying amount
of non-financial assets exceeds their recoverable amount, the assets are reduced to their recoverable amount. The recoverable amount is the higher of fair value less costs of sale and value in use. In measuring value in use, the expected future cash
flows are discounted using a pre-tax discount rate that reflects the risks specific to the asset. The recoverable amount of an asset that does not generate independent cash flows is determined for the cash-generating unit to which the asset belongs.
Impairment losses are recognized in profit or loss. An impairment loss of an asset, other than goodwill, is reversed only if there have been changes in the estimates used to determine the asset's recoverable amount since the last impairment loss was
recognized. Reversal of an impairment loss, as above, shall not be increased above the lower of the carrying amount that would have been determined (net of depreciation or amortization) had no impairment loss been recognized for the asset in prior
years and its recoverable amount. The reversal of impairment loss
of an asset presented at cost is recognized in profit or loss.
43
|
Management’s Discussion and Analysis
|
|
The following criteria are applied in assessing the impairment of these specific assets:
Goodwill in respect of subsidiaries
The Group reviews goodwill for impairment once a year, on December 31, or more frequently if events or changes in circumstances indicate impairment.
Goodwill is tested for impairment by assessing the recoverable amount of the cash-generating unit (or group of cash-generating units) to which the goodwill has been allocated. The Company identified
the operations in Israel, Canada, and Germany as three separate cash-generating units.
An impairment loss is recognized if the recoverable amount of the cash-generating unit (or group of cash-generating units) to which goodwill has been allocated is less than the carrying amount of the
cash-generating unit (or group of cash-generating units). Any impairment loss is allocated first to goodwill. Impairment losses recognized for goodwill cannot be reversed in subsequent periods.
FINANCIAL
INSTRUMENTS
Financial assets:
Financial assets are measured upon initial recognition at fair value plus transaction costs that are directly attributable to the acquisition of the financial assets, except for financial assets
measured at fair value through profit or loss in respect of which transaction costs are recorded in profit or loss.
The Group classifies and measures debt instruments in the financial statements based on the following criteria:
The Group’s business model for managing financial assets; and
The contractual cash flow terms of the financial asset.
Debt instruments are measured at amortized cost
The Group’s business model is to hold the financial assets to collect their contractual cash flows, and the contractual terms of the financial assets give rise on specified dates to cash flows that
are solely payments of principal and interest on the principal amount outstanding. After initial recognition, the instruments in this category are measured according to their terms at amortized cost using the effective interest rate method, less any
provision for impairment.
Debt instruments are measured at fair value through profit or loss
A financial asset that is a debt instrument does not meet the criteria for measurement at amortized cost or at fair value through other comprehensive income. After initial recognition, the financial
asset is measured at fair value and gains or losses from fair value adjustments are recognized in profit or loss.
Equity instruments
Investments in equity instruments do not meet the above criteria and are, accordingly, measured at fair value through profit or loss. Dividends from investments in equity instruments are recognized
in profit or loss when the right to receive the dividends is established.
Impairment of financial assets
At the end of each reporting period, the Group evaluates the loss allowance for financial debt instruments measured at amortized cost. The Group has short-term financial assets, principally trade
receivables, in respect of which the Group applies a simplified approach and measures the loss allowance in an amount equal to the lifetime expected credit losses. The impairment loss, if any, is recognized in profit or loss with a corresponding
allowance offset from the assets' carrying amount.
44
|
Management’s Discussion and Analysis
|
|
Financial liabilities:
Financial liabilities measured at amortized cost:
Financial liabilities are initially recognized at fair value less transaction costs that are directly attributable to the issue of the financial liability.
After initial recognition, the Group measures all financial liabilities at amortized cost using the effective interest rate method, except for financial liabilities at fair value through profit or
loss or when a contingent consideration recognized by an acquirer in a business combination to which IFRS 3 applies.
Financial liabilities measured at fair value through profit or loss:
At initial recognition, the Group measures financial liabilities that are not measured at amortized cost at fair value. Transaction costs incurred at initial recognition are recognized in profit or
loss.
After initial recognition, changes in fair value are recognized in profit or loss.
Derecognition of financial liabilities:
Financial liability is derecognized only when it is extinguished, that is when the obligation specified in the contract is discharged or cancelled or expires. Financial liability is extinguished when
the debtor discharges the liability by paying in cash, other financial assets, goods or services; or is legally released from the liability.
FAIR
VALUE MEASUREMENT
Fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.
Fair value measurement is based on the assumption that the transaction will take place in the asset's or the liability's principal market or, in the absence of a principal market, in the most
advantageous market.
The fair value of an asset or a liability is measured using the assumptions that market participants would use when pricing the asset or liability, assuming that market participants act in their
economic best interest.
Fair value measurement of a non-financial asset considers a market participant's ability to generate economic benefits by using the asset in its highest and best use or by selling it to another
market participant that would use the asset in its highest and best use.
45
|
Management’s Discussion and Analysis
|
|
The Group uses appropriate valuation techniques in the circumstances and for which sufficient data are available to measure fair value, maximizing the use of relevant observable inputs and minimizing
the use of unobservable inputs.
|
Level 1
|
-
|
quoted prices (unadjusted) in active markets for identical assets or liabilities.
|
|
Level 2
|
-
|
inputs other than quoted prices included within Level 1 that are observable directly or indirectly.
|
|
Level 3
|
-
|
inputs that are not based on observable market data (valuation techniques that use inputs that are not based on observable market data).
|
All assets and liabilities measured at fair value or for which fair value is disclosed are categorized into levels within the fair value hierarchy based on the lowest level input that is significant
to the entire fair value measurement:
PROVISIONS
A provision in accordance with IAS 37 is recognized when the Group has a present obligation (legal or constructive) resulting from past events, it is probable that an outflow of resources embodying
economic benefits will be required to settle the obligation and a reliable estimate can be made of the amount of the obligation. When the Group expects part or all the expense to be reimbursed, the reimbursement is recognized as a separate asset but
only when the reimbursement is virtually certain. The expense is recognized in statement of operations net of any reimbursement.
The amount recognized as a provision should be the best estimate of the expenditure required to settle the present obligation at the end of the reporting period. The best estimate of the expenditure
required to settle the present obligation is the amount that the Company would rationally pay to settle the obligation at the end of the reporting period or to transfer it to a third party at that time. Where the provision being measured involves a
large population of items, the obligation is estimated by weighting all possible outcomes by their associated probabilities. Where a single obligation is being measured, the individual most likely outcome may be the best estimate of the provision.
ISSUANCE
OF A UNIT OF SECURITIES
When multiple instruments are issued in a single transaction (package issuance), the total net proceeds from the transaction are allocated among the individual freestanding instruments identified.
The allocation occurs after identifying all freestanding instruments and the subsequent measurement basis for those instruments.
Financial instruments that are required to be subsequently measured at fair value (such as derivative liabilities) are measured at fair value and the remaining consideration is allocated to other
financial instruments that are not required to be subsequently measured at fair value (i.e. liabilities measured at amortized cost, common shares and warrants eligible for equity classification), based on the relative fair value basis for such
instruments.
Issuance costs allocated to financial instruments that are required to be subsequently measured at fair value immediately expensed. Issuance costs allocated to shares and warrants classified as
equity components and are recorded as a reduction of additional paid-in capital. Issuance costs allocated to financial liabilities measured at amortized cost are recorded as a discount and accreted over the contractual term of the financial
instrument using the effective interest method.
46
|
Management’s Discussion and Analysis
|
|
CONVERTIBLE
DEBENTURES
Upon initial recognition of convertible debentures and similar instruments, the Company considers the provisions of ASC 815-40, “Derivatives and Hedging - Contracts in Entity’s Own Equity” (“ASC
815-40”) in order to determine whether the conversion features embedded within the convertible instrument should be separated from the host instrument.
When it is determined that an embedded derivative required to be bifurcated (such as embedded conversion feature that does not qualify for equity classification), the Company recognized the embedded
derivative bifurcated as a separate derivative liability upon initial recognition and on subsequent periods at fair value. The remaining consideration amount received or allocated to the entire convertible instrument is allocated to the host debt
instrument. The difference between the face value of the host and such an allocated amount represents a discount which is amortized as finance expense to profit or loss using an effective interest method over the term of the note until its stated
maturity.
When it is determined that the embedded conversion feature qualifies for equity classification (such when the embedded conversion option, if it were freestanding, is not qualified as a derivative in
accordance with the provisions of ASC 815-10, “Derivatives and Hedging” since its terms did not require or permit net settlement or when the embedded conversion option is indexed to the entity’s own stock), the conversion option is not bifurcated.
When bifurcation is not required, the Company considers whether the debt instrument involves a significant premium (i.e. when the proceeds received or allocated upon issuance exceed the principal amount that will be paid at maturity). When it is
determined that a substantial premium exists, the entire premium is allocated to paid-in capital and when it is determined, otherwise no additional accounting is required and the convertible promissory note is accounted for at amortized cost using
effective interest method over the term of the note until its stated maturity.
SHARE-BASED
PAYMENT TRANSACTIONS
The Group's employees and service providers are entitled to remuneration in the form of equity-settled share-based payments.
Equity-settled transactions
The cost of equity-settled transactions with employees, officers and directors is measured at the fair value of the equity instruments granted at the grant date. The fair value is determined by using
an acceptable option pricing model.
The cost of equity-settled transactions with service providers is measured at the fair value of the goods or services received as consideration for equity instruments granted.
The cost of equity-settled transactions is recognized in profit or loss together with a corresponding increase in equity during the period in which the service conditions are to be satisfied ending
on the date on which the relevant employees become entitled to the award (the “Vesting Period”). The cumulative expense recognized for equity-settled transactions at the end of each reporting period until the vesting date reflects the extent to which
the Vesting Period has expired and the Group's best estimate of the number of equity instruments that will ultimately vest.
47
|
Management’s Discussion and Analysis
|
|
TREASURY
SHARES
The cost of the Common Shares held by the Company is deducted from the capital as a separate component under the "Treasury shares" section.
TRANSACTIONS
BETWEEN A COMPANY AND ITS CONTROLLING SHAREHOLDER
Assets and liabilities concerning transactions carried between the Company and its controlling shareholder or between companies under the same control are recognized at the time of the transaction at
fair value. The difference between the fair value and the consideration determined in the transaction is allocated to the capital minus the tax effect (to the extent relevant). The difference in debt is essentially a dividend, increasing the balance
of the accumulated deficit. The difference in the credit is essentially an owner's investment and is therefore credited in a separate section in the capital.
PROFIT
OR LOSS PER SHARE
Basic profit or loss per share is computed by dividing the profit or loss for the period applicable for common shareholders by the weighted average number of Common Shares outstanding, after
deduction of shares held by the Company, and Common Shares to be issued upon the vesting of Restricted Shares Units (RSUs). In computing diluted profit or loss per share, the basic profit or loss per share is adjusted to reflect the potential
dilution that could occur upon exercise of options and non-vested RSUs granted using the “treasury stock method” and using the if-converted method with respect to warrants or prefunded warrants granted or convertible debentures issued, if the effect
of each of such financial instruments is dilutive. In computing diluted profit or loss per share, the average share price for the period is used in determining the number of Common Shares assumed to be purchased from the proceeds to be received from
the exercise of options or warrants. The Company's share of profit or loss of investees is included based on its share of profit or loss per share of the investees multiplied by the number of shares held by the Company.
Shares to be issued upon exercise of options, non-vested RSUs, warrants and prefunded warrants and conversion of convertible debentures, have been excluded from the calculation of the diluted net
profit or loss per share for all the reported periods for which net profit or loss was reported because the effect of the Common Shares issuable as result of the exercise or conversion of these instruments was anti-dilutive.
NEW
STANDARDS, AMENDMENTS, AND INTERPRETATIONS TO EXISTING STANDARDS THAT ARE EFFECTIVE AND RELEVANT TO THE GROUP'S BUSINESS ACTIVITY
Amendments to IAS 21, "The Effects of Changes in Foreign Exchange Rates"
In August 2023, the IASB issued “Amendments to IAS 21: Lack of Exchangeability (Amendments to IAS 21, “The Effects of Changes in Foreign Exchange Rates”)” to clarify how an entity should assess whether a currency is
exchangeable and how it should measure and determine a spot exchange rate when exchangeability is lacking. The Amendments set out the requirements for determining the spot exchange rate when a currency lacks exchangeability. The Amendments require
disclosure of information that will enable users of financial statements to understand how a currency not being exchangeable affects or is expected to affect the entity's financial performance, financial position, and cash flows.
48
|
Management’s Discussion and Analysis
|
|
The Amendments apply for annual reporting periods beginning on or after January 1, 2025. Upon applying the Amendments, restatement of comparative information is prohibited.
The Company believes that the adoption of the Amendments has no material impact on its consolidated financial statements.
NEW STANDARDS IN THE PERIOD
PRIOR TO THEIR ADOPTION
IFRS 18, Presentation and Disclosure in Financial Statements
On April 9, 2024, the IASB published IFRS 18, which replaces IAS 1 ‘Presentation of Financial Statements’ with the objective to improve how information is communicated in an entity’s financial statements, particularly in
the statement of profit or loss and in its notes to the financial statements.
The main changes that will apply to the financial statements with the implementation of IFRS 18, in relation to the presentation and disclosure instructions that apply today include the following:
IFRS 18 changes the structure of the profit or loss report and includes three new defined categories: operating, investment and financing and adds two new interim summaries: operating profit and profit before financing
and income taxes.
IFRS 18 includes guidelines for providing disclosure on performance indicators defined by management (Management-defined performance measures).
IFRS 18 provides guidelines regarding the aggregation and disaggregation of the information in the financial statements in relation to the question of whether information should be included in the main reports or in
explanations and disclosures regarding items defined as "other".
IFRS 18 includes amendments to other standards, including limited amendments to International Accounting Standard 7, Statement of Cash Flows.
IFRS 18 will become effective, in a retrospective manner, for annual reporting periods beginning on or after January 1, 2027. Early application of IFRS 18 is permitted.
The Company is examining the possible impact of the new standard on the financial statements, but at this stage it is unable to assess such an impact. The effect of the new standard, however it may be, will only affect
matters of presentation and disclosure.
In accordance with National Instrument 52-109 – Certification of Disclosure in Issuers’ Annual and Interim Filings (“NI
52-109”) and Rule 13a-15 under the United States Securities Exchange Act of 1934, as amended (the “Exchange Act”), the establishment and maintenance of the Company’s disclosure controls and procedures
(“DC&P”) and internal control over financial reporting (“ICFR”) is the responsibility of management.
Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for
external purposes in accordance with applicable IFRS. Internal control over financial reporting should include those policies and procedures that establish the following:
| ● |
maintenance of records in reasonable detail, that accurately and fairly reflect the transactions and dispositions of assets.
|
49
|
Management’s Discussion and Analysis
|
|
| ● |
reasonable assurance that transactions are recorded as necessary to permit the preparation of financial statements in accordance with applicable IFRS.
|
| ● |
receipts and expenditures are only being made in accordance with authorizations of management or the Board; and
|
| ● |
reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial instruments.
|
NI 52-109 requires the CEO and CFO to certify that they are responsible for establishing and maintaining DC&P and ICFR for the Company and have concluded that as at December 31, 2025, those
internal controls have been designed and are effective in providing reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with IFRS.
The Company maintains a set of DC&P designed to provide reasonable assurance that information required to be publicly disclosed is recorded, processed, summarized, and reported on a timely basis.
As required by NI 52-109 and Exchange Act Rule 13a-15(b), an evaluation of the design and operation of our DC&P was completed as of December 31, 2024, under the supervision and with the participation of management, including our CEO and CFO using
the criteria set forth in the Internal Control-Integrated Framework (2013), issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based upon this evaluation, our CEO and CFO concluded that as of December 31, 2024, the
Company’s DC&P and ICFR were effective.
There have been no changes to the Company’s ICFR during the twelve months ended December 31, 2025, that have materially affected, or are likely to materially affect, the Company’s ICFR.
LIMITATIONS
OF DISCLOSURE CONTROLS AND PROCEDURES AND INTERNAL CONTROL OVER FINANCIAL REPORTING
The Company’s management, including the CEO and CFO, believes that due to inherent limitations, any DC&P or ICFR, no matter how well designed and operated, can provide only reasonable, not
absolute, assurance of achieving the desired control objectives. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Also,
projections of any evaluation of effectiveness to future periods are subject to the risk that any design will not succeed in achieving its stated goals under all potential future conditions. Accordingly, because of the inherent limitations in a
cost-effective control system, misstatements due to error or fraud may occur and not be detected. Additionally, management is required to use judgment in evaluating controls and procedures.
LIMITATION ON SCOPE OF
DESIGN
In accordance with Section 3.3 of National Instrument 52-109 – Certification of Disclosure in Issuers’ Annual and Interim Filings (“NI 52-109”), the Company
has limited the design of its DC&P and ICFR to exclude the controls, policies and procedures of Oranim Plus (the “Excluded Entity”), acquired by the Company or by one of its subsidiaries within 365 days of
the end of the period ended December 31, 2025.
As of December 31, 2025, the Company has implemented its DC&P AND ICFR in all its subsidiaries.
50
|
Management’s Discussion and Analysis
|
|
ISRAELI MARKET DEVELOPMENT 2013-2026
According to Israeli Ministry of Health, as of March 2026, there are 136,900 medical cannabis patients in Israel - 105,100 with licenses and 31,800 with prescriptions. A monthly prescription of
4,409,000 grams of medical cannabis were recorded in March 2026, an increase of 36,000 grams of cannabis from March 2025.
The chart below reflects the growth in licensed medical cannabis patients in Israel between July 2015 to March 2026.3
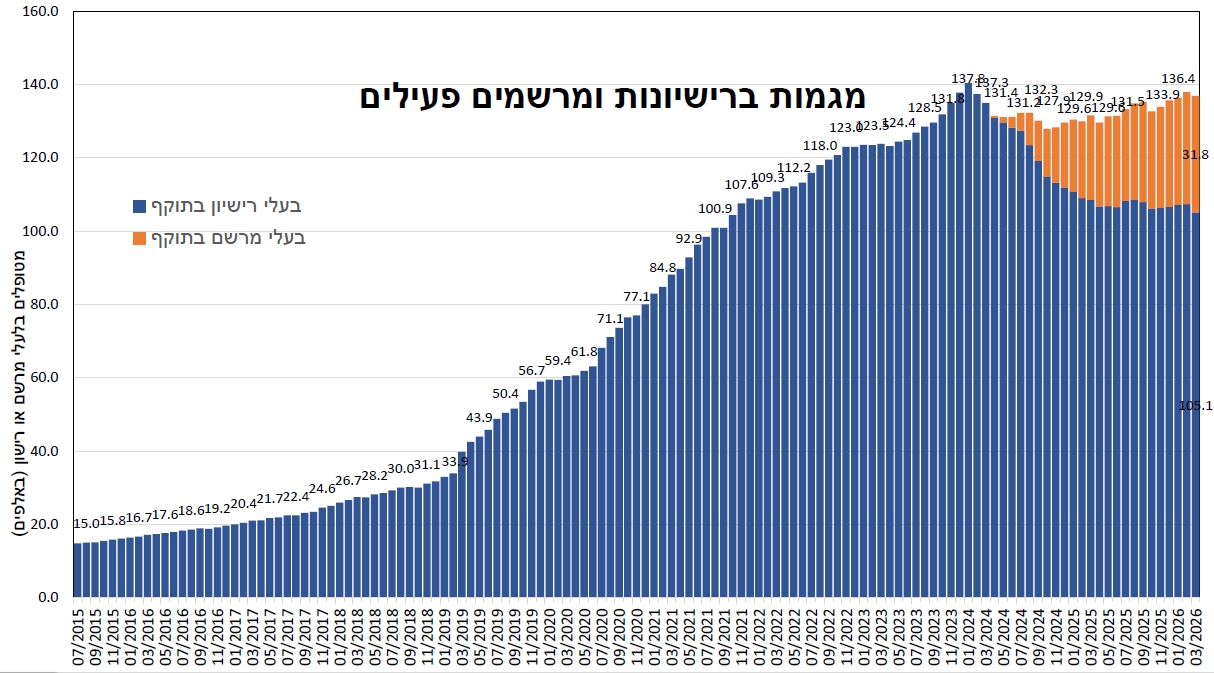
REGULATORY FRAMEWORK IN
ISRAEL
In Israel, cannabis is currently defined as a “dangerous drug” according to the Dangerous Drugs Ordinance4 (“DDO”) and the 1961 Single Convention on Narcotic Drugs (“Narcotics Convention”), to which Israel is a signatory. However, both the DDO
and the Narcotics Convention allow for the use of cannabis for medical or research purposes under a supervised and controlled regime. The competent regulatory authority in Israel in all matters concerning the oversight, control and regulation of
cannabis for medical production, consumption, and research in Israel is the IMCA, established by Government Res. No. 3069.5 The
production, distribution and consumption of adult-use recreational cannabis products is currently illegal in Israel.
3 Ministry of Health – licensed patients’ data as of March 2026 - https://www.gov.il/BlobFolder/reports/licenses-status-march-2026/he/subjects_cannabis_docs_licenses-status-march-2026.pdf
4 Cannabis is listed in schedule 1 of the Dangerous Drugs Ordinance [New
Version], 1973 [in English] https://www.health.gov.il/LegislationLibrary/Samim_01_EN.pdf
5 Israeli Government Res. No. 3609 [in Hebrew], August 7th, 2011 https://www.gov.il/he/departments/policies/2011_des3609
51
|
Management’s Discussion and Analysis
|
|
Patient Medical Consumption
The use of cannabis is allowed for patients and for medical purposes, in respect of certain medical conditions, under a special approval of the MOH. Procedure 1066 of the IMCA sets out a list of medical conditions that are allowed to be treated with medical cannabis products. Such authorized medical conditions are examined
and updated from time to time, and include, among others, cancer, pain, nausea, seizures, muscle spasms, epilepsy, Tourette syndrome, multiple sclerosis, amyotrophic lateral sclerosis, and post-traumatic stress disorder.
Licensing and Authorization for Commercial Activities in the Medical Cannabis Field
In December 2017, the IMCA issued regulations that standardized the licensing process for any cannabis related activity (the “Road Map”).7 Pursuant to the Road Map, each operation in the medical cannabis field, including the propagation, cultivation, products
manufacturing, storage and distribution to licensed pharmacies, and distribution from licensed pharmacies to licensed patients, requires compliance with the provisions of applicable laws, including the procurement of an appropriate license under the
DDO from the IMCA and the maintenance of such license in good standing. Cannabis licenses may not be transferred, exchanged or assigned without the prior approval of the IMCA. The licenses are valid for a period of up to 3 years and can be renewed
with the approval of the IMCA only.
The IMCA has issued a set of directives containing procedures and requirements for applicants for cannabis related activity licenses and has authorized certain entities to issue official certificates
upon compliance with such directives. These directives include (i) Directive 150 (GSP Standard certification); (ii) Directive 151 (GAP Standard certification); (iii) Directive 152 (GMP Standard certification); and (iv) Directive 153 (GDP Standard
certification). Regular and periodic examinations are conducted for licensed entities, in order to ensure compliance with the analytical standards and the level of quality required during each of the phases of production and distribution of medical
cannabis.
The IMCA has introduced reforms to streamline the licensing process for medical cannabis activities. These reforms aim to reduce bureaucratic hurdles and encourage
growth within the medical cannabis industry. For more information see "Regulatory Reform from Licenses to Prescriptions for Medical Treatment of Cannabis" below.
Medical Cannabis Imports and Exports
The Narcotics Convention governs the import and export of cannabis between member countries. Since Israel is a member country, any export and import of cannabis is subject to the Narcotic Convention.
In October 2020, the IMCA issued an updated procedure, titled “Guidelines for Approval of Applications for Importation of Dangerous Drug of Cannabis Type for Medical Use and for
Research” (“Procedure 109”), describing the application requirements for cannabis import licenses for medical and research purposes. Therefore, each import of medical cannabis is to be approved by the IMCA
issuing a specific import permit for each imported shipment, rather than a general license for import. An application for import of medical cannabis can be submitted by an entity licensed by the IMCA for the conduct of medical cannabis related
activity. The Israeli government approved the export of pharmaceutical-grade cannabis and cannabis-based products on January 27, 2019,8
and in December 2020, the IMCA published guidelines for the medical cannabis export permit application process.9.
6 Ministry of Health Pharmaceutical Division Policy Number 106 – Licenses for Use of Cannabis
https://www.health.gov.il/hozer/CN_106_2019.pdf (in Hebrew)
7 Directive 107 - Guidelines for the process of licensing the practice of cannabis for medical use,
as amended on October 2020 [Hebrew] - https://www.health.gov.il/hozer/CN_107_2019.pdf
8 Directive 4490 [Hebrew] - https://www.gov.il/he/departments/policies/dec4490_2019
9 Directive 110, December 2020 [Hebrew] - https://www.health.gov.il/hozer/CN_110.pdf
52
|
Management’s Discussion and Analysis
|
|
Legalization of Adult-Use Recreational Cannabis and CBD for Non-Medical Purposes in Israel
Currently, adult-use recreational cannabis use in Israel and CBD for non-medical use is illegal. In November 2020, an Israeli government committee responsible for advancing the
cannabis market reform published a report supporting and recommending the legalization of adult-use recreational cannabis in Israel. The Israeli parliament dissolved since then without applying the committee’s’ recommendations and all legislative
initiatives were suspended. However, the new government, formed on June 13, 2021, declared, and settled in the coalition agreement, its commitment to legalization of adult-use recreational cannabis. Since the formation of the new government, several
legislative initiatives were filed, including for the decriminalization of the possession of cannabis for individual recreational adult-use and the legalization of CBD for non-medical use. In February 2022, a Ministry of Health committee contemplated
the legality of CBD and published its recommendation that CBD should be excluded from the DDO. The main recommendations of the committee were adopted by the Minister of Health, however, to date, the Minister has not enacted an order directing that
CBD be removed from the DDO. On April 1, 2022, new regulations came into force which deemed the previously criminal offences of cannabis possession and use for self-consumption into administrative offences, which do not impact a criminal record, and
limited the penalty to a monetary fine only.
Regulatory Reform from Licenses to Prescriptions for Medical Treatment of Cannabis
In August 2022, the MOH published a draft outline of the transition reform from licenses to prescriptions for medical treatment of cannabis (the “Proposed Outline”). On June 13, 2023, the health committee of the Knesset approved The Dangerous Drugs Regulations (Amendment), 2023 (hereinafter referred to as the "Regulations Amendment""),
which entail a model change from issuing licenses to prescriptions permits following the publication of the Proposed Outline10.
The Regulations Amendment allows accessibility and significant bureaucratic relief for patients. The purpose of the new prescription model (as defined below) is to enable qualified specialist doctors (excluding general practitioner, family physician,
internal physician and pediatrician) to write prescriptions for medical cannabis for patients under the supervision of health care providers (widely known as Kupat Holim), without requiring a usage license from the Ministry of Health (hereinafter
referred to as "The New Prescription Model").
The main changes in the Regulations Amendment are: (i) any specialized doctor can issue permits without the need for specialized training; (ii) the permits for the use of
cannabis will be in the form of prescriptions, and not in the form of licenses from the MOH as the current framework requires; (iii) cannabis products can be sold in any pharmacy, and not only in pharmacies that have received a special permit from
the IMCA and a license from the MOH. The Regulations Amendment will come into effect within 180 days of their publication. To the best of the Company's knowledge, the indications approved as part of the Regulations Amendment encompass various
conditions, such as oncological diseases, active inflammatory bowel disease, AIDS, Multiple Sclerosis, Parkinson's disease, Tourette syndrome, epilepsy, autism, and dementia.
10 [Hebrew] - https://www.gov.il/he/Departments/policies/reform-of-drug-prescription
53
|
Management’s Discussion and Analysis
|
|
On December 8, 2023, the Company announced a 3-month delay of the anticipated medical cannabis reform announced by the Israeli ministry of health on August 7, 2023 (the "Reform"). Due to the Iron Swords War, the anticipated implementation of the medical cannabis regulatory reform, originally scheduled for December 29, 2023, has been postponed by three months. The new regulations
were designed to alleviate many of the stringent restrictions in the sector, thereby enhancing access to medical cannabis for patients.
On April 1, 2024, the Israeli cannabis reform was implemented.
“Anti-Dumping” investigation into cannabis imports from Canada
A notice on the Israeli Government’s website dated January 18, 2024, was addressed to 10 different Canadian cannabis producers: Village Farms International, Organigram Holdings, Tilray Canada, Hexo
Corp (owned by Tilray), The Green Organic Dutchman, Canopy Growth Corporation, SNDL Inc., Cronos Group, Auxly Cannabis Group, Decibel Cannabis, and all the medical cannabis manufacturers in Canada who export their goods to Israel.
The Commissioner for Trade Levies at the Ministry of Economy and Industry (the "MEI Commissioner"), announced by virtue
of his authority according to Section 24(d) of the Law on Trade Levies and Defence Measures, 5591 – 1991, of his decision to open an investigation on his own initiative into the export of cannabis from Canada, after he found that special
circumstances of actual damage exist or the probability of actual damage to the local manufacturing industry exist. The notice dated January 15, 2024 also included a letter sent to Michael Mancini, the Chief Commercial Counselor with the Embassy of
Canada, informing of the investigation. The Ministry of Economy and Industry issued a formal notice to the public to respond to questionnaires regarding the "Anti-Dumping" investigation.
Further to several requests received from the parties involved in the investigation and in accordance with section 27(b) of the Law on Trade Levies and Defense Measures, 1991 which states that "The
MEI Commissioner may, for special reasons that shall be recorded, extend the period specified in subsection (a) by an additional period that shall not exceed 30 days.", the MEI Commissioner decided that special conditions exist for extending the
deadline for the submission of the required materials as part of the investigation for 10 days until March 10, 2024, due to constraints presented by the parties following the Iron Swords war. The main reasons for the delays in the preparation of the
materials were due to the absence of many workers as part of the extensive recruitment in Israel for the reserve service and due to the unique complexity of the Israeli cannabis market where many players are required to submit data, both as producers
and importer. The Company has submitted the relevant questionnaires regarding its subsidiaries Focus and IMC Pharma, which are included in the investigation, as well as for its subsidiary Rosen Highway which is not included but is a significant
importer in Israel.
On June 18, 2024, the Ministry of Economy and Industry announced that it has decided to postpone the final deadline for obtaining its preliminary decision until July 18, 2024.
54
|
Management’s Discussion and Analysis
|
|
On July 10, 2024, the MEI Commissioner published a preliminary decision regarding the investigation and findings determining that there is dumping and consequent injury, on the basis of best
information available. The Company is evaluating the preliminary decision and its potential impact on the Company and its subsidiaries. Focus And IMC Pharma submitted their response on August 23, 2024, as required by the preliminary decision.
As part of the preliminary decision, the MEI Commissioner determined that a temporary guarantee is not necessary at this stage, and the Company is now awaiting the MEI Commissioner’s final decision.
This decision must be approved by the Ministry of Economy’s Director General, following consultation with the Ministry of Finance’s Budgets Director. The local growers have filed an administrative petition against the MEI Commissioner’s decision not
to impose a temporary guarantee. The company submitted a request to the court to join the petition to argue against the claims of the local growers and the request was approved by the court. A hearing on the petition has not yet been scheduled. The
Company will file its arguments to the court on November 21, 2024.
On November 10, 2024, the MEI Commissioner published the final report on the investigation into cannabis imports from Canada, recommending the imposition of tax levies.
According to the recommendations, a tax of 175% will be imposed on cannabis imports from Canadian companies that did not cooperate with the investigation, while major importers that participated will
be subject to lower tax rates, starting at 2% and increasing incrementally. The Company is currently reviewing these recommendations and considering steps to prevent or mitigate the final decision.
On November 24, 2024, the Company submitted its formal response to the advisory committee, which is responsible for developing and submitting recommendations to the Minister of Treasury prior to the
minister's final decision in this regard. It is currently uncertain when the advisory committee will conclude its deliberations or what outcome can be anticipated at this stage.
On December 11, 2024, the Advisory Committee held its first meeting where the Company participated and presented its arguments against the imposition of dumping tax. The Advisory Committee has not
yet sent its recommendations to the Minister of Treasury nor published any recommendations. Following the first meeting, the Company sent a letter to the Advisory Committee, presenting both new and existing arguments for consideration before the
Committee submits its recommendations to the Minister of Treasury.
On January 26, 2025, the Jerusalem District Court held a hearing on the administrative petition filed by the local growers seeking to impose a temporary guarantee. The judge recommended that the
local growers withdraw their petition. The local growers subsequently submitted their consent to withdraw the petition without costs being imposed. The parties had to submit their response regarding the request to waive costs by February 4, 2025.
On February 4, 2025, the Company submitted its response, stating that it is not waiving costs from the local growers. Following this, the Court decided to close the petition without costs.
On April 10, 2025, the Minister of Economy and Industry, Mr. Nir Barkat, decided to fully adopt the recommendations of the Public Advisory Committee and impose an anti-dumping tax of up to 165% on
imported medical cannabis flowers from Canada, for a period of four years. The tax is intended to assist Israeli growers against unfair competition from imported Canadian cannabis sold at dumped prices, which threatens the viability of the local
industry. The tax will take effect following the approval of the Minister of Treasury and the Finance Committee of the Knesset (the Israeli Parliament). Minister Barkat's decision followed a complex process, during which he was temporarily barred
from involvement due to a conflict of interest. Efforts to assign the matter to another minister were unsuccessful, and Barkat ultimately resumed responsibility and made the decision amid strong lobbying efforts both for and against the tax.
55
|
Management’s Discussion and Analysis
|
|
On April 24, 2025, the Minister of Treasury issued his final decision, rejecting the recommendation to impose anti-dumping levies on cannabis imports from Canada. The Minister's decision was based on
a comprehensive review of the investigation findings, the Commissioner’s recommendation, the advisory committee’s conclusions, the position of the Competition Authority, the Ministry of Health’s views, and additional public comments. After hearing
all relevant parties, including local growers and importers, and consulting with Ministry of Finance experts, the Minister concluded that imposing the levy would harm the macroeconomic development of the local cannabis market and the broader Israeli
economy. He emphasized that the cannabis sector is still developing, that competition has significantly improved product quality and reduced prices, and that maintaining competitive pressure from imports is essential for the industry’s healthy growth
and global competitiveness. Nonetheless, on April 29, 2025, the Minister of Economy and Industry issued his final decision, rejecting the objections raised against the imposition of the anti-dumping levy. In his statement, the Minister concluded that
the objections did not meet the legal standards set by applicable law and the Attorney General’s guidelines. He further stated that, in the absence of any substantive legal refusal submitted within the statutory timeframe, there was no basis to delay
or prevent the imposition of the levy. Accordingly, he decided to proceed with promoting the anti-dumping tax. However, pursuant to the Trade Levies and Defense Measures Law, the final authority on whether to impose such levies lies with the Minister
of Treasury. In light of this, and in response to the apparent contradiction between the decisions of the Minister of Economy and the Minister of Treasury, the Company has sent formal letters to the Attorney General of the Government and to the Legal
Advisor of the Knesset, requesting their determination that the Minister of Economy's decision to proceed despite the Minister of Treasury’s rejection is not in accordance with the law.
On June 12, 2025, an administrative petition (case number 31510-06-25) was filed with the Jerusalem District Court, requesting that the Minister of Economy impose a levy on imports of medical
cannabis imported from Canada and that the objection raised by the Minister of Finance be declared void.
On July 2, 2025, the Legal Department of the Israeli Ministry of Justice issued a detailed legal opinion in response to the Ministry of Economy’s request to overturn the Minister of Treasury’s
decision not to approve the anti-dumping levy. The Ministry of Justice concluded that although there were procedural concerns and conflicting positions among the various governmental bodies involved, there was no legal basis to invalidate the
Minister of Treasury’s decision. The opinion confirmed that the Minister of Treasury acted within the scope of his legal authority under the Trade Levies and Defense Measures Law, basing his decision on recognized macroeconomic, fiscal, and policy
considerations. While acknowledging that some of the factual findings could be debated, the opinion emphasized the need for legal restraint and ultimately upheld the Minister of Treasury’s veto as lawful.
On July 8, 2025, the Company (through its subsidiaries IMC Pharma and Focus Medical Herbs), as named respondents, submitted a preliminary response in accordance with the Court’s instructions.
The response asserts that the petition should be dismissed due to substantial delay, lack of clean hands by the petitioners, failure to include relevant parties (such as the Ministry of Health and
the Competition Authority), and the absence of legal grounds to challenge the Finance Minister’s authority. It further relies on the legal opinion issued by Legal Department of the Israeli Ministry of Justice confirming that the Finance Minister’s
decision to oppose the imposition of the levy was lawful, reasoned, and aligned with broader macroeconomic considerations, including patient access, market balance, and consistency with longstanding government policy supporting cannabis imports. The
response also emphasized that, under applicable law, the Minister of Economy cannot impose such levies without the approval of the Finance Minister, which was explicitly denied.
56
|
Management’s Discussion and Analysis
|
|
The Company believes the petition is without merit and intends to defend its position vigorously.
The matter remains in legal dispute, with the Company opposing any reversal of the Finance Minister's authority.
REGULATORY FRAMEWORK IN
GERMANY
On March 10, 2017, the German federal government enacted bill Bundestag- Drucksache 18/8965 – Law amending narcotics and other regulations that amended existing narcotics legislation to recognize
cannabis as a form of medicine and allow for the importation and domestic cultivation of medical cannabis products. Under the subsequent updated legislation, cannabis was listed in Annex 3 to the Federal Narcotics Act (“BtMG”) as a “marketable narcotic suitable for prescription” until the Medical Cannabis Act (“MedCanG”) came into force on April 1, 2024. Since then, medical cannabis has no longer been subject
to the BtMG but to the MedCanG. With regard to medical cannabis, the cultivation, production, distribution, exportation, transit and importation are currently legal in Germany, subject to regulations and licensing requirements. At the same time as
the MedCanG, the Consumer Cannabis Act (“KCanG”) also came into force on April 1, 2024. Operations involving adult-use recreational cannabis products became legal under certain conditions defined in the KCanG.
Until the KCanG came into force, legalization in Germany applied only to cannabis for medicinal purposes under state control in accordance with the Narcotic Convention. This development has its origins in the fact that the former German government
has declared in the coalition agreement at the end of 2021 its intention to open the German market also in the adult-use recreational market. In October 2022, a key points paper on the controlled supply of cannabis to adults for consumption purposes,
although a restructuring of the existing regulatory framework on cannabis in general was also discussed, published by the former cabinet, which was submitted to the European Union Commission for a preliminary legal examination. In this respect, the
Federal Government issued a declaration of interpretation with regard to existing international agreements governing the adult-use recreational cannabis usage and submitted a draft law to the European Union Commission within the framework of a
notification.
After a long political debate, the German Bundestag approved the federal government's draft law "on the controlled use of cannabis" (BT Drs. 20/8704, BT Drs. 20/8763, BT-Drs. 20/10426) on Friday,
February 23, 2024. The draft law (BT Drs. 20/8704) then came into force on April 1, 2024. An adjustment has already been made by Article 1 of the Act of June 20, 2024 (BGBl. 2024 I No. 207). Some components of the KCanG, which deal with so-called
consumer cannabis, came into force on 1 July 2024 (such as the possibility to apply for a permission to grow by and distribute recreational cannabis to members of a cultivation association. The entry into force of the law also had direct consequences
for medical cannabis, which is the subject matter of Art. 2 (Medical Cannabis Act - MedCanG) and 3 (BtMG) of the law. Since the KCanG and MedCanG came into force, cannabis is no longer a narcotic by definition and is therefore no longer subject to
the BtMG. The definition in Annex 3 of the BtMG was be replaced by that in Section 2 MedCanG: "Cannabis for medical purposes: plants, flowers and other parts of plants belonging to the genus Cannabis that are grown for medical purposes under state
control in accordance with Articles 23 and 28(1) of the Single Convention on Narcotic Drugs of 1961 of March 30, 1961 (Federal Law Gazette 1973 II p. 1354), as well as delta-9-tetrahydrocannabinol including dronabinol and preparations of all the
aforementioned substances". However, the narcotics regulations were replaced by comparable regulations and authorisations. The Federal Institute for Drugs and Medical Devices (BfArM) remains responsible for the latter as a higher federal authority.
From a regulatory perspective, medical cannabis remains a medicinal product or an active pharmaceutical ingredient, meaning that the requirements under medicinal product law will remain in place. As a result, the marketing of irradiated products
continues to require a marketing authorisation in accordance with the Ordinance on Medicinal Products Treated with Radioactive or Ionising Radiation (AMRadV). Only the narcotics licence pursuant to Section 3 BtMG is replaced by a new licence pursuant
to the MedCanG (see Section 1), which, however, largely corresponds to the previous provisions of the BtMG regarding the application process and general regulations. However, there are the following differences that are new since the entry into
force: Medical cannabis no longer has to be stored and transported like a narcotic. The corresponding safety precautions no longer apply, meaning that compliance with the provisions of pharmaceutical law is sufficient. The so-called semi-annual
reports are replaced by annual reports. The requirements for the person responsible for medical cannabis are slightly reduced compared to those for narcotics. A prescription of medical cannabis is possible without the need to use the form for
prescription for narcotics. A normal prescription is sufficient.
57
|
Management’s Discussion and Analysis
|
|
However, it is likely to be of great importance that the cultivation of medicinal cannabis based on Section 17 MedCanG is no longer subject to public tenders, but like the trading licence, it is
ultimately subject to a two-stage authorisation (at state level regarding the pharmaceutical regulations and at federal level with regard to the fact that it is medicinal cannabis).
Medical cannabis in Germany must comply with the corresponding monographs of the German and European Pharmacopoeia (“Ph. Eur.”). The Ph. Eur. Suppl. 11.5
contains the new Ph. Eur. Monograph on cannabis flowers and the new Ph. Eur. Monograph on Cannabidiol (“CBD”). With the entry into force of Suppl. 11.5 of the national edition of the Ph. Eur. on April 1, 2025,
the monograph 'Cannabis flowers / Cannabis flos [3028]' contained therein became legally binding from this date. The previous monograph 'Cannabis flowers' of the German Pharmacopoeia was deleted with the last edition of the DAB. The new monograph on
cannabis flowers includes Starting materials for the production of extracts, medicinal products that can be prescribed as such (herbal medicinal products) that are taken by patients by inhalation or oral administration. There are not entirely
irrelevant changes compared to the German monograph. The Committee on Herbal Medicinal Products (“HMPC”) as the European Medicines Agency’s (EMA) committee responsible for compiling and assessing scientific
data on herbal substances, preparations and combinations, announced that in view of uniform EU quality requirements (including with respect to import and export of cannabis), further Ph. Eur. Cannabis monographs are in preparation.
All BtMG permit applications had to specify the strains and estimated quantities of medical cannabis involved and any subsequent changes had to be reported to the Federal Opium Agency of Germany,
whereby varieties and quantities no longer appear on the permit, even if these are to be specified in the application. The same applies regarding Sections 7, 8 MedCanG in relation to the authorisation to trade in medical cannabis, although it is now
apparent that no expected annual quantities are to be specified. However, it can be assumed that the BfArM nevertheless enquire about these due to the (albeit somewhat reduced compared to the BtMG) reporting obligations in Sections 16 and 17 MedCanG
and the Foreign Narcotics Trade Regulation, which remains applicable (see Section 14 MedCanG).
CBD is neither a real subject to the KCanG nor to the MedCanG. Only in Section 1 No. 3 KCanG is there a definition, in Section 1 No. 8 b) KCanG the exemption of CBD from the term cannabis and in
Section 2 para. 2 No. 1 KCanG the exemption from the prohibition of extraction of the cannabis plant, which permits the extraction of CBD, even if it does not contain any further regulations on CBD in isolation. With regard to synthetic CBD, a
different set of regulations is important: the handling of cannabimimetics/synthetic cannabinoids is prohibited in accordance with Section 2 of the Annex in conjunction with Section 3 of the New Psychoactive Substances Act (NpSG). Product-specific
regulations relating to CBD can be found in other regulations. Thus, Annex 1 of the Ordinance on the Prescription of Medicinal Products stipulates that CBD is in principle subject to prescription but does not specify a minimum quantity or a specific
dosage form. If we look at the food sector, a distinction is made between products that naturally contain CBD and those that consist of or contain extracted CBD; the European Commission considers the latter to be novel foods under Regulation (EU)
2015/2283, which require authorisation before being placed on the market. Although applications for such authorisation have been submitted, the European Commission believes that they contain at least insufficient data on safety in food use, meaning
that none of the applications can currently lead to authorisation. Against this background, various products containing CBD can be found on the German market. There are currently various court decisions that problematize CBD in foods (especially food
supplements) and in cosmetics (especially mouth oil). On the one hand, CBD is regarded as a potential active pharmaceutical ingredient (API) to be used in medicinal products or as a novel food subject to authorisation and therefore unsuitable for a
safe use in a foodstuff, and on the other hand as unsuitable for cosmetic use in the mouth, as CBD would ultimately be consumed in this case (like a foodstuff and therefore to be regarded as foodstuff). CBD is therefore sometimes declared as a
flavoring in foods, as flavorings are excluded from the scope of the Novel Food Regulation. Nevertheless, there are already decisions that exclude suitability as a flavoring in individual cases.
58
|
Management’s Discussion and Analysis
|
|
Cultivation in Germany and Distribution of Medical Cannabis Cultivated in Germany
In the past, the Federal Opium Agency of Germany’s BfArM formed a cannabis division (the “Cannabis Agency”) to oversee cultivation, harvesting, processing, quality control, storage, packaging and
distribution to wholesalers, pharmacists and manufacturers. The Cannabis Agency also regulated pricing of German-produced medical cannabis products and served as an intermediary of medical cannabis product sales between manufacturers, wholesalers and
pharmacies on a non-profit basis so far. In late 2018, the Cannabis Agency issued a call for tenders to award licenses for local medical cannabis cultivation and distribution of German-cultivated medical cannabis products (the “German Local Tender”).
The Cannabis Agency served as an intermediary in the supply chain between such cultivation and distribution. In April 2019, three licenses for local cultivation were granted. In consequence three companies in Germany received the permission to
cultivate on behalf of the Cannabis Agency of the BfArM.
Since the entry into force of the MedCanG, the granting of licences for domestic cultivation is no longer subject to tendering but governed by §§ 4 et seq. MedCanG. The previously time-consuming
tendering and awarding of contracts for the domestic cultivation of cannabis for medical purposes by the Cannabis Agency and the subsequent purchase and distribution of the domestic harvest yields by the Cannabis Agency from the economic operators
determined during the tendering procedure are no longer necessary. Ultimately, only the corresponding licences in accordance with the MedCanG and - from implementation of manufacturing steps relevant under pharmaceutical law - the AMG are required in
compliance with the respective conditions and the associated regulations.
Import volumes and procedures regarding Medical Cannabis
The past and present regime permits the importation of cannabis plants and plant parts for medicinal purposes under state control subject to the requirements under the Narcotic Convention, according
to which, Germany must estimate the expected demand of medical cannabis products for medical and research purposes for the following year and report such estimates to the International Narcotics Control Board. In September 2025, Germany already
reached the maximum import quota of 122 tonnes of medical cannabis approved by the International Narcotics Control Board. The BfArM was therefore temporarily suspending new import licences until the quota was increased.
As a prerequisite to obtaining a German import license, the supplier must grow and harvest in compliance with EU-GACP-Guidelines and manufacture in compliance with EU-GMP-Guidelines and
certifications, or alternatively, it is a pure EU-GACP product, and the EU-GMP manufacturing steps then take place in Germany. With regard to imports from third countries and the associated testing and assessment of EU GMP compliance, the relevant
pharmaceutical regulations are in force, which also provide for on-site inspections by the EU authorities, provided that no MRA or similar is in force for the specific product type. All medical cannabis products imported to Germany must derive from
plant material cultivated in a country whose regulations comply with Art. 23 and 38 of the Narcotic Single Convention and must comply with the relevant and applicable monographs described in the German and European pharmacopeias.
59
|
Management’s Discussion and Analysis
|
|
Dispensing Exclusively via Pharmacies
Medical cannabis products imported pursuant to an import license under the MedCanG and AMG permits are sold exclusively to pharmacies for final dispensing to patients on a prescription basis as
‘magistral preparations’, a term used in Europe to refer to medical products prepared in a pharmacy in accordance with a medical prescription for an individual patient. Magistral preparations require certain manufacturing steps in the pharmacy. Such
manufacturing steps of the pharmacist typically include the testing and dosing of pre-packaged cannabis inflorescences (typically referred to as “flos”), medical cannabis products for oral administration (dronabinol), medical cannabis products for
inhalation upon evaporation and medical cannabis-infused teas. In addition to magistral preparations, medical cannabis products are also marketable as pre-packaged, licensed drugs (e.g. Sativex®).
Telemedicine and Mail-order-Pharmacies
According to the Federal Ministry of Health (“BMG”), the aforementioned increase in import volumes is due to a rise in the number of prescriptions for self-payers, which are not
covered by statutory health insurance. Only a very slight increase has been observed in statutory health insurance funds, which does not reflect the increase in imports to this extent.
In order to prevent these developments in prescribing practice, the BMG drafted a bill to amend the MedCanG (RefE)1 in July 2025. Such legislation amendment was then approved by the German Federal
Cabinet on October 8, 20252 and provides for a significant tightening of the MedCanG with regard to the prescription and dispensing of medical cannabis. In the draft, the BMG justifies this necessity with the need to ensure a secure supply of
medicines and patient safety. On the one hand, the BMG expresses concerns about the growing use of telemedicine platforms, since it believes that prescriptions for medical cannabis are often issued there without personal contact. The BMG assumes that
prescriptions are regularly issued solely on the basis of a questionnaire and sent via (cooperating) mail-order pharmacies. In the view of the ministry, this could cause risks as cannabis flowers for medical purposes are prescribed as a magistral
formulation and in consequence without a marketing authorisation. For this reason, according to RefE, Section 3 Med-CanG shall stipulate that the prescription of cannabis flowers would require in-person physician consultation (e.g. in a doctor's
office or during a home visit). This shall apply to both: initial prescriptions and repeat prescriptions, for which personal contact between doctor and patient must have taken place within the last four quarters. Furthermore, the distribution of
cannabis flowers for medical purposes within Germany by mail-order would be prohibited, while delivery only would be permitted. If implemented, these changes could materially adversely affect our German operations, including by limiting distribution
channels and reducing patient accessibility, which may, in return, negatively impact revenues and operating results.
For clarification: cross-border dispatch has always been prohibited under the World Postal Convention. The change would only affect dispatch within Germany. Pharmacies would only be able to deliver
using their own employees as couriers.
However, it should be noted that this drastic complication of prescribing could have significant consequences for certain patient groups. The purpose of telemedicine is precisely to further develop
healthcare by providing treatment options, especially for patients in rural areas. The BMG describes precisely these points on its website3. For this reason, there are also dissenting voices with regard to the RefE. It therefore remains to be seen
whether and to what extent the legislative changes envisaged in the RefE will ultimately be implemented in the MedCanG. This will be decided in the further course of the proceedings.
60
|
Management’s Discussion and Analysis
|
|
If the legislative changes are actually adopted as decided by the Federal Cabinet, this would affect not only the patients mentioned above, but also providers of telemedicine services and mail-order
pharmacies in particular.
NO U.S. CANNABIS-RELATED
ACTIVITIES
The Group does not engage in any U.S. cannabis-related activities as defined in Canadian Securities Administrators Staff Notice 51-352 (Revised) – Issuers with U.S.
Marijuana-Related Activities.
The Company has implemented risk management governance processes that are led by the Board, with the active participation of management, and updates its assessment of its business risks on an annual
basis. Notwithstanding, it is possible that the Company may not be able to foresee all the risks that it may have to face. The market in which IM Cannabis currently competes is complex, competitive and changing rapidly, and its business is subject to
risks inherent in a high growth, heavily regulated enterprise, and the Company has identified certain risks pertinent to the Group’s business that may have affected or may affect the Group’s business, financial conditions, results of operations and
cash flows, as further described throughout this MD&A and under “Risk Factors” in the Company’s Annual Report on Form 20-F for the fiscal year ended December 31, 2025 available on the Company’s profile on SEDAR+ at www.sedarplus.ca and on
EDGAR at www.sec.gov/edgar . For additional risk factors, readers are directed to the Annual Report. Sometimes new risks emerge, and management may not be able to predict all of them or be able to predict how they may cause actual results to
be different from those contained in forward looking statements. Readers of this MD&A should not rely upon forward looking statements as a prediction of future results.
CREDIT RISK
The maximum credit exposure as of March 31, 2026, is the carrying amount of cash and cash equivalents, trade receivables and other current assets. The Group does not have
significant credit risk with respect to outstanding trade receivables. All cash and cash equivalents are placed with major Israeli financial institutions.
Loan receivable credit risk is managed by each loan separately according to the Company’s policy, procedures and control relating to the borrower’s credit risk management. At the
end of each period, the individual loan values are assessed based on a credit risk analysis.
The expected credit loss analysis is generally based on management’s understanding of the borrower’s experience/integrity, financial health, business plans, capacity, products,
customers, contracts, competitive advantages/disadvantages, and other pertinent factors when assessing credit risk. This would also include the assessment of the borrower’s forecasts as well as taking into consideration any security and/or collateral
the Company has on the outstanding balance.
LIQUIDITY RISK
As of March 31, 2026, the Group's financial liabilities with liquidity risk consist of trade payables and other accounts payable which have contractual maturity dates within one year, bank loans and,
checks receivables and lease liabilities. The Group manages its liquidity risk by reviewing its capital requirements on an ongoing basis. Based on the Group's working capital position at March 31, 2026, management considers liquidity risk to be high.
61
|
Management’s Discussion and Analysis
|
|
CURRENCY RATE RISK NTD:
As of March 31, 2026, a portion of the Company's financial assets and liabilities are held in Euro and NIS consisting of cash in the amount of EUR 460 thousand (approximately
$737) and NIS 459 thousand (approximately $202), respectively. The Company's objective in managing its foreign currency risk is to minimize its net exposure to foreign currency cash flows by transacting with third parties in NIS to the greatest
extent possible. The Company does not currently use foreign exchange contracts to hedge its exposure of its foreign currency cash flows as management has determined that this risk is not significant at this point of time.
SHARE PRICE RISK
The Group's investments in unlisted shares are sensitive to market price risk arising from uncertainties about future value of these investments. The Group manages the price risk through
diversification and by placing limits on individual and total investment in shares. The Company's Board of directors reviews and approves all decisions related to investments in shares. At the reporting date, the Group's exposure to investments in
unlisted shares measured at fair value was $1,819.
TAX
REMITTANCE
The Company is subject to the provisions of the ITA12 and to review by CRA13. The Company files its annual tax compliance based on its interpretation of the ITA and CRA’s guidance. There is no
certainty that the returns and tax position of the Company will be accepted by CRA as filed. Any difference between the Company’s tax filings and CRA’s final assessment could impact the Company’s results and financial position.
There can be no assurance that income tax laws or the interpretation thereof in any of the jurisdictions in which the Company operates will not be changed or interpreted or administered in a manner
which adversely affects the Company and its shareholders. In addition, there is no assurance that CRA will agree with the manner in which the Company calculates taxes payable or that any of the other tax agencies will not change their administrative
practices to the detriment of the Company or its shareholders.
By Notice of Assessment for Excise Tax dated October 23, 2023 and covering the period January 1, 2020 to December 31, 2020, the Company was assessed tax on insurance of approximately $199, arrears
interest of approximately $36 and a failure to file penalty of approximately $8 (collectively, the “2020 Assessment”).
By Notice of Assessment for Excise Tax dated October 23, 2023 and covering the period January 1, 2021 to December 31, 2021, the Company was assessed excise tax on insurance of approximately $73,
arrears interest of approximately $2 and a failure to file penalty of approximately $1 (collectively, the “2021 Assessment”).
If a person files a Notice of Objection (Excise Tax Act), the CRA cannot take collection action on amounts in dispute until 90 days after the Notice of Decision is sent to that person. However,
interest and penalty continue to accrue on any amount owing.
On November 29, 2023, the Company filed Notices of Objection (Excise Tax Act) to the 2020 Assessment and the 2021 Assessment. Therefore, the CRA cannot take collection action on the amounts noted
above until 90 days after Notices of Decision are sent to the Company.
On April 26, 2024, the Company received a letter from the CRA that the Notice of Assessment for Excise Tax that the Company objected to will be voided and no outstanding balance will be owed with
respect to such assessments. Based on the forgoing, this matter has been resolved to the Company's satisfaction and the objections were finalized.
62
|
Management’s Discussion and Analysis
|
|
CYBERSECURITY
RISKS
The Company’s information systems and its third-party service providers and vendors are vulnerable to increasing threat of continually evolving cybersecurity risks, resulting in data breaches and
data losses. These risks arising from events including without limitation malware, computer viruses, employee error, extortion, malfeasance, system errors, and hacking. In order to minimize the risk of these events from occurring, the Group is
performing timely maintenance, upgrade and replacement of networks, equipment, IT systems and software and other protective measures. However, any failure or delay in maintaining, upgrading or replacing such systems and software could materially
increase the risk of cybersecurity incident and data breach or data loss, and the Company may experience operational delays, information system failures, and/or increases in capital expenses. Ultimately, the Company’s business, financial condition,
operating results and reputation may be impacted adversely by such occurrences.
The Company’s risk and exposure to these matters cannot be fully mitigated because of, among other things, the evolving nature of these threats. As a result, cyber security and the continued
development and enhancement of controls, processes and practices designed to protect systems, computers, software, data and networks from attack, damage or unauthorized access is a priority. As cyber threats continue to evolve, the Company may be
required to expend additional resources to modify or enhance protective measures or to investigate and remediate any security vulnerabilities.
CONSOLIDATION
OF CERTAIN FINANCIAL RESULTS UNDER IFRS 10 AND MAINTENANCE OF COMMON CONTROL
The Company complies with IFRS 10, which applies a single consolidation model using a definition of “control” that requires an investor (as defined in IFRS 10) to consolidate an investee (as defined
in IFRS 10) where: (i) the investor has power over the investee; (ii) the investor has exposure or rights to variable returns from involvement with the investee; and (iii) the investor can use its power over the investee to affect the amount of the
investor’s returns.
Subsequent to the restructuring of IMC Holdings on April 2, 2019, the Company analyzed the terms of the contractual agreements with Focus in accordance with IFRS 10 to conclude whether it should
continue to consolidate the accounts of Focus in its financial statements.
Under IFRS 10, consolidation occurs when an investor can exercise control over an investee. Control is achieved through voting rights or other evidence of power. Where there are no direct holdings,
under IFRS 10, an investor (as defined in IFRS 10) should consider other evidence of power and ability to unilaterally direct an investee’s (as defined in IFRS 10) relevant activities. In view of the contractual agreements and the guidance in IFRS
10, notwithstanding that the Company has no direct or indirect ownership of Focus Medical, it has sufficient rights to unilaterally direct the relevant activities (a concept known as “de facto control”), mainly due to the following:
| (a) |
the Company receiving economic benefits from Focus (and the terms of the contractual agreements between the Company and Focus cannot be changed without the approval of IMC Holdings);
|
| (b) |
IMC Holdings holds 74% interest in Focus;
|
| (c) |
Messrs. Shuster and Gabay each being a director of Focus (while Mr. Shuster concurrently being a CEO, director, and substantial shareholder of the Company and Mr. Gabay concurrently being a substantial shareholder of the Company); and
|
| (d) |
the Company providing management and support activities to Focus through a services agreement.
|
63
|
Management’s Discussion and Analysis
|
|
Accordingly, under IFRS 10, the Company has “de facto control” over Focus, and therefore consolidates the financial results of Focus in the Company’s financial statements.
Any failure of the Company or Messrs. Oren Shuster and Rafael Gabay to maintain “de facto control” over Focus as defined under IFRS 10 could alter the Company’s consolidation model, potentially
resulting in a material adverse effect on the business, results of operations and financial condition of the Company.
On November 30, 2023, IMC Holdings acted to exercise its option to purchase the divested 74% interest in Focus held by Oren Shuster, and Rafael Gabay by submitting a request to the "IMCA," an agency
operated by the Israeli Ministry of Health that will allow the option exercise.
On February 26, 2024, IMCA approved the persons who will be acting on behalf on IMC Holding pursuant to the exercise of the option, allowing to complete the transaction.
On February 26, 2024, IMC Holdings has exercised its option and as of that date, IMC holds 74% in Focus. The Company will continue to consolidate the financial results of Focus in the Company’s
financial statements.
The Company, through I.M.C. Holdings acquired from Ewave the remaining 26% interest in Focus following the approval of the Focus Transaction at the shareholder's Meeting held in May 23, 2025. The
Focus Transaction was closed effective May 26, 2025, following receipt of disinterested shareholder approval at the Meeting.
POSSIBLE
DIRECT INVOLVEMENT IN THE ISRAELI CANNABIS INDUSTRY
According to current Israeli regulatory medical cannabis framework, any engagement in Cannabis Activities requires receiving the applicable license from the “IMCA”, an agency operated by the Israeli
Ministry of Health, which requires, among other things, pre-approvals by the IMCA (the “IMCA Pre-Approval Requirement”) of the directors, officers and shareholders holding 5% or more of the shares of the
license applicant (“Material Holders”), and of all directors, officers and shareholders that become Material Holders following the grant of the applicable license. Therefore, if the Company will be considered
by the IMCA as directly engaged in Cannabis Activities the aforementioned approvals by the IMCA might apply, on future security holdings, as described above.
Furthermore, any failure of the Company or its shareholders to comply with the IMCA Pre-Approval Requirement may impact the Group’s ability to continue operating in compliance with any licenses to
engage in Cannabis Activities or to renew such licenses. Any inability of the Group to maintain licenses for Cannabis Activities in good standing may result in a material adverse effect on the Group’s business, financial condition, results of
operations and prospects.
COMPANY’S ABILITY TO
CONTINUE AS A GOING CONCERN
The Group’s current operating budget includes various assumptions concerning the level and timing of cash receipts from sales and cash outlays for operating expenses and capital expenditures,
including cost saving plans. In 2025 the company will continue its efforts for efficiency operations.
Despite the cost savings plan as described above, the projected cash flows for 2025 indicates that it is uncertain that the Group will generate sufficient funds to continue its operations and meet
its obligations as they become due. The Group continues to evaluate additional sources of capital and financing. However, there is no assurance that additional capital and or financing will be available to the Group, and even if available, whether it
will be on terms acceptable to the Group or in amounts required.
These conditions raise substantial doubt about the Company’s ability to continue as a going concern. The consolidated financial statements do not include any adjustments relating to the
recoverability and classification of assets or liabilities that might be necessary should the Company be unable to continue as a going concern.
64
|
Management’s Discussion and Analysis
|
|
CONFLICT AND POLITICAL
INSTABILITY IN EASTERN EUROPE
Throughout 2024 and continuing into Q3 2025, global financial markets have experienced elevated levels of volatility driven by persistent uncertainty around the timing, scale, and impact of monetary
policy responses to inflationary pressures, as well as ongoing geopolitical instability stemming from Russia’s invasion of Ukraine. This volatility has resulted in a decline in the level of activity in the financial markets. Continued market
volatility or uncertainty related to actions taken or to be taken by central banks, a decline in the global macroeconomic outlook, including as a result of Russia’s invasion of Ukraine and the threat, or outbreak of more widespread armed conflict in
Eastern Europe would cause financial market activity to continue to decrease, which would negatively affect the Group’s revenues and capital markets activity.
CONFLICT AND POLITICAL
INSTABILITY IN ISRAEL - THE IRON SWORDS WAR
The Group is vulnerable to the political, economic, legal, regulatory, and military conditions affecting Israel and the Middle East. Armed conflicts between Israel and its neighbouring countries and
territories occur periodically in the region and may adversely affect the Group’s business, results of operations and financial condition. In addition, the Group may be adversely affected by other events or factors affecting Israel such as the
interruption or curtailment of trade between Israel and its trading partners, or any restrictions or pressure on the Group’s partners or customers or others to prevent or discourage them from doing business activities with Israel or Israeli
businesses, a significant downturn in the economic or financial condition of Israel, a significant downgrading of Israel’s internal credit rating, labour disputes and political instability, including riots, uprisings and government failures.
Restrictive laws or policies directed towards Israel or Israeli businesses could have a material adverse effect on the Group’s business, results of operations, financial condition and prospects.
Any armed conflicts, terrorist activities or political instability in the region could adversely affect business conditions, could harm the Group’s results of operations, and could make it more
difficult for the Company to raise capital. Parties with whom the Group does business may decline to travel to Israel during periods of heightened unrest or tension, forcing the Group to make alternative arrangements when necessary in order to meet
our business partners face to face. In addition, the political and security situation in Israel may result in parties with whom we have agreements involving performance in Israel claiming that they are not obligated to perform their commitments under
those agreements pursuant to force majeure provisions in such agreements. Further, in the past, the State of Israel and Israeli companies have been subjected to economic boycotts. Several countries still restrict business with the State of Israel and
with Israeli companies. These restrictive laws and policies may have an adverse impact on our operating results, financial condition or the expansion of our business.
Furthermore, under Israeli law, citizens and permanent residents of Israel are obligated to perform military reserve duty for extended periods of time and are subject to being called to active duty
at any time under emergency circumstances. In response to increased hostilities, there have been periods of significant call-ups of military reservists.
On October 7, 2023, a war between the terror organization Hamas and Israel began, or the Iron Swords War, and has led to heightened security concerns and economic instability in Israel. There remains
significant uncertainty regarding the duration and escalation of the conflict, and further military actions, restrictions, or government-imposed measures could adversely affect our operations and financial condition.
The Iron Swords War has had an impact on the Company's business operations, which may or may not continue in the short and long term. The Company has experienced interruptions affecting various
aspects of its business, including related to employees, supplies, imports and sales. In addition, as a result of the Iron Swords War, legislative measures in Israel related to medical cannabis reform, which was initially set to commence on December
29, 2023, were postponed. While the reform eventually came into effect on April 1st, 2024, the healthcare system in Israel has experienced significant bureaucratic
challenges, including delays in renewing existing prescriptions and in issuing new prescriptions for medical cannabis patients. These administrative disruptions have directly impacted patient access to products and, consequently, affected the
Company’s local sales and distribution activities
65
|
Management’s Discussion and Analysis
|
|
Other regional hostilities, since October 7, 2023, have concurrently become more pronounced. This includes and has included a northern front war between Israel and Hezbollah and continued conflict
with the Houthi Movement in Yemen. Such potential disruption to our operations may include certain delays and diversions of the import of certain components for manufacturing and production as a result of reduced air travel and the attacks on
container ships on the Red Sea route by the Iranian-backed Houthi Movement.
In April 2024 and October 2024, Iran launched direct attacks on Israel involving hundreds of drones and missiles and has threatened to continue to attack Israel. On June 13, 2025, in light of
continued nuclear threats and intelligence assessments indicating imminent attacks, Israel launched a strike directly targeting military and nuclear infrastructure inside Iran aimed to disrupt Iran’s capacity to coordinate or launch further
hostilities against Israel, as well as disrupt its nuclear program. On June 13, 2025, a full-scale war has erupted between Israel and Iran, or the Iran War. On June 21, 2025, the United States military conducted targeted air strikes against three
nuclear sites within Iran and on June 23, 2025 Iran retaliated against U.S. interests in the region. Hostilities between Iran and Israel and the United States may further escalate, which could create significant volatility in the global economy as
well as disruptions to global supply chains. Additionally, in light of the national emergency declared by the Israeli government, the Company operated according to the government’s emergency guidelines, which significantly limited its operational
capacity, until a ceasefire came into effect on June 24, 2025.
Until the ceasefire went into effect, Israel’s airspace and commercial ports closure severely impacted logistics and the Company’s ability to receive goods and raw materials from overseas. As a
result, certain shipments and deliveries of certain components for manufacturing and production were significantly delayed or diverted, leading to supply limitations and delayed shipments to customers.
The intensity and duration of the multi-front conflict are difficult to predict, as are such conflict’s economic implications on the Company’s business and operations and on Israel’s economy in
general. The potential deterioration of Israel’s economy, as a direct and indirect result of these events, may have a material adverse effect on the Company and its ability to effectively conduct its operations.
On October 9, 2025, Israel and Hamas signed an agreement in Sharm el-Sheikh, mediated by the United States, Egypt, Qatar, and Turkey, as the first phase of a U.S. 21-point plan. The agreement was
announced on October 9 and approved by Israel’s government in the early hours of October 10, after which the initial ceasefire took effect. While implementation is ongoing and violations have been reported, the agreement’s scope, durability, and
downstream economic effects remain uncertain. The Company continues to monitor developments and will adjust operations and disclosures as appropriate.
66
|
Management’s Discussion and Analysis
|
|
JUDICIAL AND LEGISLATIVE
REFORMS IN ISRAEL
Israel is undergoing political and social instability relating to the judicial and legislative reforms proposed by the current government, creating certain instability and uncertainty. This
instability which has a certain effect on the activity of the financial markets may cause material impact on the Groups’ ability to operate in the Israeli market, which derives, among other, from: exposure to currency exchange rate and interest rate,
reduced sales due to disruptive days and lower probability for capital investments.
On April 1, 2024, the April 2024 Israeli Cannabis Reform was implemented. For more information, see “Regulatory Reform from Licenses to
Prescriptions for Medical Treatment of Cannabis” above.
CCAA
PROCEEDINGS
On September 14, 2023, a CCAA Termination Order was granted by the Honourable Justice Osborne (upon service on the Service List of an executed certificate and the above CCAA proceedings under the Companies Creditors’ Arrangement Act and the Stay Period were terminated without any further act or formality. On September 29th,
2023, Trichome Financial Corp. filed (or was deemed to have filed) an assignment (or a bankruptcy order was made against Trichome Financial Corp.), and Goldhar & Associates Ltd., was appointed as trustee of the estate of the bankrupt by the
official receiver (or the Court). The first meeting of creditors of the bankrupt was held on October 17th, 2023.
As a direct or indirect shareholder of the entities that make up the Trichome Group, the Company was subject to the priorities of other stakeholders in the CCAA proceedings and ultimately did not
realize any return in the restructuring of the Trichome Group business.
ANTI-DUMPING
INVESTIGATION
In January 2024, Israel’s Ministry of Economy and Industry launched an “anti-dumping” investigation into Canadian cannabis imports, targeting several producers including Tilray,
Canopy Growth, and Cronos Group. The investigation aimed to determine whether Canadian companies were harming Israel’s domestic cannabis industry by selling at unfairly low prices. The process included deadline extensions due to the Iron Swords war,
public consultations, a preliminary ruling confirming dumping and harm, and legal battles over the lack of a temporary guarantee. While the MEI Commissioner recommended a tiered tax on imports, the final decision required approval from both the
Minister of Economy and the Minister of Treasury.
Despite the Minister of Economy’s April 2025 decision to impose a levy of up to 165%, the Minister of Treasury rejected the proposal, citing broader economic harm and the
benefits of import competition. The Ministry of Justice later upheld the Treasury’s veto as lawful, confirming that without Treasury approval, the levy could not be imposed. This conflict led to further court petitions, where the Company, through its
subsidiaries, argued that the Treasury’s decision was legally binding and supported by sound macroeconomic reasoning. The matter remains in legal dispute, with the Company opposing any reversal of the Finance Minister's authority.
For more information, see “Anti-Dumping” investigation into cannabis imports from Canada" above.
ENVIRONMENTAL
RISKS
The Group’s operations are subject to environmental and occupational safety laws and regulations in certain jurisdictions, concerning, among other things, emissions and discharges to water, air and
land, the handling and disposal of hazardous and nonhazardous materials and wastes, and employee health and safety. The Group incurs ongoing costs and obligations related to compliance with environmental and employee health and safety matters. Any
failure to comply or maintain compliance with environmental and occupational safety laws and regulations may result in additional costs for corrective measures, penalties or restrictions on manufacturing operations and could have a material adverse
effect on the business, results of operations and financial condition of the Group.
67
|
Management’s Discussion and Analysis
|
|
RISKS
INHERENT IN THE AGRICULTURAL BUSINESS
The Company’s business involves the growing of cannabis products by third party suppliers, which are agricultural products. As such, the business is subject to the risks inherent in the agricultural
business, such as pests, plant diseases and similar agricultural risks. Although, the third-party cultivators the Company partner with carefully monitor the growing conditions with trained personnel and applicable equipment, there can be no assurance
that natural elements will not have a material adverse effect on the production of its products and results of operations. Any decline in production could have a material adverse effect on the Group’s business, operating results or financial
condition.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
Certain statements in this MD&A may contain “forward-looking statements” or “forward-looking information,” within the meaning of applicable Canadian and United States securities legislation
(collectively referred to herein as “forward-looking statements”). All statements other than statements of fact may be deemed to be forward-looking statements, including statements with regard to expected financial performance, strategy and business
conditions. The words “believe”, “plan”, “intend”, “estimate”, “expect”, “anticipate”, “continue”, or “potential”, and similar expressions, as well as future or conditional verbs such as “will”, “should”, “would”, and “could” often identify
forward-looking statements. These statements reflect management’s current expectations and plans with respect to future events and are based on information currently available to management including based on reasonable assumptions, estimates,
internal and external analysis and opinions of management considering its experience, perception of trends, current conditions and expected developments as well as other factors that management believes to be relevant as at the date such statements
are made. No assurance can be given that the expectations in any forward-looking statement will prove to be correct and, as such, the forward-looking statements included in this MD&A should not be unduly relied upon. Forward-looking statements is
by its nature prospective and requires IM Cannabis to make certain assumptions and is subject to inherent risks and uncertainties. All forward-looking statements are provided as of the date of this MD&A. The Company does not undertake to update
any such forward-looking statements whether as a result of new information, future events or otherwise, except as required by law.
Forward-looking statements in this MD&A may include, without limitation, forward-looking statements pertaining to:
| • |
the Company’s business objectives and milestones and the anticipated timing of execution;
|
| • |
the performance of the Company’s business, strategies and operations;
|
| • |
the Company’s intentions to expand its business, operations and potential activities;
|
| • |
the Company’s plans to expand sales channels, distribution, delivery, storage capacity, and reach to medical cannabis patients;
|
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets in the jurisdictions in which the Company operates;
|
| • |
the Company’s ability to maintain or grow market share and maintain its competitive advantages;
|
| • |
statements relating to the Company’s commitment to responsible growth and compliance with the strictest regulatory environments;
|
68
|
Management’s Discussion and Analysis
|
|
| • |
the Company’s focus on providing premium cannabis products to medical patients in the jurisdictions in which the Company conducts business and any other jurisdiction in which the Company may conduct business in the future;
|
| • |
the Company’s plans to amplify its commercial and brand power to become a global high-quality cannabis player;
|
| • |
the Company’s primary goal of sustainably increasing revenue in its core markets;
|
| • |
the demand and momentum in the Company’s Israeli and Germany operations;
|
| • |
how the Company intends to position its brands;
|
| • |
the efficiencies and synergies of the Company as a global organization with domestic expertise in Israel and Germany;
|
| • |
expectations that providing high-quality, reliable supply to the Company’s customers and patients will lead to recurring sales;
|
| • |
expectations related to the Company’s introduction of new SKUs
|
| • |
anticipated cost savings from the reorganization of the Company and the completion thereof upon the timelines disclosed herein;
|
| • |
geographic diversification and brand recognition and the growth of the Company’s brands in the jurisdictions that the Company operates in or may expand to;
|
| • |
expectations related to the Company’s ability to address the ongoing needs and preferences of medical cannabis patients;
|
| • |
the Company’s retail presence, distribution capabilities and data-driven insights;
|
| • |
the future impact of the Regulations Amendment (as defined herein) regarding the transition from licenses to prescriptions for medical treatment of cannabis in Israel;
|
| • |
the Company’s continued partnerships with third party suppliers and partners and the benefits thereof;
|
| • |
the Company’s ability to achieve profitability in 2025;
|
| • |
the number of patients in Israel licensed by the Israeli Ministry of Health (“MOH”) to consume medical cannabis;
|
| • |
expectations relating to the number of patients paying out-of-pocket for medical cannabis products in Germany;
|
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany;
|
| • |
expectations related to the demand and the ability of the Company to source premium and ultra-premium cannabis products exclusively and competition in this product segment;
|
| • |
the anticipated impact of inflation and liquidity on the Company’s performance;
|
| • |
expectations with respect to the Company’s operating budget and the assumptions related thereto;
|
| • |
expectations relating to the Company as a going concern and its ability to conduct business under the ordinary course of operations;
|
| • |
expectations related to the collection the payment awarded in the Judgment and the chances of the claim advancing or the potential outcome of the Test Kits Appeal (as defined herein);
|
| • |
the continued listing of the Common Shares on Nasdaq;
|
| • |
cannabis licensing in the jurisdictions in which the Company operates;
|
| • |
the renewal and/or extension of the Company’s licenses;
|
| • |
the Company’s anticipated operating cash requirements and future financing needs;
|
69
|
Management’s Discussion and Analysis
|
|
| • |
the Company’s expectations regarding its Gross Margins, EBITDA, Adjusted EBITDA, revenue, expenses, profit margins and operations;
|
| • |
the expected increase in revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
future opportunities for the Company in Israel, particularly in the retail and distribution segments of the cannabis market;
|
| • |
future expansion and growth opportunities for the Company in Germany and Europe and the timing of such; and
|
| • |
contractual obligations and commitments; and
|
| • |
the Company’s ability to maintain compliance with Nasdaq’s continued listing standards, including regaining and sustaining the minimum stockholders’ equity requirements or satisfying alternative criteria;
|
| • |
the potential impact of a delisting from Nasdaq on the Company’s operations, financing ability, and shareholder value;
|
| • |
the Company’s plans to access the capital markets under the Shelf Registration Statement filed on Form F-3 and the use of proceeds from any such offerings;
|
| • |
expectations relating to the Company’s ability to complete future private placements or loan agreements to meet liquidity needs;
|
| • |
the Company’s ability to successfully implement its cost-reduction and operational efficiency plans to support its going concern assumptions;
|
| • |
expectations relating to potential acceleration rights or repayment terms under outstanding loan agreements;
|
| • |
the expected resolution of delays in Canadian export permits and the impact on Israeli operations and supply chain continuity;
|
| • |
the anticipated benefits of the acquisition of the remaining interest in Focus Medical Herbs Ltd. and the expected synergies;
|
| • |
the Company’s ability to achieve sales growth through the introduction of new cannabis strains and product lines in Israel and Germany;
|
| • |
expectations related to ongoing tax obligations, debenture conversions, or settlement agreements.
|
With respect to the forward-looking statements contained in this MD&A, the Company has made assumptions regarding, among other things:
| • |
the Company has the ability to achieve its business objectives and milestones under the stated timelines;
|
| • |
the Company will succeed in carrying out its business, strategies and operations;
|
| • |
the Company will realize upon its intentions to expand the business, operations and potential activities of the Company;
|
| • |
the Company will expand its sales channels, distribution, delivery and storage capacity, and reach to medical cannabis patients;
|
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis in the jurisdictions in which the Company operates;
|
| • |
the competitive conditions of the industry will be favorable to the Company, and the Company has the ability to maintain or grow its market share and maintain its competitive advantages;
|
| • |
the Company will commit to responsible growth and compliance with the strictest regulatory environments;
|
70
|
Management’s Discussion and Analysis
|
|
| • |
the Company will remain focused on providing premium cannabis products to medical patients in the jurisdictions in which the Company conducts business and any other jurisdiction in which the Company may conduct business in the future;
|
| • |
the Company has the ability to amplify its commercial and brand power to become a global high-quality cannabis player;
|
| • |
the Company will maintain its primary goal of sustainably increasing revenue in its core markets;
|
| • |
the demand and momentum in the Company’s Israeli and Germany operations will be favorable to the Company;
|
| • |
the Company will carry out its plans to position its brands as stated;
|
| • |
the Company’s Company has the ability to realize upon the stated efficiencies and synergies the Company as a global organization with domestic expertise in Israel and Germany;
|
| • |
providing a high-quality, reliable supply to the Company’s customers and patients will lead to recurring sales;
|
| • |
the Company will introduce new SKUs;
|
| • |
the Company will realize the anticipated cost savings from its reorganization;
|
| • |
the Company has the ability to achieve geographic diversification and brand recognition and the growth of the Company’s brands in the jurisdictions that the Company operates in or may expand to;
|
| • |
the Company’s has the ability to address the ongoing needs and preferences of medical cannabis patients;
|
| • |
the Company has the ability to realize upon its retail presence, distribution capabilities and data-driven insights;
|
| • |
the future impact of the Regulations Amendment will be favorable to the Company;
|
| • |
the Company will maintain its partnerships with third parties, suppliers and partners;
|
| • |
the Company has the ability to achieve profitability in 2025;
|
| • |
the accuracy of number of patients in Israel licensed by the MOH to consume medical cannabis;
|
| • |
the accuracy of the number of patients paying out-of-pocket medical cannabis products in Germany;
|
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany will occur;
|
| • |
the Company has the ability to source premium and ultra-premium cannabis products exclusively and competition in this product segment;
|
| • |
the anticipated impact of inflation and liquidity on the Company’s performance will be as forecasted;
|
| • |
the accuracy with respect to the Company’s operating budget and the assumptions related thereto;
|
| • |
the Company will remain as going concern;
|
| • |
a favorable outcome with respect to the collection of the awards in successful judgements, and the success of other ongoing claims the Company is involved in;
|
| • |
the Company’s Common Shares will remain listed on the Nasdaq;
|
| • |
the Company’s ability to maintain cannabis licensing in the jurisdictions in which the Company operates;
|
| • |
the Company has the ability to obtain the renewal and/or extension of the Company’s licenses;
|
| • |
the Company has the ability to meet operating cash requirements and future financing needs;
|
71
|
Management’s Discussion and Analysis
|
|
| • |
the Company will meet or surpass its expectations regarding its Gross Margins, EBITDA, Adjusted EBITDA, revenue, expenses, profit margins and operations;
|
| • |
the Company will increase its revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
the Company has the ability to capitalize on future opportunities for the Company in Israel, particularly in the retail and distribution segments of the cannabis market;
|
| • |
the Company will carry out its future expansion and growth opportunities for the Company in Germany and Europe and the timing of such; and
|
| • |
the Company will fulfill its contractual obligations and commitments;
|
| • |
the Company’s plan submitted to Nasdaq to regain compliance will be accepted and effectively executed; and
|
| • |
the Company’s continued access to debt and equity financing under acceptable terms through private placements, loan facilities, or under its Shelf Registration; and
|
| • |
the ability of the Company to comply with covenants and repayment terms under existing and future loan agreements; and
|
| • |
the Company will overcome export permit delays and secure a reliable supply chain to support its sales channels in Israel; and
|
| • |
the acquisition of the remaining stake in Focus Medical Herbs will contribute positively to the Company’s financial results and operational structure; and
|
| • |
that recent and future board and management appointments will support improved governance and financial performance; and
|
| • |
the proposed “Quantum” transaction progressing from the signed non-binding term sheet to definitive agreements and closing, on terms substantially consistent with the term sheet and within anticipated timelines, subject to customary
conditions;
|
| • |
there will be no material adverse changes to the macroeconomic environment (e.g., inflation, interest rates, currency exchange) that impact financing or operations.
|
Readers are cautioned that the above lists of forward-looking statements and assumptions are not exhaustive. Since forward-looking statements address future events and conditions, by their very nature they involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated or implied by such forward-looking statements due to a number of factors and risks. These include:
| • |
the Company’s inability to achieve its business objectives and milestones under the stated timelines;
|
| • |
the Company inability to carry out its business, strategies and operations;
|
| • |
the Company’s inability to realize upon its intentions to expand the business, operations and potential activities of the Company;
|
| • |
the Company will not expand its sales channels, distribution, delivery and storage capacity, and reach to medical cannabis patients;
|
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets will be unfavorable to the Company in the jurisdictions in which the Company operates;
|
| • |
the competitive conditions of the industry will be unfavorable to the Company, and the Company’s inability to maintain or grow its market share and maintain its competitive advantages;
|
| • |
the Company will not commit to responsible growth and compliance with the strictest regulatory environments;
|
72
|
Management’s Discussion and Analysis
|
|
| • |
the Company’s inability to remain focused on providing premium cannabis products to medical patients in the jurisdictions in which the Company conducts business and any other jurisdiction in which the Company may conduct business in the
future;
|
| • |
the Company inability to amplify its commercial and brand power to become a global high-quality cannabis player;
|
| • |
the Company will not maintain its primary goal of sustainably increasing revenue in its core markets;
|
| • |
the demand and momentum in the Company’s Israeli and Germany operations will be unfavorable to the Company;
|
| • |
the Company will not carry out its plans to position its brands as stated;
|
| • |
the Company’s inability to realize upon the stated efficiencies and synergies of the Company as a global organization with domestic expertise in Israel and Germany;
|
| • |
providing a high-quality, reliable supply to the Company’s customers and patients will not lead to recurring sales;
|
| • |
the Company will not introduce new SKUs;
|
| • |
the Company’s inability to realize upon the anticipated cost savings from the reorganization;
|
| • |
the Company’s inability to achieve geographic diversification and brand recognition and the growth of the Company’s brands in the jurisdictions that the Company operates in or may expand to;
|
| • |
the Company’s inability to address the ongoing needs and preferences of medical cannabis patients;
|
| • |
the Company’s inability to realize upon its retail presence, distribution capabilities and data-driven insights;
|
| • |
the future impact of the Regulations Amendment will be unfavorable to the Company;
|
| • |
the Company will not maintain its partnerships with third party suppliers and partners;
|
| • |
the Company’s inability to achieve profitability in the next quarters of 2025;
|
| • |
the inaccuracy of number of patients in Israel licensed by the MOH to consume medical cannabis;
|
| • |
the inaccuracy of the number of patients paying out-of-pocket for medical cannabis products in Germany;
|
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany will not occur;
|
| • |
the Company’s ability to source premium and ultra-premium cannabis products exclusively and competition in this product segment;
|
| • |
the anticipated impact of inflation and liquidity on the Company’s performance will not be as forecasted;
|
| • |
the inaccuracy with respect to the Company’s operating budget and the assumptions related thereto;
|
| • |
the Company will not remain as going concern;
|
| • |
an unfavorable outcome of legal proceedings the Company is involved in;
|
| • |
an unfavorable outcome with respect to the collection of the award in the Judgment of the Test Kits Appeal and the Company being unsuccessful in other ongoing claims the Company is involved in;
|
| • |
the Company’s Common Shares will not remain listed on the Nasdaq;
|
| • |
the Company’s inability to maintain cannabis licensing in the jurisdictions in which the Company operates;
|
| • |
the Company’s inability to obtain the renewal and/or extension of the Company’s licenses;
|
| • |
the Company’s inability to meet operating cash requirements and future financing needs;
|
73
|
Management’s Discussion and Analysis
|
|
| • |
the Company will not meet or surpass its expectations regarding its Gross Margins, EBITDA, Adjusted EBITDA, revenue, expenses, profit margins, and operations;
|
| • |
the Company will not increase its revenue and margins in its Israeli medical cannabis market activities arising from its acquisitions;
|
| • |
the Company’s ability to capitalize on future opportunities for the Company in Israel, particularly in the retail and distribution segments of the cannabis market;
|
| • |
the Company will not carry out its future expansion and growth opportunities for the Company in Germany and Europe and the timing of such;
|
| • |
the Company will not fulfill its contractual obligations and commitments;
|
| • |
the Company may fail to regain or maintain compliance with Nasdaq listing standards, resulting in delisting and reduced access to capital markets;
|
| • |
the Company may not be able to raise sufficient capital under its Shelf Registration or other financing initiatives, leading to liquidity constraints;
|
| • |
the risk of default or acceleration under loan agreements, including those subject to pledges or performance conditions;
|
| • |
continued supply chain disruptions, including export permit delays from Canada, may impact revenue and inventory availability;
|
| • |
the acquisition of Focus Medical Herbs may not yield the anticipated benefits or operational synergies; and
|
| • |
the Company may be unable to collect amounts owed from prior judgments or settlements, affecting its financial position.
|
Readers are cautioned that the foregoing list of risk factors is not exhaustive. Additional information on these and other factors that could affect the business, operations or financial results of
the Company are detailed under the headings “Risk and Factors” and “Contingent Liabilities and Commitments” of this MD&A. The Company and management caution readers
not to place undue reliance on any forward-looking statements, which speak only as of the date made. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it can give no assurance that such
expectations will prove to have been correct. The Company and management assume no obligation to update or revise them to reflect new events or circumstances except as required by applicable securities laws.
Additional information about the assumptions, risks and uncertainties of the Company’s business and material factors or assumptions on which information contained in forward-looking statements is
based is provided in the Company’s disclosure materials, including in this MD&A under “Legal and Regulatory – Risk Factors” and the Company’s Annual Report under “Risk
Factors”, available on the Company’s profile on SEDAR+ at www.sedarplus.ca and on EDGAR at www.sec.gov/edgar.
All forward-looking statements in this MD&A is qualified by these cautionary statements.
Additional information about the Company, including its Annual Report, is available on the Company’s profile on SEDAR+ at www.sedarplus.ca and on EDGAR at www.sec.gov/edgar.
74