SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
|
REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR 12(g) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended |
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period from ____________________ to ____________________ |
|
SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
(Exact name of Registrant as specified in its charter)
(Jurisdiction of incorporation or organization)
(Address of principal executive offices)
(Name, Telephone, E-Mail and/or Facsimile number and Address of Company Contact Person)
|
Title of each class
|
Trading Symbol
|
Name of each exchange on which registered
|
||
|
|
|
The
|
Yes ☐
Yes ☐
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|
|
|
Emerging growth company
|
|
U.S. GAAP ☐
|
|
Other ☐
|
Yes ☐ No

|
Legal Entity |
Jurisdiction |
Relationship with the Company |
|
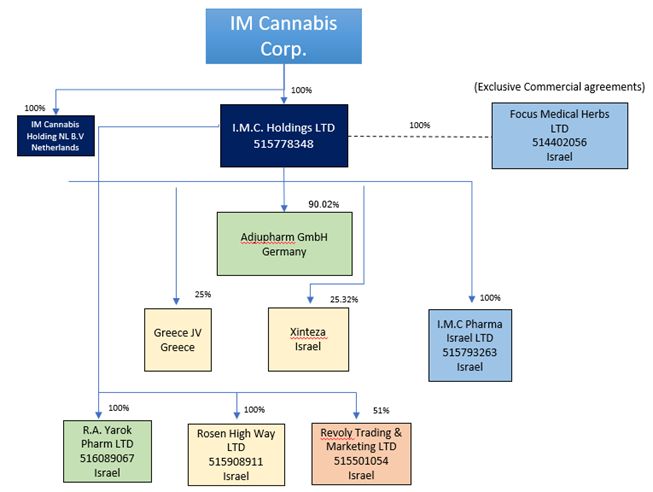
I.M.C. Holdings Ltd. (“IMC Holdings”)
|
Israel |
Wholly-owned subsidiary |
|
I.M.C. Pharma Ltd. (“IMC Pharma”)
|
Israel |
Wholly-owned subsidiary of IMC Holdings |
|
Focus Medical Herbs Ltd. (“Focus")(1)
|
Israel |
Wholly-owned subsidiary of IMC Holdings |
|
R.A. Yarok Pharm Ltd. (“Pharm Yarok”)
|
Israel |
Wholly-owned subsidiary of IMC Holdings |
|
Rosen High Way Ltd. (“Rosen High Way”)
|
Israel |
Wholly-owned subsidiary of IMC Holdings |
|
Rivoly Trading and Marketing Ltd. d/b/a Vironna Pharm (“Vironna”)
|
Israel |
Subsidiary of IMC Holdings |
|
Adjupharm GmbH (“Adjupharm”)
|
Germany |
Subsidiary of IMC Holdings |
|
Trichome Financial Corp. (“Trichome")
(2) |
Canada |
Former wholly-owned subsidiary |
|
Xinteza API Ltd (“Xinteza”)
|
Israel |
Subsidiary of IMC Holdings |
|
Shiran Societe Anonyme (“Greece”)
|
Greece |
Subsidiary of IMC Holdings |
|
IM Cannabis Holding NL B.V Netherlands (“IMC
Holdings NL”) |
Netherlands |
Wholly-owned subsidiary of IMC Holdings |
| (1) |
Effective February 26, 2024, IMC Holdings exercised its option to acquire a 74% ownership stake in Focus, and effective May 26, 2025,
IMC Holdings acquired the remaining 26% from Ewave Group Ltd. |
| (2) |
Discontinued operations. |
| • |
our business objectives and milestones and the anticipated timing of execution; |
| • |
the performance of our business, strategies and operations; |
| • |
our intentions to expand our business, operations, sales channels, distribution, delivery and storage capacity; |
| • |
our ability to reach to medical cannabis patients and address their ongoing needs and preferences; |
| • |
the competitive conditions of the cannabis industry and the growth of medical or adult-use recreational cannabis markets in the jurisdictions
in which we operate; |
| • |
our ability to maintain or grow our market share and maintain our competitive advantages; |
| • |
our focus on providing premium cannabis products and strengthening our brands and commercial position; |
| • |
demand trends and momentum in our Israeli and Germany operations; |
| • |
the benefits of our retail presence, distribution capabilities, data-driven insights and third-party partnerships; |
| • |
expectations regarding recurring sales and operating efficiencies; |
| • |
anticipated cost savings from our reorganization and the timing thereof; |
| • |
the impact of the Regulations Amendment (as defined herein) and other regulatory developments; |
| • |
our ability to achieve profitability, improve margins and manage liquidity; |
| • |
the number of patients in Israel licensed by the Israeli Ministry of Health (“MOH”)
to consume medical cannabis and the number of patients paying out-of-pocket for medical cannabis products in Germany; |
| • |
the anticipated decriminalization or legalization of adult-use recreational cannabis in Israel and Germany; |
| • |
our operating budget and the assumptions underlying it; |
| • |
our ability to continue as a going concern; |
| • |
the outcome of legal proceedings; |
| • |
the continued listing of our Common Shares on the Nasdaq Stock Market LLC (“Nasdaq”);
|
| • |
our ability to maintain, renew and extend cannabis licenses; |
| • |
our anticipated operating cash requirements and future financing needs; |
| • |
our expectations regarding revenue, expenses, profit margins and operations; and |
| • |
our entry into, and development of, new lines of business; |
| • |
future opportunities for growth in Israel, Germany and Europe. |
| • |
We have incurred significant losses since our inception and anticipate that we will incur continued losses;
|
| • |
We have generated insufficient revenues from product sales and may never be profitable; |
| • |
If our operational cash flows continue to be negative, we may be required to fund future operations with
alternative financing options such as offerings of shares; |
| • |
We may not be able to secure the funds necessary to implement our strategies; |
| • |
In pursuit of new opportunities in the cannabis industry, we may fail to select appropriate investment
candidates and negotiate acceptable arrangements; |
| • |
Unfavorable divestments could have a material adverse effect on us; and |
| • |
We are subject to the inherent liquidity risk that we will not be able
to pay our financial obligations as they become due; |
| • |
Regulatory authorities in Israel may determine that we are in contravention of Israeli cannabis regulations;
|
| • |
Our operations are subject to various laws, regulations and guidelines, and any potential noncompliance
could cause our business, financial condition and results of operations to be adversely affected; |
| • |
Our operations are subject to a variety of laws, regulations, and guidelines, to which any changes could
have a material adverse effect on our business, results of operations, financial condition and prospects; |
| • |
Failure to meet target production capacity may result in a material adverse effect on our business, financial
condition; and |
| • |
Our operations are subject to environmental and occupational safety laws and regulations,
any failure to comply with such laws and regulations could have a material adverse effect on us; and |
| • |
Our ability to produce, store, import, distribute and sell cannabis and derivative products is dependent
on licensing and any failure to maintain such licenses would have a material adverse impact; |
| • |
Increased competition could materially and adversely affect our business, financial condition and results
of operations; |
| • |
We are vulnerable due to the results of the “Anti-Dumping” investigation into cannabis imports
from Canada that could have a material adverse effect on our business; |
| • |
We rely on business licenses, permits and approvals and the failure to maintain those could have a material
adverse effect on our business, financial condition and results of the operations; |
| • |
We rely on key business inputs and any failure to secure required supplies and services or to do so on
appropriate terms could also have a material adverse effect on our business, financial condition, and operating results; |
| • |
We rely upon the ability, judgment, discretion and good faith of key personnel, and the inability to attract,
develop, motivate and retain highly qualified employees could have a material adverse effect on our business, financial condition and
results of operations; |
| • |
Future acquisitions or dispositions could result in the failure to realize anticipated benefits of such
transactions; |
| • |
The failure to design, develop or maintain effective internal controls may affect our ability to prevent
fraud, detect material misstatements, and fulfill reporting obligations. |
| • |
The insurance purchased by the us cannot cover all risks that we are exposed to, and any uninsured amounts
of liabilities incurred by our member(s) may be paid directly by such members; and |
| • |
Our products face an inherent risk of exposure to product liability claims, regulatory
action and litigation if such products are alleged to have caused significant loss or injury; |
| • |
Our principal shareholders, officers and directors beneficially own approximately 23.4% of our Common
Shares, and therefore be able to exert significant control over matters submitted to shareholders’ approval; We may issue additional
securities in the future, which may dilute our shareholder’s holdings. |
| • |
The possible lack of liquidity of securities may cause difficulty for security holders to re-sell securities
at desired prices; |
| • |
Loss of foreign private issuer status under United States securities laws could increase our regulatory
and compliance costs; and |
| • |
We may be a “passive
foreign investment company” (“PFIC”), for U.S. federal income tax purposes in
the current taxable year or become a PFIC in the future. There generally would be negative tax consequences for U.S. taxpayers that are
holders of the Common Shares if we are a PFIC. |
| • |
We use intellectual property protections to protect our products, brands and technologies. Maintaining
such protections and paying any costs attributed to the enforcement of such protections does not completely eliminate the risk of litigation,
which may result in a material adverse effect on our business, financial condition, results of operations and prospects. |
| • |
We are vulnerable to the political, economic, legal, social, regulatory, and military conditions
affecting Israel and the Middle East that could have a material adverse effect on our business, results of operations, financial condition
and our prospects. |
| • |
It may be difficult to enforce a U.S. judgment against us, our officers and directors in Israel or the
United States, or to assert U.S. securities laws claims in Israel or serve process on our officers and directors; and |
| • |
Since we are incorporated in British Columbia and substantially all of our directors and officers reside
outside the United Stated, and some outside Canada, it may be difficult in the United States to enforce civil liabilities against us based
solely upon the federal securities laws of the United States. Similarly, it may be difficult to enforce civil liabilities against our
directors and officers residing outside of Canada. |
| • |
are subject to receipt of applicable regulatory approvals and changes in applicable regulations and laws; |
| • |
continue operations in Israel, which is vulnerable to security and military conditions due to heightened tension in the Middle East.
For more information, see “Risks Related to Israeli Law and Our Operations in Israel” –
“We are vulnerable to the political, economic, legal, social, regulatory, and military conditions affecting Israel and the Middle
East that could have a material adverse effect on our business, results of operations, financial condition and our prospects”
in this “Risk Factors” Section. |
| • |
Continue the research and development of our products; |
| • |
Seek regulatory and marketing approval for our products; |
| • |
Seek to attract and retain skilled personal; |
| • |
Create additional infrastructure to support our operations as a public company and our planned future commercialization efforts;
|
| • |
Experience any delays or encounter issues with respect to the above. |
| • |
finance our operations and the import of cannabis to Israel and Germany; |
| • |
establish and maintain supply and, if applicable, manufacturing relationships with third parties that can provide, in both amount
and quality, adequate products to support development and the market demand for our products, and any future products; |
| • |
any delays and disruptions we encounter in our supply chains; |
| • |
the stability of the operations of our suppliers and service providers, and our relationship with them; |
| • |
maintain sufficient average selling price for our products and the revenues margin we generate; |
| • |
enter into agreements with commercial partners; |
| • |
obtain market acceptance to our products and any future products; |
| • |
accurately identifying demand for our products and any future products; |
| • |
launch and commercialize any products for which we obtain regulatory and marketing approval; |
| • |
address any competing technological and market developments that impact our products and their usage; |
| • |
negotiate favorable terms in any collaboration, licensing or other arrangements into which we may ener and perform our obligations
under such collaborations; |
| • |
divest our cannabis operations and retain our existing pharmacy business in Israel; |
| • |
diversifying into a new business line, unrelated to the legacy cannabis business or the existing pharmacy business in Israel;
|
| • |
attract, hire and retain qualified personnel; and |
| • |
any changes in applicable regulations and laws; |
| ● |
diversion of management time and focus from operating our business to addressing acquisition integration challenges; |
| ● |
coordination of research and development and sales and marketing functions; |
| ● |
retention of employees from the acquired company; |
| ● |
cultural challenges associated with integrating employees from the acquired company into our organization; |
| ● |
integration of the acquired company's accounting, management information, human resources, and other administrative systems;
|
| ● |
the need to implement or improve controls, procedures, and policies at a business that prior to the acquisition may have lacked effective
controls, procedures, and policies; |
| ● |
potential write-offs of intangible assets or other assets acquired in transactions that may have an adverse effect on our operating
results in a given period; |
| ● |
liability for activities of the acquired company before the acquisition, including patent and trademark infringement claims, violations
of laws, commercial disputes, tax liabilities, and other known and unknown liabilities; and |
| ● |
litigation or other claims in connection with the acquired company, including claims from terminated employees, consumers, former
stockholders, or other third parties. |
| • |
engagement of a new third-party processing facility. |
| • |
changes to shipping and distribution service providers. |
| • |
reductions in headcount and closure of certain trading activities.
adjustments to operations in response to logistical disruptions associated with regional hostilities. |
| - |
The Craft Collection – IMC brand’s premium product line with indoor-grown, hand-dried
and hand-trimmed high-THC cannabis flowers. The Craft Collection includes exotic and unique cannabis strains such as Sup.S. |
| - |
The Top-Shelf Collection – IMC’s premium product line which offers indoor-grown,
high-THC cannabis flowers with strains such as Lemon Rocket, Diesel Drift, Tropicana Gold, Lucy Dreamz, Santa Cruz, Or’enoz, and
Banjo. Inspired by the 1970's cannabis culture in America, the Top-Shelf Collection targets the growing segment of medical patients who
are cannabis culture enthusiasts. |
| - |
The Signature Collection – IMC brand’s high-quality product line with greenhouse-grown
or indoor grown, high-THC cannabis flowers. The Signature Collection currently includes well known proprietary cannabis dried flowers
such as Chemchew, Rockabye, FLO OG, Roma T15, Roma T20, Karma lada, Sydney, MOTORBRTH and B.F LMO, which are all indoor-grown flowers.
|
| - |
The Full Spectrum Extracts – IMC brand’s full spectrum, strain-specific cannabis
extracts, which includes high-THC Roma®T20 oil and OIL GLTO 33. |
| - |
Roma® Product Portfolio – IMC's Roma® portfolio
also includes oils. IMC’s Roma® strain is a high-THC medical cannabis flower that offers a therapeutic continuum and is known
for its strength and longevity of effect. |
| - |
BLKMKT™ – this brand is our second Canadian brand. It is a super-premium product
line with indoor-grown, hand-dried and hand-trimmed high-THC cannabis flowers. The BLKMKT™ includes BLK MLK, YA HEMI, PURPLE RAIN,
JEALOUSY, Hemi GLTO, RAINBOW P, GUVA BOBA, Sunsets.rudel, Park fire OG and Up side down C. |
| - |
LOT420 – this brand launched in Israel in
the second quarter of 2023, with super-premium indoor-grown cannabis with high-THC imported
from Canada. The LOT420 brand includes GLTO 33, Apps and Bans and O.C. We
ceased selling Atomic APP. |
| - |
The PICO collection (minis) - Under the BLKMKT™ and LOT420 brands, we launched in 2023
a new type of product (small flowers), which is a super-premium indoor-grown cannabis imported from Canada with high-THC. The PICO collection
includes the following products: PICO PURPLE RAIN, PICO YA HEMI, PICO JEALOUSY, Pico upside Down, PICO RAIN BOW, Pico California love,
PICO BLK MLK and PICO Bacio Glto. |
| - |
Flower – In the second quarter of 2024, we launched a super-premium indoor-grown cannabis
with high-THC imported from Canada. The Flower brand includes cannabis strains called California Love and Face Sherb. |
|
Revenues from Continuing Operations - By Product Type
(in thousands of Canadian dollars) | |||
|
Financial Year |
Medical Cannabis Products |
Other Products |
Total |
|
2025 |
$53,186 |
$1,545 |
$54,731 |
|
2024 |
$51,355 |
$2,696 |
$54,031 |
|
2023 |
$44,246 |
$4,558 |
$48,804 |
|
Revenues from Continuing Operations - By Geographic Market
(in thousands of Canadian dollars) | |||
|
Financial Year |
Israel |
Germany |
Total |
|
2025 |
$18,383 |
$36,348 |
$54,731 |
|
2024 |
$38,523 |
$15,508 |
$54,031 |
|
2023 |
$43,316 |
$5,488 |
$48,804 |
| 1. |
Change in the prescription process: patients with a wide range of diseases and medical conditions from Oncology to Parkinsons will
no longer be required to obtain a license to receive medical cannabis. Patients will receive a prescription similar to those for other
prescription medications. Pain and PTSD are not included in the April 2024 Regulatory Reform yet. |
| 2. |
Medical cannabis will now be prescribed through the HMO's, Israel's public healthcare system: until the April 2024 Regulatory Reform,
cannabis could not be prescribed through the HMO's which cover the majority of the Israeli population. |
| 3. |
The number of prescribing physicians is expected to increase: as of today, HMO physicians, who are dully trained and certified within
their field of expertise, can prescribe medical cannabis as a first line treatment, as opposed to a last resort, based on medical discretion
for the approved indications. |
| 4. |
The cost for prescription is anticipated to be reduced: the Ministry of Health limited the cost for a medical cannabis prescription.
|
|
Name |
Position(s) with us |
Age |
Other Directorships |
Date of Initial Appointment |
|
Oren Shuster(4)
|
Chief Executive Officer and Director |
57 |
IMC Holdings; Focus; Pharm Yarok; Rosen High Way; IMC Pharma; IMC Farms; Ewave Group Ltd (“Ewave”) and its subsidiaries; |
October 11, 2019 |
|
Asi Levi |
Chief Financial Officer |
46 |
- |
December 11, 2025 |
|
Oz Adler |
Chairman of the Board |
39 |
Elbit Imaging Ltd;
Clearmind Inc;
Rail Vision Ltd;
Jeffs’ Brands Ltd; and
Polyrizon Ltd. |
July 7 ,2025 |
|
Alon Dayan |
Director |
50 |
SciSparc Ltd; and
ViewBix Inc. |
December 31, 2025 |
|
Assaf Yitzhaik(1)
|
Director |
53 |
Rani Zim Shopping Centers Ltd;
Gix Internet Ltd;
Clearmind Medicine Inc;
Save Foods Inc;
Jeffs’ Brands Ltd;
Plyrizon Ltd; and
Plentify Ltd; |
September 15, 2025 |
|
Einat Zakariya(1)(2)(3)
|
Director |
55 |
- |
September 1, 2022 |
|
Eli Zamir(1)(2)
|
Director |
56 |
Newmed Energy |
September 15, 2025 |
|
Moti Marcus(1)(2)(3)
|
Director |
63 |
- |
September 12, 2022 |
|
Richard Balla |
Chief Executive Officer of Adjupharm, a wholly owned subsidiary of the Company
|
40 |
- |
March 21, 2019 |
| (1) |
Member of the Audit Committee. |
| (2) |
Member of the Compensation Committee |
| (3) |
Member of the Governance and Nomination Committee. |
| (1) |
Oren Shuster, CEO, Executive Chairman and a director; |
| (2) |
Asi Levi, CFO; |
| (3) |
Oz Adler, Chairman of the Board; |
| (4) |
Richard Balla, Chief Executive Officer of Adjupharm, a wholly owned subsidiary of the Company;
|
| (5) |
Michal Lebovitz Nissimov, former General Counsel and Secretary; |
| (6) |
Uri Birenberg, former CFO. |
| 1. |
base salary; |
| 2. |
cash bonuses; and/or |
| 3. |
long-term incentives. |
| 1. |
Base Salary |
| 2. |
Cash Bonuses |
| 3. |
Long Term Incentives |
| • |
The aggregate number of Common Shares issuable pursuant to all Awards shall not exceed 20% of the issued
and outstanding Common Shares at the time of granting Awards (on a non-diluted basis). |
| • |
If any Award expires or otherwise terminates for any reason without having been exercised, vested or purchased
(as applicable) in full, the Common Shares in respect of such expired or terminated Award shall again be available for the purposes of
granting Awards pursuant to the Equity Incentive Plan. |
| • |
Subject to the provisions of the Equity Incentive Plan, the Board, or its delegate, shall have authority
to interpret the Equity Incentive Plan and all Award agreements entered into in connection with the grant of Awards under the Equity Incentive
Plan, to define the terms used in the Equity Incentive Plan and in all Award agreements entered into thereunder, to prescribe, amend and
rescind the terms of the Equity Incentive Plan and to make all other determinations necessary or advisable for the administration of the
Equity Incentive Plan. |
| • |
The price per share at which any Common Share which is the subject of an Option may be purchased (the “Option
Exercise Price”) will be established by the Board, or its delegate, subject to the rules of the regulatory authorities having
jurisdiction over our securities, provided that the Option Exercise Price shall not be less than the greater of $0.05, and the closing
market prices of the underlying securities on (a) the trading day prior to the date of grant of the Options, and (b) the date of grant
of the Options. The term of each Option will be fixed by the Board, or its delegate, and set out in the applicable Award Certificate,
subject to compliance with the Equity Incentive Plan and applicable securities laws and stock exchange policies. |
| • |
Options granted pursuant to the Equity Incentive Plan shall be exercisable at such times and on the occurrence
of such events, and be subject to such restrictions and conditions, as the Board, or its delegate, shall in each instance approve, which
need not be the same for each grant or for each Awardee. Without limiting the foregoing, the Board, or its delegate, may permit the exercise
of an Option through either a cashless exercise mechanism or net exercise mechanism pursuant to the terms of the Equity Incentive Plan.
|
| • |
Awards may be granted to Awardees as compensation for employment or consulting services or services as
a director or officer and may entitle Awardees to receive, for no additional cash consideration, Common Shares upon specific time or other
vesting conditions being met as determined by the Board, or its delegate. The value of Awards is influenced by the fair market value of
the underlying Common Shares, as determined by the Board, or its delegate, pursuant to the terms of the Equity Incentive Plan. |
| • |
If an Award expires, terminates or is cancelled within or immediately after a Black‑Out, the holder
may elect to extend the term to the date that is 10 business days after the last day of the Black‑Out. |
| • |
The maximum number of Common Shares which may be issued to any one Awardee, who is a Related Party may
not exceed: (a) within any 12-month period under the Equity Incentive Plan is 5% of the number of Common Shares outstanding (on a fully-diluted
basis) from time to time; and (b) at any time under the Equity Incentive Plan is 5% of the Common Shares outstanding on a fully-diluted
basis from time to time, in each case unless shareholder approval is obtained pursuant to the Regulatory Rules. |
| • |
The maximum number of Common Shares which may be issuable to all Investor Relations Service Providers within
any 12-month period under the Equity Incentive Plan shall not exceed 2% of the number of Common Shares outstanding. |
| • |
The maximum number of Common Shares which may be issuable to all Related Persons at any time under the
Equity Incentive Plan shall not exceed (a) 15% of the Common Shares outstanding on a fully-diluted basis from time to time; and (b) within
any 12 month period under the Equity Incentive Plan 15% of the number of outstanding Common Shares on a fully-diluted basis from time
to time, in each case unless shareholder approval is obtained pursuant to the Regulatory Rules. All Awards granted under the Equity Incentive
Plan must be granted in accordance with the Equity Incentive Plan’s requirements and applicable Regulatory Rules, including where
securityholder approval is required if, after the grant, the fully‑diluted thresholds for Related Person(s) would be exceeded.
|
| • |
In the event of an Award Holder’s death, any Vested Options pass to the Personal Representative and
are exercisable on or before the earlier of (i) one year following the date of death and (ii) the applicable Expiry Date. In addition,
any unvested RSUs or Stock Awards previously credited to the Award Holder’s account will be cancelled, and vested RSUs or Stock
Awards will be paid to the Award Holder’s estate, with any settlement or redemption to occur within 12 months following the termination
date. Unless the Board, or its delegate, determines otherwise, Awards held by or exercisable by a Personal Representative shall continue
to vest in accordance with any Vesting schedule. |
| • |
If an Award Holder ceases to hold the position for which an Option was granted (other than by reason of
death or Disability), the Expiry Date of the Option is generally the earlier of (i) the original Expiry Date and (ii) 90 days after the
cessation date; however, if the Award Holder resigns, the Expiry Date is generally 30 days after the cessation date (in each case unless
otherwise determined by the Board, or its delegate, and expressly provided in the Award Certificate). For greater certainty, any unvested
Options are immediately cancelled and forfeited to the Company for no consideration. |
| • |
Where an Award Holder is terminated for cause, the Expiry Date of the Option is the cessation date, and
any unvested Options are immediately cancelled and forfeited to the Company for no consideration. |
| • |
Upon the occurrence of a Triggering Event, the Board, or its delegate, may, without the consent of Award
Holders, (i) cause all or a portion of Awards to terminate, (ii) cause Awards to be exchanged for stock awards of another corporation,
and/or (iii) cause Awards or portions thereof to become immediately exercisable, in each case as the Board, or its delegate, determines
and subject to any required Regulatory Approvals. If the Board, or its delegate, wishes to cause all or a portion of Awards to terminate
on the occurrence of a Triggering Event, it must give written notice to the applicable Award Holders not less than 10 days prior to consummation
of the Triggering Event to permit exercise or purchase of the Vested portion of Awards prior to termination. |
| • |
Subject to any required Regulatory Approvals, the Board, or its delegate, may amend the Equity Incentive
Plan or any existing Award; however, if an amendment to an existing Award would (i) materially decrease the rights or benefits of an Award
Holder or (ii) materially increase the obligations of an Award Holder, the Board, or its delegate, must also obtain the written consent
of the Award Holder, unless otherwise excepted by the Equity Incentive Plan. |
|
|
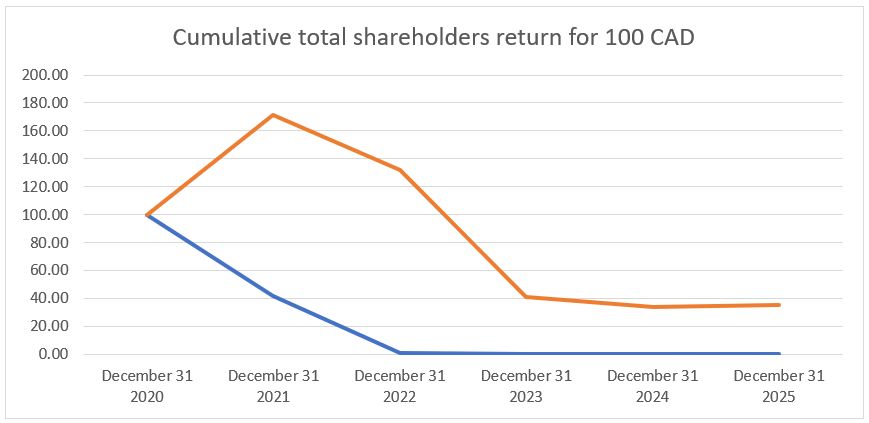
December 31, 2020 |
December 31, 2021 |
December 31, 2022 |
December 31, 2023 |
December 31, 2024 |
December 31, 2025 |
|
IM Cannabis Corp. |
$100.00 |
$42.03 |
$1.29 |
$0.46 |
$0.54 |
$0.23 |
|
CSE Composite Index |
$100.00 |
$171.57 |
$132.1 |
$41.3 |
$33.71 |
$35.07 |
| 1. |
Einat Zakariya (Chair); |
| 2. |
Eli Zamir; and |
| 3. |
Moti Marcus, |
| • |
Salary Benchmarking for Management Positions – Conducting a comparative analysis of
executive salary levels based on a tailored sample of companies. This includes the CEO and senior management positions, with a breakdown
of base salary, bonuses, and equity compensation. |
| • |
Compensation Analysis for International Operations – Assessing salary levels for executives
in Adjupharm, using MERCER and Accumulate market data. This includes benchmarking compensation components such as base salary, bonuses,
and equity grants. |
| • |
Equity Grant Policy Review – Evaluating our equity compensation strategy for Board members
and executives, including grant methods, frequency, and valuation. |
| • |
Equity Compensation Policy Development – Establishing an equity-based compensation framework,
including: |
| o |
Defining a capital compensation strategy. |
| o |
Structuring an allocation model for existing and new employees, from the CEO and Board members downward. |
| o |
Setting guidelines for equity content and reallocation policies. |
| o |
Developing a multi-year allocation plan; and |
| o |
Supporting Compensation Committee approval processes. |
|
Name and Principal Position |
Year |
Salary
($)(1)
|
Share-
Based
Awards
($) |
Option-Based
Awards
($)(2)
|
Non-Equity Incentive Plan
Compensation
($) |
Pension
Value
($) |
All Other
Compensation ($)(3)
|
Total
Compensation ($) | |
|
Annual Incentive Plans |
Long-Term Incentive Plans | ||||||||
|
Oren Shuster(4)
CEO, Executive
Chairman and Director |
2025 |
526,397 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
526,397 |
|
2024 |
484,697 |
Nil |
45,591 |
Nil |
Nil |
Nil |
Nil |
530,288 | |
|
2023 |
476,266 |
Nil |
331,802 |
Nil |
Nil |
Nil |
Nil |
818,068 | |
|
Asi Levi(5)
CFO |
2025 |
12,455 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
12,455 |
|
Uri Birenberg(6)
Former CFO |
2025 |
384,701 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
384,701 |
|
2024 |
330,238 |
Nil |
Nil |
Nil |
Nil |
Nil |
26,064 |
356,302 | |
|
2023 |
73,558 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
73,558 | |
|
Richard Balla
Chief Executive Officer of Adjupharm
|
2025 |
192,000 |
Nil |
Nil |
96,000 |
Nil |
Nil |
29,133 |
317,133 |
|
2024 |
193,608 |
Nil |
Nil |
88,908 |
Nil |
Nil |
31,035 |
313,551 | |
|
2023 |
175,385 |
Nil |
Nil |
87,692 |
Nil |
Nil |
30,895 |
293,972 | |
|
Michal Lebovitz Nissimov
Former General Counsel and
Secretary(7)
|
2025 |
242,509 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
242,509 |
|
2024 |
192,877 |
Nil |
828 |
Nil |
Nil |
Nil |
Nil |
193,705 | |
|
2023 |
119,088 |
Nil |
1,099 |
Nil |
Nil |
Nil |
Nil |
120,187 | |
|
Marc Lustig, former
Executive Chairman(8)
|
2024 |
22,400 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
22,400 |
|
2023 |
129,920 |
79,959 |
Nil |
Nil |
Nil |
Nil |
Nil |
209,879 | |
|
Oz Adler, Executive Chairman(9)
|
2025 |
36,293 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
36,293 |
|
(1) |
Each of Messrs. Shuster, Levi, Birenberg, Fisher, and Mrs. Lebovitz Nissimov received
their compensation in NIS and Mr. Balla received his compensation in Euros. All salaries were converted to CAD pursuant to the average
Bank of Canada rate for the applicable fiscal year. |
|
(2)
|
We used the Black-Scholes pricing model as the methodology to calculate the grant
date fair value, and relied on the following the key assumptions and estimates for each calculation under the following assumptions: (i)
risk free interest rate of 0.42% to 4.21% (ii) expected dividend yield of 0%; (iii) expected volatility of 76.28% to 82.31%; and (iv)
a term of 2 to 10 years. The Black-Scholes pricing model was used to estimate the fair value as it is the most accepted methodology.
|
|
(3) |
All other compensation includes cash bonuses. |
|
(4)
|
Oren Shuster, through Ewave, a company in which
he controls, entered into a consulting agreement with us pursuant to which he is paid NIS 108,350 plus VAT per month (approximately $43,900
plus tax per month) in consideration of his services as CEO. Mr. Shuster did not earn consideration for his role as Chairman and nor as
a director of us during the fiscal years ended December 31, 2025, 2024 and 2023. |
|
(5) |
Mr. Levi was appointed as our CFO effective December 11, 2025. |
|
(6) |
Mr. Birenberg was appointed our CFO effective October 10, 2023 and served as our CFO
until January 5, 2026. |
|
(7) |
Michal Lebovitz Nissimov was appointed as our General Counsel and Secretary effective
April 14, 2023 and served as our General Counsel and Secretary until January 15, 2026. |
|
(8) |
21,949 Warrants are held by Marc Lustig. Mr. Lustig resigned from his role as a director
effective June 5, 2024. |
|
(9) |
Mr. Adler was appointed as our Chairman of the Board effective July 7, 2025.
|
|
Option-based
Awards | ||||
|
Name |
Number of securities underlying unexercised
Options
(#)(1)
|
Option exercise price
($) |
Option expiration date |
Value of unexercised
in-the-money Options ($)(2)
|
|
Oren Shuster |
21,875 |
3.00 |
October 4, 2026 |
Nil |
|
Asi Levi |
Nil |
Nil |
Nil |
Nil |
|
Uri Birenberg
(Former CFO) |
Nil |
Nil |
Nil |
Nil |
|
Oz Adler |
Nil |
Nil |
Nil |
Nil |
|
Richard Balla |
625 |
96.00 |
July 30, 2029 |
Nil |
|
Michal Lebovitz Nissimov
(Former General Counsel and Secretary) |
500 |
3.00 |
October 4, 2026 |
Nil |
| (1) |
Each Option entitles the holder to purchase one Common Share. |
| (2) |
As of December 31, 2025, the closing price of the Common Shares, was US$1.39 per Common Share. |
|
Name |
Option-based awards – Value
vested during the year
($)(1)
|
Share-based awards – Value vested during the year ($) |
Non-equity incentive plan
compensation – Value earned
during the year
($) |
|
Oren Shuster |
69 |
Nil |
Nil |
|
Asi Levi |
Nil |
Nil |
Nil |
|
Uri Birenberg |
Nil |
Nil |
Nil |
|
Oz Adler |
Nil |
Nil |
Nil |
|
Richard Balla |
Nil |
Nil |
Nil |
|
Michal Lebovitz Nissimov |
2 |
Nil |
Nil |
|
(1) |
As of December 31, 2025, the closing price of the Common Shares was US$1.39 per Common Share. |
|
Name(1)
|
Fees
earned ($)(2) |
Share-based
awards
($) |
Option-based
awards ($)(3) |
Non-equity incentive plan compensation ($) |
Pension value ($) |
All other compensation ($) |
Total ($) |
|
Oz Adler(4)
|
36,293 |
Nil |
Nil |
Nil |
Nil |
Nil |
36,293 |
|
Brian Schinderle(5)
|
54,799 |
Nil |
Nil |
Nil |
Nil |
Nil |
54,799 |
|
Moti Marcus(6)
|
80,635 |
Nil |
Nil |
Nil |
Nil |
Nil |
80,635 |
|
Einat Zakariya(8)
|
76,054 |
Nil |
Nil |
Nil |
Nil |
Nil |
76,054 |
|
Shmulik Arbel |
44,373 |
Nil |
Nil |
Nil |
Nil |
Nil |
44,373 |
|
Alon Dayan(8)
|
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Eli Zamir(9)
|
24,150 |
Nil |
Nil |
Nil |
Nil |
Nil |
24,150 |
|
Assaf Yitzhaik(10)
|
24,150 |
Nil |
Nil |
Nil |
Nil |
Nil |
24,150 |
| (1) |
Each of Mr. Marcus Ms. Zakariya and Mr. Arbel received their compensation in NIS and Mr. Schinderle received his compensation in
USD. All salaries were converted to CAD pursuant to the average Bank of Canada rate for the applicable fiscal year. L5 Capital received
their fees in CAD. |
| (2) |
Other than with respect to Mr. Lustig, each director was entitled to a $13,750 payment per quarter for their role as a director .
For each Audit Committee meeting, the Chair received a $1,500 payment and each other member received a $1,000 payment and for each of
the Compensation Committee and Governance and Nomination Committee meetings, the Chair received a $1,200 payment and each other member
received a $700 payment. |
| (3) |
We used the Black-Scholes pricing model as the methodology to calculate the grant date fair value and relied on the following key
assumptions and estimates for each calculation under the following assumptions: (i) risk free interest rate of 3.23% (ii) expected dividend
yield of 0%; (iii) expected volatility of 128.1% to 137.34%; and (iv) a term of 2 years. The Black-Scholes pricing model was used to estimate
the fair value as it is the most accepted methodology. |
| (4) |
Mr. Adler was appointed as director and Chairman of the Board on September 15, 2025. |
| (5) |
Mr. Schinderle resigned from the Board effective September 15, 2025. As a director, Mr. Schinderle received compensation through
Solidum Capital Advisors LLC. |
| (6) |
Mr. Marcus receives compensation through Marcus Management Services Ltd. |
| (7) |
Mr. Arbel resigned from the Board effective September 15, 2025. As a director, Mr. Arbel received additional compensation for his
services as an adviser to us. This compensation was received in NIS and converted to CAD pursuant to the average Bank of Canada rate for
the applicable fiscal year. |
| (8) |
Mr. Dayan was appointed as director on December 31, 2025. |
| (9) |
Mr. Zamir was appointed as director on September 15, 2025. |
| (10) |
Mr. Yitzhaik was appointed as director on September 15, 2025. |
|
Option-based
Awards |
Share-based
Awards | ||||||
|
Name |
Number of
securities
underlying
unexercised
Options
(1)
(#) |
Option exercise price
($) |
Option expiration
date |
Value
of unexercised
in-the-money
Options(2)
($) |
Number of
shares or
units of
shares that
have not vested
(#) |
Market or
payout value of
share-based
awards that
have not
vested
($) |
Market or
payout value of
vested share-
based awards
not paid out or
distributed
($) |
|
Oz Adler |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
|
Brian Schinderle |
1,500 |
3.00 |
October 4, 2026 |
Nil |
Nil |
Nil |
Nil |
|
Moti Marcus |
1,500 |
3.00 |
October 4, 2026 |
Nil |
Nil |
Nil |
Nil |
|
Einat Zakariya |
1,500 |
3.00 |
October 4, 2026 |
Nil |
Nil |
Nil |
Nil |
|
Shmulik Arbel |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
|
Marc Lustig |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
|
Alon Dayan |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
|
Assaf Yitzhaik |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
|
Eli Zamir |
Nil |
N/A |
N/A |
Nil |
Nil |
N/A |
N/A |
| (1) |
Each Option entitles the holder to purchase one Common Share. |
|
(2) |
The closing price of the Common Shares as at December 31, 2025 was US$1.39 per Common Share. |
|
Name |
Option-based awards – Value
vested during the year(1)
($) |
Share-based awards – Value vested during the year ($) |
Non-equity incentive plan
compensation – Value earned during
the year
($) |
|
Brian Schinderle |
62 |
Nil |
Nil |
|
Moti Marcus |
62 |
Nil |
Nil |
|
Einat Zakariya |
62 |
Nil |
Nil |
|
Assaf Yitzahik |
NA |
NA |
NA |
|
Eli Zamir |
NA |
NA |
NA |
|
(1) |
The closing price of the Common Shares as of December 31, 2025 was US$1.39 per Common Share. |
|
Name of Director |
Board |
Audit Committee |
Compensation Committee |
Special Committee |
|
Oz Adler |
8/19 |
1/7 |
NA |
N/A |
|
Oren Shuster |
19/19 |
1/7 |
NA |
N/A |
|
Moti Marcus |
19/19 |
7/7 |
NA |
1/1 |
|
Einat Zakariya |
18/19 |
6/7 |
2/2 |
N/A |
|
Brian Schinderle(2)
|
13/19 |
5/7 |
NA |
N/A |
|
Shmulik Arbel(3)
|
12/19 |
1/7 |
NA |
N/A |
|
Alon Dayan(4)
|
NA |
NA |
NA |
N/A |
|
Assaf Yitzahik(5)
|
6/19 |
2/7 |
2/2 |
1/1 |
|
Eli Zamir(6)
|
6/19 |
2/7 |
2/2 |
1/1 |
| (1) |
Marc Lustig resigned as Executive Chairman and a director effective June 5, 2024. |
| (2) |
Brian Schinderle resigned as a director effective September 15, 2025. |
| (3) |
Shmulik Arbel resigned as a director effective September 15, 2025. |
| (4) |
Alon Dayan was appointed as director effective December 31, 2025. |
| (5) |
Assaf Yitzhaik was appointed as director effective September 15, 2025. |
| (6) |
Eli Zamir was appointed as director effective September 15, 2025. |
| (a) |
overseeing that our day-to-day business affairs are appropriately managed and taking steps to maintain and enhance an effective senior
management team reporting to the CEO; |
| (b) |
recommending to the Board our financial and operating goals and objectives and, following approval by the Board thereof, consistently
striving to achieve such goals and objectives; |
| (c) |
formulating, and presenting to the Board for approval, long-term business plans, strategies and policies having the objective of
maximizing our long-term success and the creation of shareholder value; |
| (d) |
together with other senior management, as are appropriate, developing and recommending to the Board annual business plans and budgets
that support our long term business plans and strategies; |
| (e) |
developing and implementing, with our senior management, plans, strategies, budgets and policies necessary to achieve our goals and
objectives; |
| (f) |
supervising, maintaining and deploying our resources – human, financial or otherwise – with the purpose and objective
of achieving our operating goals and objectives; |
| (g) |
keeping the Board informed in a timely and candid manner of our progress towards the achievement of its strategic and operational
goals and objectives and of all material deviations from the goals, objectives, plans, strategies, budgets or policies established by
the Board; |
| (h) |
overseeing, evaluating and taking steps to enhance, where necessary, the integrity and reliability of our internal controls, including
its management information systems and financial reporting, and establishing, maintaining, designing and evaluating disclosure controls
and procedures for us; |
| (i) |
identifying and managing business risks we faced, including overseeing the design and implementation of appropriate systems and procedures
to effectively monitor, manage and mitigate such risks; |
| (j) |
ensuring that the Board has regular exposure to our senior management and overseeing the development and succession of our senior
management team; |
| (k) |
evaluating the performance of our senior management and making recommendations with respect to their compensation; |
| (l) |
maintaining a positive and ethical work climate that is conducive to attracting, retaining and motivating a diverse group of top-quality
employees at all levels; |
| (m) |
serving as our principal spokesperson and ensuring that information communicated to the public fairly portrays our position and that
timely and continuous disclosure of our obligations are met; |
| (n) |
representing us in a such a way so as to enhance and maintain our reputation and to promote positive relationships with shareholders,
suppliers, contractors, clients, service providers, strategic partners, creditors, financial institutions, local communities, all levels
of government and the media; and |
| (o) |
fulfilling all other responsibilities as assigned by the Board, in the manner expected by the Board. |
|
Name |
Independence(1)
|
Financial
Literacy(2) |
|
Moti Marcus (Chair) |
Independent |
Financially literate |
|
Asaf Yitzhaik |
Independent |
Financially literate |
|
Eli Zamir |
Independent |
Financially literate |
| 1. |
Within the meaning of subsection 1.4 of NI 52-110 and as determined under Exchange Act Rule 10A-3 and Rule 5605(a)(2) of the
Nasdaq Stock Market Rules. |
| 2. |
Within the meaning of subsection 1.6 of NI 52-110, Item 407(d)(5)(ii)-(iii) of Regulation S-K under the Exchange Act) and Rule 5605(c)(2)(A)
of the Nasdaq Stock Market Rules. |
| (i) |
matters of governance; and |
| (ii) |
the nomination of directors to the Board. |
|
Name |
Independence(1)
|
|
Einat Zakariya (Chair) |
Independent |
|
Oren Shuster |
Non - Independent |
|
Moti Marcus |
Independent |
|
Year |
Full Time |
Part Time |
Total | |||
|
Fiscal 2023 |
95 |
- |
95 | |||
|
Fiscal 2024 |
55 |
- |
55 | |||
|
Fiscal 2025 |
44 |
- |
44 |
|
Year |
Israel |
Germany |
Canada |
Total | ||||
|
Fiscal 2023 |
77 |
18 |
- |
95 | ||||
|
Fiscal 2024 |
35 |
20 |
- |
55 | ||||
|
Fiscal 2025 |
21 |
23 |
- |
44 |
| • |
each person, or group of affiliated persons, known by us to beneficially own five percent (5%) or more of any class of our shares;
|
| • |
each of our Named Executive Officers; |
| • |
each of our directors; and |
| • |
all of our directors and executive officers as a group. |
|
Name of Beneficial Holder |
Number of Common Shares Beneficially Held |
Number of Common Shares Underlying Options |
Option Exercise Price ($) |
Option Expiration Date |
Restricted Share Units |
Debentures |
Warrants |
Total Convertible Securities |
Percentage of Common Shares Beneficially Held Undiluted |
Percentage of Common Shares Beneficially Held Partially Diluted
|
|
Holders of more than 5% of our voting securities: | ||||||||||
|
Oren Shuster*(1)
|
898,350 |
21,875 |
3.00 |
October 4, 2026 |
Nil |
46,512 |
194,111 |
262,498(1)
|
14.72% |
19.02% |
|
Rafael Gabay(2)
|
518,525 |
Nil |
Nil |
Nil |
Nil |
46,396 |
194,087 |
240,483 |
8.49% |
12.43% |
|
Directors and senior management who are not 5% holders: | ||||||||||
|
Oz Adler* |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Alon Dayan* |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Asi Levi |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Assaf Yitzhaik* |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Einat Zakariya* |
10,200 |
1,500 |
3.00 |
October 4, 2026 |
Nil |
Nil |
Nil |
1,500 |
** |
** |
|
Eli Zamir* |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
Moti Marcus* |
Nil |
1,500 |
3.00 |
October 4, 2026 |
Nil |
Nil |
Nil |
1,500 |
Nil |
** |
|
Richard Balla |
Nil |
625 |
96.00 |
July 30, 2029 |
Nil |
Nil |
Nil |
625 |
Nil |
** |
|
All directors and senior management as a group (8 persons): | ||||||||||
|
908,550 |
21,875 |
Nil |
46,512 |
194,111 |
266,292 |
15.41% |
19.93% | |||
| (1) |
769,532 Common Shares and 194,111 Warrants are held by Oren Shuster directly and 128,818 Common Shares are held indirectly through
Ewave, a privately-held entity of which Mr. Shuster owns and controls 50% of the outstanding voting rights. Mr. Shuster also holds 46,512
convertible debentures. |
| (2) |
518,500 Common Shares and 194,087 Warrants are held by Rafael Gabay directly and 25 Common Shares are held indirectly by Ewave, a
privately-held entity of which Mr. Gabay owns and controls 50% of the outstanding voting rights. Mr. Gabay also holds 46,396 convertible
debentures. |
| • |
The contractual party of the Company was not the Stroakmont & Atton. The contract with Stroakmont & Atton was only concluded
as a sham transaction in order to cover up a contract with a company named Uniclaro GmbH (“Uniclaro”). Therefore, Stroakmont
& Atton is not the real purchaser rather than Uniclaro. |
| • |
The Company allegedly placed an order with Uniclaro for a total of 4.3 million Clongene COVID-19 tests, of which Uniclaro claims
to have a payment claim against the Company for a partial delivery of 380,400 Clongene COVID-19 tests in a total amount of EUR 941,897.
Uniclaro has assigned this alleged claim against the Company to Stroakmont & Atton Trading GmbH, and Stroakmont & Atton Trading
GmbH has precautionary declared a set-off against the Company’s claim. |
| 1. |
under the age of 18 years; |
| 2. |
found by a court, in Canada or elsewhere, to be incapable of managing the individual’s own affairs, unless a court, in Canada
or elsewhere, subsequently finds otherwise; |
| 3. |
an undischarged bankrupt; or |
| 4. |
convicted in or out of the Province of British Columbia of an offence in connection with the promotion, formation or management of
a corporation or unincorporated business, or of an offence involving fraud, unless: |
| a. |
the court orders otherwise; |
| b. |
5 years have elapsed since the last to occur of: |
| i. |
the expiration of the period set for suspension of the passing of sentence without a sentence having been passed; |
| ii. |
the imposition of a fine; |
| iii. |
the conclusion of the term of any imprisonment; and |
| iv. |
the conclusion of the term of any probation imposed; or |
| c. |
a pardon was granted or issued, or a record suspension ordered, under the Criminal Records Act (Canada) and the pardon or record
suspension, as the case may be, has not been revoked or ceased to have effect. |
| • |
Focus IP Agreement and Cancellation Note: The intellectual property agreement dated
April 2, 2019, as amended on January 1, 2021, between IMC Holdings and Focus, and the related services agreement dated April 2, 2019,
as amended on January 1, 2021, pursuant to which IMC Holdings derived economic benefits from Focus and Focus (i) used the IMC brand on
an exclusive basis for the sale of cannabis products; and (ii) engaged IMC Holdings to provide certain management and consulting services.
On February 26, 2024, the parties executed a cancellation note terminating the Focus IP Agreement as of such date. |
| • |
Telecana Loan Agreement: On November 29, 2022, IMC Holdings entered into a convertible loan
agreement (the “Telecana Loan Agreement”) with Telecana Ltd. (“Telecana”)
and its sole shareholder, pursuant to which IMC Holdings loaned an aggregate of NIS 1,545 thousand (approximately C$605 thousand) in installments
between January and May 2023. Pursuant to the Telecana Loan Agreement, subject to approval from the IMCA, the loan may be converted into
51% of the share capital of Telecana upon the earlier of (i) receipt of a preliminary license from the IMCA, or (ii) at the sole discretion
of IMC Holdings. |
| • |
Kadimastem Term Sheet and Loan Agreement: On February 28, 2024, we entered into a non-binding
term sheet (the “Kadimastem Term Sheet”) and a loan agreement (the “Kadimastem
Loan Agreement”) with Kadimastem Ltd., in connection with a proposed business combination that would have constituted a reverse
merger. Under the Kadimastem Loan Agreement, Kadimastem agreed to provide a loan of up to $650 thousand to IMC Holdings, secured by certain
assets and a personal guarantee by our Chief Executive Officer. On May 28, 2024, we announced the termination of the Kadimastem Term Sheet.
Pursuant to a separation agreement, the outstanding loan of $300 thousand was repaid with accrued interest by July 31, 2024. |
| • |
Oranim Pharmacy Acquisition and Termination: On March 28, 2022, IMC Holdings acquired 51%
of the rights in the Oranim Pharm Partnership (the “Oranim Pharmacy Acquisition”).
On April 16, 2024, we announced that we would not complete the remaining installment payments, and as a result transferred the 51% interest
back to the seller. On July 8, 2024, the parties executed a cancellation agreement addressing the transfer of shares and related matters.
|
| • |
an individual who is a citizen or resident of the United States; |
| • |
a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) organized under the laws of the United
States, any state thereof or the District of Columbia; |
| • |
an estate whose income is subject to U.S. federal income taxation regardless of its source; or |
| • |
a trust that (i) is subject to the primary supervision of a court within the United States and the control of one or more U.S. persons
for all substantial decisions or (ii) has a valid election in effect under applicable Treasury Regulations to be treated as a U.S. person.
|
| • |
at least 25% of the issued shares of any class or series of our capital stock was owned by or belonged to any combination of
(a) the Non-Canadian Holder, (b) persons with whom the Non-Canadian Holder does not deal at arm’s length for purposes of the Canadian
Tax Act, and (c) partnerships in which the Non-Canadian Holder or a person described in (b) holds a membership interest directly or indirectly
through one or more partnerships, and |
| • |
more than 50% of the fair market value of the Common Shares was derived, directly or indirectly, from one or any combination of:
(i) real or immoveable property situated in Canada, (ii) “Canadian resource properties” (as that term is defined in the Canadian
Tax Act), (iii) “timber resource properties” (as that term is defined in the Canadian Tax Act) or (iv) options in respect
of, or interests in, or for civil law rights in, a property described in any of the foregoing whether or not the property exists.
|
|
(in thousands of U.S. dollars) |
2025 |
2024 |
|
Audit Fees(1) |
$386 |
$281 |
|
Audit-related Fees(2)
|
$- |
$- |
|
Tax Fees(3) |
$64 |
$44 |
|
All Other Fees(4)
|
$- |
$29 |
|
Total |
$450 |
$354 |
| 1. |
Audit Fees consist of fees for professional services rendered for the audit of our annual consolidated financial statements and review
of our interim financial statements, as well as services that are normally provided in connection with statutory and regulatory filings
or engagements. |
| 2. |
Audit-Related Fees consist of assurance and related services reasonably related to the performance of the audit or review of our
financial statements and not reported under “Audit Fees.” |
| 3. |
Tax Fees consist of fees for tax compliance, tax advice and tax planning services. |
| 4. |
All Other Fees consist of fees for products and services other than those described above. |
| • |
an Information Security Policy that articulates our information security practices and procedures to maintain confidence in our business and to protect the confidentiality, integrity, and availability of the information we handle;
|
| • |
a dedicated Cyber Security company responsible for executing on relevant internal and external requirements and identifying appropriate technical and organizational measures to deliver information security in compliance with those requirements;
|
| • |
a Cyber Security company, principally responsible for driving our cybersecurity risk assessment processes, including a formal information security risk assessment on an at least annual basis; our security controls framework and risk remediation and prioritizations; and risk awareness or education programs for employees relating to cybersecurity;
|
| • |
the use of external resources, such as assessors, consultants, and auditors, where appropriate, to assess, test, or otherwise assist with aspects of our security controls;
|
| • |
an external audit of our systems and environments, including an external penetration test, on an annual basis;
|
| • |
cybersecurity training of our incident response personnel and senior management;
|
| • |
a cybersecurity incident response plan that includes procedures for assessing, responding to, remediating, resolving, and conducting post-analysis of cybersecurity incidents;
|
| • |
a vendor assessment program designed to identify and mitigate cybersecurity risks associated with our use of
|
| • |
contractual obligations on third-party vendors to report security incidents, risk identification, or other security-related issues promptly to our designated contact personnel.
|
|
Consolidated Financial Statements for the Years Ended December 31, 2025 and 2024
|
|
Independent Auditors’ Reports
|
|
Consolidated Statements of Financial Position
|
|
Consolidated Statements of Net Loss and Comprehensive Loss
|
|
Consolidated Statements of Changes in Shareholders’ Equity
|
|
Consolidated Statements of Cash Flows
|
|
Notes to the Consolidated Financial Statements
|
|
Description
|
Page
|
|
|
Consolidated Financial Statements and Notes
|
F-1 - F-64
|
|
Exhibit
|
|
|
No.
|
Description of Exhibit
|
|
101.INS*
|
XBRL Instant Document
|
|
101.SCH*
|
XBRL Taxonomy Extension Schema Document
|
|
101.CAL*
|
XBLR Taxonomy Extension Calculation Linkbase Document
|
|
101.DEF*
|
XBRL Taxonomy Extension Definition Linkbase
|
|
101.LAB*
|
XBRL Taxonomy Extension Label Linkbase
|
|
101.PRE*
|
XBRL Taxonomy Extension Presentation Linkbase
|
|
104*
|
Cover Page Interactive Data File – (formatted as Inline XBRL and contained in Exhibit 101)
|
|
Date: March 30, 2025
|
IM Cannabis Corp.
By: /s/ Oren Shuster
Name: Oren Shuster |

|
Page
|
|
|
Report of Independent Registered Public Accounting Firm (PCAOB ID:
|
F-2
|
|
Report of Independent Registered Public Accounting Firm (PCAOB ID:
|
F-3
|
|
F-4 - F-5
|
|
|
F-6 - F-7
|
|
|
F-8 - F-10
|
|
|
F-11 - F-12
|
|
|
F-13 - F-64
|

|
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
|
Fahn Kanne & Co.
Head Office
32 Hamasger Street
Tel-Aviv 6721118, ISRAEL
PO Box 36172, 6136101
T +972 3 7106666
F +972 3 7106660
www.gtfk.co.il
|
|
Board of Directors and Shareholders
IM Cannabis Corp.
|
|
|
|
 |
Kost Forer Gabbay & Kasierer
144 Menachem Begin Road, Building A,
Tel-Aviv 6492102, Israel
|
Tel: +972-3-6232525
Fax: +972-3-5622555
ey.com
|
The Group’s Ability to Continue as a Going Concern
|
December 31,
|
||||||||||||
|
Note
|
2025
|
2024
|
||||||||||
|
ASSETS
|
||||||||||||
|
CURRENT ASSETS:
|
||||||||||||
|
Cash
|
$
|
|
$
|
|
||||||||
|
Restricted cash
|
|
|
||||||||||
|
Trade receivables
|
5
|
|
|
|||||||||
|
Other current assets
|
6
|
|
|
|||||||||
|
Inventory
|
7
|
|
|
|||||||||
|
|
|
|||||||||||
|
NON-CURRENT ASSETS:
|
||||||||||||
|
Investments in affiliate
|
8
|
|
|
|||||||||
|
Property, plant and equipment, net
|
9
|
|
|
|||||||||
|
Intangible assets, net
|
10
|
|
|
|||||||||
|
Goodwill
|
10
|
|
|
|||||||||
|
Right-of-use assets, net
|
11
|
|
|
|||||||||
|
|
|
|||||||||||
|
Total assets
|
$
|
|
$
|
|
||||||||
|
December 31,
|
||||||||||||
|
Note
|
2025
|
2024
|
||||||||||
|
LIABILITIES AND SHAREHOLDERS' EQUITY (DEFICIT)
|
||||||||||||
|
CURRENT LIABILITIES:
|
||||||||||||
|
Current maturities of operating lease liabilities
|
11
|
$
|
|
$
|
|
|||||||
|
Trade payables
|
12
|
|
|
|||||||||
|
Other current liabilities
|
13
|
|
|
|||||||||
|
Overdraft and Credit from bank institution and others
|
14
|
|
|
|||||||||
|
Convertible debentures
|
15
|
|
|
|||||||||
|
Derivative warrants liabilities and prefunded warrants
|
16
|
|
|
|||||||||
|
|
|
|||||||||||
|
NON-CURRENT LIABILITIES:
|
||||||||||||
|
Operating lease liabilities
|
11
|
|
|
|||||||||
|
Credit from bank institution and others
|
14
|
|
|
|||||||||
|
Deferred tax liabilities
|
|
|
||||||||||
|
|
|
|||||||||||
|
Total liabilities
|
|
|
||||||||||
|
CONTINGENT LIABILITIES
|
17
|
|||||||||||
|
EQUITY (DEFICIT) ATTRIBUTABLE TO SHAREHOLDERS OF THE COMPANY:
|
18
|
|||||||||||
|
Share capital and premium
|
|
|
||||||||||
|
Capital reserve from share-based payment transactions
|
|
|
||||||||||
|
Amount received on account of financial instruments and other
|
|
|
||||||||||
|
Capital reserve from translation differences of foreign operations
|
(
|
)
|
(
|
)
|
||||||||
|
Capital reserve from transaction with non-controlling interests
|
18B6
|
(
|
)
|
|
||||||||
|
Capital reserve from transaction with controlling shareholder
|
18C
|
|
|
|
||||||||
|
Accumulated deficit
|
(
|
)
|
(
|
)
|
||||||||
|
Total equity (deficit) attributable to shareholders of the Company
|
(
|
)
|
|
|||||||||
|
Non-controlling interests
|
|
(
|
)
|
|||||||||
|
Total shareholders' equity (deficit)
|
(
|
)
|
|
|||||||||
|
Total liabilities and shareholders' equity (deficit)
|
$
|
|
$
|
|
||||||||
|
March 30, 2026
|
/s/ Oren Shuster
|
/s/ Oz Adler
|
||
|
Date of approval of the
financial statements
|
Oren Shuster
Chief Executive Officer and Director
|
Oz Adler
Chairman of the Board
|
|
Year ended
December 31,
|
||||||||||||||||
|
Note
|
2025
|
2024
|
2023
|
|||||||||||||
|
Revenue
|
19A
|
$
|
|
$
|
|
$
|
|
|||||||||
|
Cost of revenue
|
19B
|
|
|
|
||||||||||||
|
Gross profit before fair value adjustments
|
|
|
|
|||||||||||||
|
Fair value adjustments:
|
||||||||||||||||
|
Realized fair value adjustments on inventory sold or impaired
|
|
|
(
|
)
|
||||||||||||
|
Total fair value adjustments
|
|
|
(
|
)
|
||||||||||||
|
Gross profit after fair value adjustments
|
|
|
|
|||||||||||||
|
Selling and marketing expenses
|
19C
|
|
|
|
||||||||||||
|
General and administrative expenses
|
19D
|
|
|
|
||||||||||||
|
Restructuring expenses
|
19E
|
|
|
|
||||||||||||
|
Other expenses
|
19F
|
|
|
|
||||||||||||
|
Share-based compensation
|
18D
|
|
|
|
||||||||||||
|
Total operating expenses
|
|
|
|
|||||||||||||
|
Operating loss
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Finance income
|
19G
|
|
|
|
||||||||||||
|
Finance expenses
|
19G
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Finance income (expense), net
|
(
|
)
|
(
|
)
|
|
|||||||||||
|
Loss before taxes on income (tax benefit)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Taxes on income (tax benefit)
|
20
|
|
(
|
)
|
|
|||||||||||
|
Net loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||
| Note |
Year ended
December 31,
|
||||||||||||||
|
|
2025
|
2024
|
2023 (*)
|
|
|||||||||||
|
Other comprehensive income that will not be reclassified to profit or loss in subsequent periods:
|
|||||||||||||||
|
Remeasurement gain on defined benefit plans
|
$
|
|
$
|
|
$
|
|
|||||||||
|
Total other comprehensive income that will not be reclassified to profit or loss in subsequent periods
|
|
|
|
||||||||||||
|
Other comprehensive loss that will be reclassified to profit or loss in subsequent periods:
|
|||||||||||||||
|
Adjustments arising from translation of financial statements of foreign operations
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Total other comprehensive loss
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Total comprehensive loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Net loss attributable to:
|
|||||||||||||||
|
Shareholders of the Company
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Non-controlling interests
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||
|
Total comprehensive loss attributable to:
|
|||||||||||||||
|
Shareholders of the Company
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Non-controlling interests
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||
|
Loss per share attributable to shareholders of the Company from net loss:
|
|||||||||||||||
|
Basic loss per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
|
Diluted loss per share (in CAD)
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||
| (*) |
Loss per share includes the effect of Reverse Share Split (see also Note 18A below).
|
|
Share capital and premium
|
Capital reserve from share-based payment transactions
|
Capital reserve from translation difference of foreign operations
|
Accumulated deficit
|
Total
|
Non-controlling interests
|
Total
Shareholders' equity
|
||||||||||||||||||||||
|
Balance as of January 1, 2023
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
|
$
|
|
|||||||||||||
|
Net loss
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||
|
Total other comprehensive income (loss)
|
|
|
(
|
)
|
|
(
|
)
|
|
(
|
)
|
||||||||||||||||||
|
Total comprehensive loss
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||
|
Common shares issued through private placements transactions, net of issuance costs (Note 18B1)
|
|
|
|
|
|
|
|
|||||||||||||||||||||
|
Common shares issued as debts settlement with related party (Note 18B2)
|
|
|
|
|
|
|
|
|||||||||||||||||||||
|
Other comprehensive loss classification
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|||||||||||||||||||||
|
Expired options
|
|
(
|
)
|
|
|
|
|
|
||||||||||||||||||||
|
Balance as of December 31, 2023
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||||||||
|
Share capital and premium
|
Capital reserve from share-based payment transactions
|
Amount received on account of financial instruments and other
|
Capital reserve from translation difference of foreign operations
|
Accumulated deficit
|
Total
|
Non-controlling interests
|
Total
Shareholders' equity
|
|||||||||||||||||||||||||
|
Balance as of January 1, 2024
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||||||||||
|
Net loss
|
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Total other comprehensive income (loss)
|
|
|
|
(
|
)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||
|
Total comprehensive loss
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||||
|
Common shares issued through private placement transaction, net of issuance costs (Note 18B3)
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Common shares issued as share-based compensation with related party (Note 18B4)
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Recognition of conversion feature related to convertible debentures (Note 15)
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Other comprehensive income classification
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||
|
Expired and exercised options
|
|
(
|
)
|
|
|
|
|
|
|
|||||||||||||||||||||||
|
Balance as of December 31, 2024
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
$
|
|
|||||||||||||
|
Share capital and premium
|
Capital reserve from share-based payment transactions
|
Amount received on account of financial instruments and other
|
Capital reserve from translation difference of foreign operations
|
Capital reserve from transaction with non-controlling interests
|
Capital reserve from transaction with main shareholder |
Accumulated deficit
|
Total
|
Non-controlling interests
|
Total
Shareholders' equity (deficit)
|
|||||||||||||||||||||||||||||||
|
Balance as of January 1, 2025
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
$
|
|
|||||||||||||||||
|
Net loss
|
|
|
|
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||||||||||||||||||
|
Total other comprehensive income (loss)
|
|
|
|
(
|
)
|
|
|
|
(
|
)
|
|
(
|
)
|
|||||||||||||||||||||||||||
|
Total comprehensive loss
|
|
|
|
(
|
)
|
|
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Expiration of conversion feature related to convertible debentures (Note 15)
|
|
|
(
|
)
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
|
Recognition of conversion feature related to convertible debentures (Note 15)
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||
|
Common shares issued upon partial conversion of convertible debentures (Notes 15 and 18B5)
|
|
|
(
|
)
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
|
Common shares issued as consideration upon acquisition on non-controlling interest (Note 18B6)
|
|
|
|
|
(
|
)
|
|
|
(
|
)
|
|
|
||||||||||||||||||||||||||||
|
Common shares issued upon debt settlement (Note 18B7)
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||
|
Net proceeds received upon completion of private placement transaction (Note 18B8)
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||
|
Common shares issued upon exercise of pre-funded warrants (Notes 18B4 and 18B8)
|
|
|
(
|
)
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
|
Recognition of capital contribution from a main shareholder (Note 18C)
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||
|
Share-based compensation
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||||||
|
Balance as of December 31, 2025
|
$
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
(
|
)
|
$
|
|
$
|
(
|
)
|
||||||||||||||||
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Cash flows provided by (used in) operating activities:
|
||||||||||||
|
Net loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Adjustments for non-cash items:
|
||||||||||||
|
Realized fair value adjustments on inventory sold or impaired
|
|
|
|
|||||||||
|
Revaluation of financial instruments
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Issuance costs allocated to warrants granted
|
|
|
|
|||||||||
|
Disposal of property, plant and equipment
|
|
|
|
|||||||||
|
Common shares and prefunded warrants issued as share-based compensation with related party
|
|
|
|
|||||||||
|
Discount expenses in respect of convertible debentures
|
|
|
|
|||||||||
|
Depreciation of property, plant and equipment
|
|
|
|
|||||||||
|
Amortization of intangible assets
|
|
|
|
|||||||||
|
Depreciation of right of use assets
|
|
|
|
|||||||||
|
Impairment of goodwill
|
|
|
|
|||||||||
|
Impairment of intangible assets
|
|
|
|
|||||||||
|
Finance income, net
|
|
|
|
|||||||||
|
Deferred tax payments (benefit), net
|
(
|
)
|
(
|
)
|
|
|||||||
|
Share-based payments
|
|
|
|
|||||||||
|
Loss from deconsolidation of Oranim
|
|
|
|
|||||||||
|
Revaluation expenses of investment in affiliate
|
|
|
|
|||||||||
|
Revaluation expenses (income) of loans receivables
|
|
(
|
)
|
|
||||||||
|
Changes in employee benefit liabilities, net
|
|
(
|
)
|
(
|
)
|
|||||||
|
Gain from debts restructuring
|
|
(
|
)
|
|
||||||||
|
Discount expenses in respect of credit
|
|
|
|
|||||||||
|
|
|
|
||||||||||
|
Changes in non-cash working capital:
|
||||||||||||
|
(Increase) decrease in trade receivables
|
|
(
|
)
|
|
||||||||
|
Decrease in other current assets
|
|
|
|
|||||||||
|
(Increase) decrease in inventory, net of fair value adjustments
|
(
|
)
|
|
|
||||||||
|
(Increase) decrease in trade payables
|
|
|
(
|
)
|
||||||||
|
(Increase) decrease in other current liabilities
|
|
(
|
)
|
(
|
)
|
|||||||
|
|
|
|
||||||||||
|
Taxes paid
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Net cash provided by (used in) operating activities
|
|
(
|
)
|
(
|
)
|
|||||||
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Cash flows used in investing activities:
|
||||||||||||
|
Purchase of property, plant and equipment
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds from sales of property, plant and equipment
|
|
|
|
|||||||||
|
Deconsolidation of subsidiary
|
|
(
|
)
|
|
||||||||
|
Loan granted
|
|
|
(
|
)
|
||||||||
|
Change in restricted cash
|
(
|
)
|
(
|
)
|
|
|||||||
|
Net cash used in investing activities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Cash provided by financing activities:
|
||||||||||||
|
Net proceeds allocated to issuance of share capital
|
|
|
|
|||||||||
|
Net proceeds allocated to issuance of warrants measured at fair value
|
|
|
|
|||||||||
|
Proceeds received from common shares issued upon pre-funded warrants exercised
|
(*)
|
|
|
|
||||||||
|
Repayment of lease liabilities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Payment of interest on lease liabilities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds from loans received
|
|
|
|
|||||||||
|
Repayment of loans
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Interest paid
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Proceeds received from discounted checks
|
(
|
)
|
|
|
||||||||
|
Net cash provided by financing activities
|
|
|
|
|||||||||
|
Effect of foreign exchange on cash
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Change in cash
|
|
(
|
)
|
(
|
)
|
|||||||
|
Cash at the beginning of year
|
|
|
|
|||||||||
|
Cash at the end of year
|
|
$
|
|
$
|
|
|||||||
|
Supplemental disclosure of non-cash activities:
|
||||||||||||
|
Right of use assets recognized with corresponding lease liabilities (Note 11)
|
$
|
|
$
|
|
$
|
|
||||||
|
Issuance of convertible debentures in exchange for loans (principal and interest) received (Note 15)
|
$
|
|
$
|
|
$
|
|
||||||
|
Common shares and warrants issued as debts settlement with related party (Note 18B1 and 18B2)
|
$
|
|
$
|
|
$
|
|
||||||
|
Common shares issued upon exercise of pre-funded warrants (Note 18B4)
|
$
|
|
$
|
|
$
|
|
||||||
|
Common shares issued upon partial conversion of convertible debentures (Note 18B5)
|
$
|
|
$
|
|
$
|
|
||||||
|
Common shares issued as debt settlement (Note 18B7)
|
$
|
|
$
|
|
$
|
|
||||||
|
Revaluation of put option liability versus equity
|
$
|
|
$
|
|
$
|
|
||||||
| (*) |
Represents an amount less than $1.
|
| NOTE 1 - |
GENERAL
|
| A. |
Corporate information
|
| B. |
Definitions
|
|
The Company, or IMCC
|
-
|
IM Cannabis Corp.
|
|
The Group
|
-
|
IM Cannabis Corp., its Subsidiaries
|
|
Subsidiaries
|
-
|
Companies that are controlled by the Company (as defined in IFRS 10) and whose accounts are consolidated with those of the Company
|
|
CAD or $
|
-
|
Canadian Dollar
|
|
US$
|
-
|
United States dollar
|
|
EUR |
- |
EURO |
|
NIS
|
-
|
New Israeli Shekel
|
| C. |
Impact of potential Germany's legalization of cannabis
|
| NOTE 1 - |
GENERAL (Cont.)
|
| D. |
Liquidity and capital resources and going concern
|
| E. |
Impact of continued interest rate on the Group's business activity
|
| NOTE 1 - |
GENERAL (Cont.)
|
| F. |
Impact of the security situation on the Group's business activity
|
| A. |
Statement of Compliance
|
| B. |
Basis of Measurement
|
| C. |
Use of Significant Accounting Estimates and Assumptions and Judgements
|
| D. |
The Functional Currency and the Presentation Currency
|
| A. |
Consolidated financial statements
|
|
Percentage ownership
|
||||
|
Subsidiaries
|
2025
|
2024
|
||
|
I.M.C. Holdings Ltd.
|
|
|
||
|
Focus Medical Herbs Ltd. (*)
|
|
|
||
|
Oranim Plus Pharm Ltd. (**)
|
|
|
||
|
Oranim Pharm (**)
|
|
|
||
|
IM Cannabis Holding NL B.V (***)
|
|
|
||
|
Adjupharm GmbH
|
|
|
||
|
I.M.C. Pharma Ltd.
|
|
|
||
|
I.M.C. Farms Israel Ltd. (****) |
|
|
||
|
I.M.C.C. Medical Herbs Ltd. (****)
|
|
|
||
|
R.A. Yarok Pharm Ltd.
|
|
|
||
|
Rosen High Way Ltd.
|
|
|
||
|
Revoly Trading and Marketing Ltd.
|
|
|
||
| (*) |
IMC Holdings held an option to acquire from main shareholders of the Company an ownership which represents a rate of
|
| (**) |
Was deconsolidated effective April 15, 2024 (see also Note 19F1 below).
|
| (***) |
Inactive entity.
|
| (****) |
In January 2025, the Israeli Companies Registrar approved a liquidation status for such entities which was completed during the year.
|
| B. |
Cash
|
| C. |
Inventories
|
| ◾ |
Raw materials - at cost of purchase using the "first-in, first-out" method.
|
| ◾ |
Work in progress and finished goods - on basis of average costs including materials, labor and other direct and indirect manufacturing costs based on normal capacity.
|
| ◾ |
Purchased merchandise and products - using the weighted average cost method or using the "first-in, first-out" method.
|
| D. |
Revenue recognition
|
| ◾ |
The reason for the bill-and-hold arrangement is substantive (for example, the customer has requested the arrangement);
|
| ◾ |
The product is identified separately as belonging to the customer;
|
| ◾ |
The product currently is ready for physical delivery to the customer;
|
| ◾ |
The Group does not have the ability to use the product by selling it or delivering it to another customer.
|
| E. |
Post-employment benefits
|
| F. |
Leases
|
In lease transaction under which the Group subleases underlying right-of-use asset while the lease (‘head lease’) with the original lessor remains in effect and the sublease term is for a period that is shorter than the remaining term of the head lease, it was determined that the sublease is classified as an operating lease. Thus, the Group retains the lease liability and the right-of-use asset relating to the head lease in the statement of financial position and continues to account for the original lease as described above. In addition, the Group recognizes lease income from sublease on a straight-line basis over the term of the sublease.
| G. |
Property, plant and equipment, net
|
|
%
|
|||
|
Building
|
|
||
|
Equipment and furniture
|
|
||
|
Vehicles
|
|
||
|
Computer, software and equipment
|
|
||
|
Leasehold improvements
|
()
|
| (*) |
Leasehold improvements are depreciated on a straight-line basis over the shorter of the lease term and the useful life of the improvement.
|
| H. |
Intangible assets, net
|
|
Years
|
|||
|
Cultivations and processing licenses
|
()
|
||
|
Customer relationships
|
|
||
|
Trade name
|
|
| (*) |
The licenses consisted of GMP and GDP licenses in Germany which have determined to have an indefinite useful life but were impaired in total amount of $
|
| I. |
Impairment of non-financial assets
|
| J. |
Financial instruments
|
| 1. |
Financial assets:
|
| - |
The Group’s business model for managing financial assets; and
|
| - |
The contractual cash flow terms of the financial asset.
|
| L. |
Financial instruments (Cont.)
|
| 2. |
Financial liabilities:
|
| K. |
Fair value measurement
|
|
Level 1
|
-
|
quoted prices (unadjusted) in active markets for identical assets or liabilities.
|
|
Level 2
|
-
|
inputs other than quoted prices included within Level 1 that are observable directly or indirectly.
|
|
Level 3
|
-
|
inputs that are not based on observable market data (valuation techniques which use inputs that are not based on observable market data).
|
| L. |
Provisions
|
| M. |
Taxes on income
|
| N. |
Issuance of a unit of securities
|
| O. |
Convertible debentures
|
| P. |
Put option granted to non-controlling interests
|
| Q. |
Share-based payment transactions
|
| R. |
Loss per share
|
| S. |
Operating cycle
|
| T. |
New standards adopted at January 1, 2025
|
| U. |
A summary of new reporting standards not yet effective and which are relevant to the Company's activities
|
| 1. |
IFRS 18, Presentation and Disclosure in Financial Statements
|
| ◾ |
IFRS 18 changes the structure of the profit or loss report and includes three new defined categories: operating, investment and financing and adds two new interim summaries: operating profit and profit before financing and income taxes.
|
| ◾ |
IFRS 18 includes guidelines for providing disclosure on performance indicators defined by management (management-defined performance measures).
|
| ◾ |
IFRS 18 provides guidelines regarding the aggregation and disaggregation of the information in the financial statements in relation to the question of whether information should be included in the main reports or in explanations and disclosures regarding items defined as "other".
|
| ◾ |
IFRS 18 includes amendments to other standards, including limited amendments to International Accounting Standard 7, Statement of Cash Flows.
|
| 2. |
Amendments to the Classification and Measurement of Financial Instruments - Amendments to IFRS 9 and IFRS 7
|
| ◾ |
A clarification that financial liability is derecognized on the ‘settlement date’ and introduction of an accounting policy choice (if specific conditions are met) to derecognize financial liabilities settled using an electronic payment system before the settlement date.
|
| ◾ |
Additional guidance on how the contractual cash flows for financial assets with environmental, social and corporate governance (ESG) and similar features should be assessed.
|
| ◾ |
Classification on what constitute ‘non-recourse features’ and what are the characteristics of contractually linked instruments.
|
| ◾ |
Introduction of disclosures for financial instruments with contingent features and additional disclosure requirements for equity instruments classified at fair value through other comprehensive income (OCI).
|
| NOTE 4 - |
SIGNIFICANT ACCOUNTING ESTIMATES AND ASSUMPTIONS USED IN THE PREPARATION OF THE FINANCIAL STATEMENTS
|
| A. |
Estimates and assumptions:
|
| - |
Impairment of inventory:
|
| - |
Determining the fair value of unquoted financial assets:
|
| - |
Impairment of goodwill and other intangible assets:
|
| - |
Legal claims:
|
| - |
Tax provision:
|
| NOTE 5 - |
TRADE RECEIVABLES
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Open balance
|
$
|
|
$
|
|
||||
|
Checks at a discount (*)
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| (*) |
Represents several identified outstanding unpaid invoices of certain customers that are used as guarantees for short-term loans received under execution of financing transactions of the Company with non-banking credit services entities. For more information, see Note 14C below.
|
| (**) |
Trade receivables are non-interest bearing and are generally on terms of
|
| NOTE 6 - |
OTHER CURRENT ASSETS
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Prepaid expenses
|
$
|
|
$
|
|
||||
|
Advances to suppliers
|
|
|
||||||
|
Government authorities
|
|
|
||||||
|
Former non-independent director
|
|
|
||||||
|
Shareholders (see Note 22A below)
|
|
|
||||||
|
Loans receivables
|
|
|
||||||
|
Other receivables
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 7 - |
INVENTORY
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Work in progress:
|
||||||||
|
Bulk cannabis
|
$
|
|
$
|
|
||||
|
Finished goods:
|
||||||||
|
Packaged dried cannabis
|
|
|
||||||
|
Other products
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 8 - |
INVESTMENT IN AFFILIATE
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Open balance
|
$
|
|
$
|
|
||||
|
Change of fair value in investment in affiliate (Note 19G)
|
|
(
|
)
|
|||||
|
Foreign currency translation
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 9 - |
PROPERTY, PLANT AND EQUIPMENT, NET
|
|
Building and
leasehold improvements |
Equipment and furniture
|
Computer, software and equipment
|
Vehicles
|
Total
|
||||||||||||||||
|
Cost:
|
||||||||||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|||||||||||||||
|
Additions during the year
|
|
|
|
|
|
|||||||||||||||
|
Disposals
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Deconsolidation of Oranim
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Foreign currency translation
|
|
|
|
(
|
)
|
|
||||||||||||||
|
Balance at December 31, 2024
|
|
|
|
|
|
|||||||||||||||
|
Additions during the year
|
|
|
|
|
|
|||||||||||||||
|
Foreign currency translation
|
|
|
|
(
|
)
|
|
||||||||||||||
|
Balance at December 31, 2025
|
|
|
|
|
|
|||||||||||||||
|
Accumulated depreciation:
|
||||||||||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|
|
|||||||||||||||
|
Depreciation during the year
|
|
|
|
|
|
|||||||||||||||
|
Disposals
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Deconsolidation of Oranim
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||||||
|
Foreign currency translation
|
|
|
|
(
|
)
|
|
||||||||||||||
|
Balance at December 31, 2024
|
|
|
|
|
|
|||||||||||||||
|
Depreciation during the year
|
|
|
|
|
|
|||||||||||||||
|
Foreign currency translation
|
|
|
|
(
|
)
|
|
||||||||||||||
|
Balance at December 31, 2025
|
|
|
|
|
|
|||||||||||||||
|
Depreciated cost at December 31, 2025
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||
|
Depreciated cost at December 31, 2024
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||
| NOTE 10 - |
GOODWILL AND INTANGIBLE ASSETS, NET
|
|
Cultivations and processing licenses (*)
|
Customer relationships
|
Trade name
|
Goodwill
|
Other
|
Total
|
|||||||||||||||||||
|
Cost:
|
||||||||||||||||||||||||
|
Balance at January 1, 2024
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
Impairment
|
|
|
|
(
|
)
|
|
(
|
)
|
||||||||||||||||
|
Deconsolidation of Oranim
|
|
(
|
)
|
|
(
|
)
|
|
(
|
)
|
|||||||||||||||
|
Foreign currency translation adjustments
|
|
|
|
|
|
|
||||||||||||||||||
|
Balance at December 31, 2024
|
|
|
|
|
|
|
||||||||||||||||||
|
Impairment
|
(
|
)
|
|
|
(
|
)
|
|
(
|
)
|
|||||||||||||||
|
Foreign currency translation adjustments
|
|
|
|
|
|
|
||||||||||||||||||
|
Balance at December 31, 2025
|
|
|
|
|
|
|
||||||||||||||||||
|
Accumulated amortization:
|
||||||||||||||||||||||||
|
Balance at January 1, 2024
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortization during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Deconsolidation of Oranim
|
|
(
|
)
|
|
|
|
(
|
)
|
||||||||||||||||
|
Balance at December 31, 2024
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortization during the year
|
|
|
|
|
|
|
||||||||||||||||||
|
Balance at December 31, 2025
|
|
|
|
|
|
|
||||||||||||||||||
|
Amortized cost at December 31, 2025
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
|
Amortized cost at December 31, 2024
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||
| (*) |
The licenses consisted of GMP and GDP licenses in Germany that had indefinitely useful life. The Company recognized an impairment loss in total amount of $
|
| NOTE 10 - |
GOODWILL AND INTANGIBLE ASSETS, NET (Cont.)
|
| NOTE 11 - |
LEASING
|
| A. |
Right of use assets:
|
|
|
Premises
|
Vehicles
|
Total
|
||||||||||
|
Cost:
|
||||||||||||
|
Balance at December 31, 2023
|
$
|
|
$
|
|
$
|
|
||||||
|
Changes during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Deconsolidation of Oranim
|
(
|
)
|
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
(
|
)
|
|
|
||||||||
|
Balance at December 31, 2024
|
|
|
|
|||||||||
|
Changes during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
|
|
|
|||||||||
|
Balance at December 31, 2025
|
|
|
|
|||||||||
|
Accumulated depreciation:
|
||||||||||||
|
Balance at December 31, 2023
|
|
|
|
|||||||||
|
Changes during the year:
|
||||||||||||
|
Depreciation and amortization
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Deconsolidation of Oranim
|
(
|
)
|
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
(
|
)
|
|
(
|
)
|
|||||||
|
Balance at December 31, 2024
|
|
|
|
|||||||||
|
Changes during the year:
|
||||||||||||
|
Depreciation and amortization
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
|
|
|
|||||||||
|
Balance at December 31, 2025
|
|
|
|
|||||||||
|
Depreciated cost at December 31, 2025
|
$
|
|
$
|
|
$
|
|
||||||
|
Depreciated cost at December 31, 2024
|
$
|
|
$
|
|
$
|
|
||||||
| NOTE 11 - |
LEASING (Cont.)
|
| B. |
Operating lease liabilities:
|
|
Premises
|
Vehicles
|
Total
|
||||||||||
|
Balance at January 1, 2024
|
$
|
|
$
|
|
$
|
|
||||||
|
Changes during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Payment of lease liabilities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Interest on lease liabilities
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Deconsolidation of Oranim
|
(
|
)
|
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
(
|
)
|
|
|
||||||||
|
Balance at December 31, 2024
|
|
|
|
|||||||||
|
Changes during the year:
|
||||||||||||
|
New leases
|
|
|
|
|||||||||
|
Payment of lease liabilities
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Interest on lease liabilities
|
|
|
|
|||||||||
|
Termination of leases
|
|
(
|
)
|
(
|
)
|
|||||||
|
Currency translation adjustments
|
(
|
)
|
|
(
|
)
|
|||||||
|
Balance at December 31, 2025
|
$
|
|
$
|
|
$
|
|
||||||
|
December 31
|
||||||||
|
2025
|
2024
|
|||||||
|
Current liability
|
$
|
|
$
|
|
||||
|
Non-current liability
|
|
|
||||||
|
Balance at December 31
|
$
|
|
$
|
|
||||
| C. |
Amounts recognized in statements of cash flow:
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Amortization of the right for use assets
|
$
|
|
$
|
|
$
|
|
||||||
|
Interest on lease liabilities
|
$
|
|
$
|
|
$
|
|
||||||
|
Payment of lease liabilities
|
$
|
|
$
|
|
$
|
|
||||||
| D. |
Analysis of contractual payment dates of leasing liability at December 31, 2025:
|
|
Up to a year
|
|
|||
|
Between 1-3 years
|
|
|||
|
Total (undiscounted)
|
|
| NOTE 12 - |
TRADE PAYABLES
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Open accounts in Israel
|
$
|
|
$
|
|
||||
|
Open accounts abroad
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 13 - |
OTHER CURRENT LIABILITIES
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Accrued expenses (*)
|
$
|
|
$
|
|
||||
|
Employees and payroll accruals
|
|
|
||||||
|
Government authorities
|
|
|
||||||
|
Advances from customers
|
|
|
||||||
|
Liability for restructuring
|
|
|
||||||
|
Other payables (**)
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| (*) |
Including outstanding amount related to management fee to be paid to entity controlled by main shareholders of the Company in total amount of $
|
| (**) |
Including subleasing income received in advance from an entity controlled by main shareholders in total amount of $
|
| NOTE 14 - |
OVERDRAFT AND CREDITS FROM BANK INSTITUTION AND OTHERS
|
| A. |
On May 17, 2023, the Company and Bank Mizrahi entered into credit facility with total commitment of up to NIS
In March 2025, the Company and Bank Mizrahi signed an agreement under which a short-term loan of NIS
|
| B. |
From time to time, in the normal course of business, the Company enters into financing transactions with non-financial institutions under which the Company receives loans that bear an interest at a fixed rate which shall be repaid together with the principal amount over a limited period (mainly short-term) as defined in each loan agreement. When loans received from related parties are considered as free interest loans or loans with reduced interest which do not represent the applicable rate of risk for the Company, the transaction is accounted for as a capital contribution from a main shareholder.
|
| C. |
From time to time, in the normal course of business, the Company enters into financing transactions with non-banking credit services entities under which the Company receives short-term loans that are guaranteed by certain identified outstanding unpaid invoices of certain customers (the “Selected Trade Receivables”). As it was determined that the Company has retained substantially all the risks and rewards of ownership of the Selected Trade Receivables, the Company continues to recognize the Selected Trade Receivables in their entirety and recognizes financial liability for consideration received as short-term loans. During the year ended December 31, 2025, the Company received sort-term loans under such arrangement in an aggregate amount of $
|
|
December 31,
|
||||||||||||
|
Interest rate
|
2025
|
2024
|
||||||||||
|
Overdraft and credit from bank institution
|
|
%
|
$
|
|
$
|
|
||||||
|
Credit from non-financial institutions
|
|
%
|
|
|
||||||||
|
Check receivables
|
|
%
|
|
|
||||||||
|
$
|
|
$
|
|
|||||||||
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Current maturity
|
$
|
|
$
|
|
||||
|
Long-term credit facility
|
|
|
||||||
|
$
|
|
$
|
|
|||||
| NOTE 15 - |
CONVERTIBLE DEBENTURES OFFERING
|
|
May 26, 2025
|
||||
|
Debentures (host instrument) (*)
|
$
|
|
||
|
Embedded conversion feature (residual amount)
|
|
|||
|
Total fair value of the convertible debentures
|
$
|
|
||
| (*) |
The fair value of the host instrument was measured by management using the assistance of an external appraiser taking into account a debt discount rate of
|
| NOTE 15 - |
CONVERTIBLE DEBENTURES OFFERING (Cont.)
|
|
Year ended
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Balance at January 1
|
$
|
|
$
|
|
||||
|
Issued of debentures as part of debt settlement
|
|
|
||||||
|
Derecognition of debentures as part of modification of terms
|
(
|
)
|
|
|||||
|
Issuance of debentures as part of modification of terms
|
|
|
||||||
|
Recognition of discount equal to embedded conversion feature
|
(
|
)
|
(
|
)
|
||||
|
Partial conversion into common shares (see Note 18B5 below)
|
(
|
)
|
|
|||||
|
Amortization of discount expenses
|
|
|
||||||
|
Balance at December 31
|
$
|
|
$
|
|
||||
| NOTE 16 - |
DERIVATIVE WARRANTS LIABILITIES AND PREFUNDED WARRANTS
|
| A. |
From time to time the Company entered into a non-brokered private placement offering transactions or settlement agreements under which the Company issued units that consist of common shares and warrants which are exercisable into common shares over a limited period of the time at an exercise price which is denominated in foreign currency and/or as the warrants might be exercisable to variable number of shares due to cashless exercise mechanism. As a result of the above, such warrants are accounted for as a derivative warrants liability which is measured at fair value through profits and losses.
|
|
December 31, 2025
|
December 31, 2024
|
|||||||||||||||||||||||
|
Series 2024 (*)
|
Series 2023
|
Series 2021
|
Series 2024
|
Series 2023
|
Series 2021
|
|||||||||||||||||||
|
Share price (in CAD)
|
|
|
|
|
|
|
||||||||||||||||||
|
Exercise price (in CAD)
|
|
|
|
|
|
|
||||||||||||||||||
|
Expected volatility (%)
|
|
%
|
|
%
|
|
%
|
|
%
|
|
%
|
|
%
|
||||||||||||
|
Risk-free interest rate (%)
|
|
%
|
|
%
|
|
%
|
|
%
|
|
%
|
|
%
|
||||||||||||
|
Dividend yield (%)
|
|
|
|
|
|
|
||||||||||||||||||
|
Expected term (years)
|
|
|
|
|
|
|
||||||||||||||||||
|
Number of warrants
|
|
|
|
|
|
|
||||||||||||||||||
|
Fair value per warrant (in CAD)
|
|
|
|
|
|
|
||||||||||||||||||
|
Series 2024
|
Series 2023
|
Series 2021
|
Total
|
|||||||||||||
|
Balances at January 1, 2023
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Issued (see Notes 18B1-18B2 below)
|
|
|
|
|
||||||||||||
|
Changes in fair value (**)
|
|
(
|
)
|
(
|
)
|
(
|
)
|
|||||||||
|
Balances at December 31, 2023
|
|
|
|
|
||||||||||||
|
Issued (see Note 18B3 below)
|
|
|
|
|
||||||||||||
|
Changes in fair value (**)
|
(
|
)
|
|
|
(
|
)
|
||||||||||
|
Balances at December 31, 2024
|
|
|
|
|
||||||||||||
|
Changes in fair value (**)
|
(*) (
|
)
|
(
|
)
|
|
(
|
)
|
|||||||||
|
Balances at December 31, 2025
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
| (*) |
Including, inter alia, the implication of execution of agreements dated August 13, 2025, with the holders of certain common share purchase warrants (the “Warrants”) originally issued as part of its private placement offering that closed on November 12, 2024 (the “2024 Private Placement”), pursuant to which the Company reduced the exercise price of each Warrant from $
|
| (**) |
See Note 19F below.
|
| B. |
For more information regarding prefunded warrants granted to Company's Chief Executive Officer, then Chairman of the Board and main shareholder, see also Note 18B4 below.
|
| NOTE 17 - |
CONTINGENT LIABILITIES
|
| A. |
Legal proceedings:
|
| 1. |
On November 19, 2021, Adjupharm filed a statement of claim (the “Claim”) to the District Court of Stuttgart (the “Stuttgart Court”) against Stroakmont & Atton Trading GmbH (“Stroakmont & Atton”), its shareholders and managing directors regarding a debt owed by Stroakmont & Atton to Adjupharm of approximately EUR
|
| NOTE 17 - |
CONTINGENT LIABILITIES (Cont.)
|
| A. |
Legal proceedings (Cont.):
|
| 2. |
On December 22, 2022, Uniclaro filed a statement of claim against Adjupharm with the district court in Hamburg, pursuant to which Uniclaro is claiming the purchase price for
|
| NOTE 17 - |
CONTINGENT LIABILITIES (Cont.)
|
|
|
A. |
Legal proceedings (Cont.):
|
| 3. |
On November 17, 2023, we received a copy of the 35 Oak Statement of Claim that was filed in the ONSC by 35 Oak Holdings Ltd., MW Investments Ltd., 35 Oak Street Developments Ltd., Michael Wiener, Kevin Weiner, William Weiner, Lily Ann Goldstein-Weiner, in their capacity as trustees of the Weiner Family Foundation (collectively the “MYM Shareholder Plaintiffs”) against us and our Board and officers, (collectively, the “MYM Defendants”).
|
|
NOTE 17 -
|
CONTINGENT LIABILITIES (Cont.)
|
|
|
B. |
Tax Remittance:
On October 31, 2023, notices of assessment for excise tax have been received covering the period commencing January 1, 2020 through December 31, 2021, under which the Company was assessed for tax on insurance, arrears interest and failure to file penalty in aggregate amount of $
On November 29, 2023, the Company filed notices of objection (Excise Tax Act) to the 2020-2021 Assessments. The Company assesses the filed notices of objection (Excise Tax Act) to be low to medium complexity.
On April 26, 2024, the Company received a letter from the Canada revenue agency that the notice of assessment for excise tax that the Company objected to is voided and no outstanding balance is owed with respect to such assessments. Based on the foregoing, this matter has been resolved to the Company's satisfaction and the objections were finalized.
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT)
|
| A. |
Composition of share capital:
|
|
|
December 31, 2025
|
December 31, 2024
|
|||||||||||||||
|
Authorized
|
Issued and outstanding
|
Authorized
|
Issued and outstanding
|
|||||||||||||
|
Number of shares
|
||||||||||||||||
|
Common Shares without par value
|
Unlimited
|
|
Unlimited
|
|
||||||||||||
| B. |
Changes in issued and outstanding share capital:
|
|
|
Number of shares
|
||||||||||||
|
2025
|
2024
|
2023 (*)
|
|
|||||||||
|
Balance as of January 1
|
|
|
|
|||||||||
|
Common shares issued through private placements offerings (1), (3), (8)
|
|
|
|
|||||||||
|
Common shares issued as debts settlement (2), (7)
|
|
|
|
|||||||||
|
Common shares issued as compensation to a related party (4)
|
|
|
|
|||||||||
|
Common shares issued upon RSUs vested
|
|
|
||||||||||
|
Common shares issued upon pre-funded warrants exercised (4), (8)
|
|
|
|
|||||||||
|
Common shares issued upon debentures converted (5)
|
|
|
|
|||||||||
|
Common shares issued as consideration upon acquisition of non-controlling interest (6)
|
|
|
|
|||||||||
|
Balance as of December 31
|
|
|
|
|||||||||
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| B. |
Change in issued and outstanding share capital (Cont.):
|
| 1. |
In January and February of 2023, the Company issued
|
| 2. |
On May 8, 2023, the Company closed a debt settlement transaction (the “Debt Settlement”) with L5 Capital Inc., a company controlled by Marc Lustig, the then executive chairman of the Board of the Company (“L5 Capital”), pursuant to which the Company settled outstanding indebtedness of US$
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| B. |
Change in issued and outstanding share capital Cont.):
|
| 3. |
On November 12, 2024, the Company closed a non-brokered private placement offering through issuance of
|
| 4. |
Since October 2022, the Company has borrowed from various institutions more than US$
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| B. |
Change in issued and outstanding share capital (Cont.):
|
| 4. |
(Cont.)
|
| 5. |
During the period commencing May 26, 2025 through December 31, 2025, total carrying amount of debentures of approximately $
|
| 6. |
In February 2024, the Company, through IMC Holdings, exercised an Option and acquired
|
| 7. |
On June 12, 2025, the Board approved the Company to enter into a debt settlement agreement with an unrelated service provider, pursuant to which unpaid fees, charges, and disbursements for legal services rendered to the Company in previous periods in total amount of $
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| B. |
Change in issued and outstanding share capital (Cont.):
|
|
On July 30, 2025 (the “Closing Date”), the Company closed a Private Placement Offering (the “Offering”) through issuance of
|
||
| 8. |
Each Warrant Share entitles its holder to purchase one common share at an exercise price of $
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| C. |
Capital reserve from transaction with main shareholder
|
| D. |
Share-based payment
|
| 1. |
Share option plan
|
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| D. |
Share option plan (Cont.)
|
| 2. |
The following table presents the Company’s options activity under the Stock Option Plan for the periods reported:
|
|
Year ended
December 31, 2025
|
||||||||
|
Number of options
|
Weighted average exercise price
|
|||||||
|
in CAD
|
||||||||
|
Options outstanding at the beginning of the year
|
|
|
||||||
|
Options forfeited during the year
|
(
|
)
|
|
|||||
|
Options outstanding at the end of year
|
|
|
||||||
|
Options exercisable at the end of year
|
|
|
||||||
|
Year ended
December 31, 2024
|
||||||||
|
Number of options
|
Weighted average exercise price
|
|||||||
|
in CAD
|
||||||||
|
Options outstanding at the beginning of the year
|
|
|
||||||
|
Options granted during the year
|
|
|
||||||
|
Options forfeited during the year
|
(
|
)
|
|
|||||
|
Options outstanding at the end of year
|
|
|
||||||
|
Options exercisable at the end of year
|
|
|
||||||
|
Year ended
December 31, 2023
|
||||||||
|
Number of options
|
Weighted average exercise price
|
|||||||
|
in CAD
|
||||||||
|
Options outstanding at the beginning of the year
|
|
|
||||||
|
Options granted during the year
|
|
|
||||||
|
Options forfeited during the year
|
(
|
)
|
|
|||||
|
Options outstanding at the end of year
|
|
|
||||||
|
Options exercisable at the end of year
|
|
|
||||||
| NOTE 18 - |
SHAREHOLDERS' EQUITY (DEFICIT) (Cont.)
|
| D. |
Share option plan (Cont.)
|
| 3. |
The following table presents the assumptions used to estimate the fair values of the options granted in the periods reported:
|
|
Year ended
December 31,
|
||||||||
|
2024
|
2023
|
|||||||
|
Share price (in CAD)
|
$
|
|
$
|
|
||||
|
Exercise price (in CAD)
|
$
|
|
$
|
|
||||
|
Expected life (years)
|
|
|
||||||
|
Volatility (%)
|
|
|
||||||
|
Annual risk-free rate (%)
|
|
|
||||||
|
Dividend yield (%)
|
|
|
||||||
| NOTE 19 - |
ADDITIONAL INFORMATION TO PROFIT OR LOSS ITEMS
|
| A. |
Additional information on revenue:
|
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Cannabis products
|
$
|
|
$
|
|
$
|
|
||||||
|
Other products
|
|
|
|
|||||||||
|
$
|
|
$
|
|
$
|
|
|||||||
| B. |
Cost of revenue:
|
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Salaries and related expenses
|
$
|
|
$
|
|
$
|
|
||||||
|
Materials
|
|
|
|
|||||||||
|
Write-off
|
|
|
|
|||||||||
|
Professional fees
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Miscellaneous
|
|
|
|
|||||||||
|
$
|
|
$
|
|
$
|
|
|||||||
| C. |
Selling and marketing expenses:
|
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Salaries and related expenses
|
$
|
|
$
|
|
$
|
|
||||||
|
Advertising
|
|
|
|
|||||||||
|
Professional fees
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Miscellaneous
|
|
|
|
|||||||||
|
$
|
|
$
|
|
$
|
|
|||||||
| D. |
General and administrative expenses:
|
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Salaries and related expenses
|
$
|
|
$
|
|
$
|
|
||||||
|
Insurance
|
|
|
|
|||||||||
|
Professional fees, net (*)
|
|
|
|
|||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Miscellaneous
|
|
|
|
|||||||||
|
$
|
|
$
|
|
$
|
|
|||||||
| (*) |
Includes management fee incurred indirectly through an entity controlled by the main shareholder. See Note 22B below.
|
| NOTE 19 - |
ADDITIONAL INFORMATION TO PROFIT OR LOSS ITEMS (Cont.)
|
| E. |
Restructuring expenses:
|
| F. |
Other expenses:
|
| 1. |
On April 15, 2024, the Company and the former shareholder of Oranim Plus Pharm Ltd. (“Oranim”) mutually agreed to terminate previous arrangements between then for purchasing
|
|
CURRENT ASSETS:
|
||||
|
Cash
|
$
|
|
||
|
Trade receivables
|
|
|||
|
Other current assets
|
|
|||
|
Inventory
|
|
|||
|
|
||||
|
NON-CURRENT ASSETS:
|
||||
|
Property, plant and equipment, net
|
|
|||
|
Right-of-use assets, net
|
|
|||
|
Intangible assets, net
|
|
|||
|
Goodwill
|
|
|||
|
|
||||
|
Total assets
|
$
|
|
||
|
CURRENT LIABILITIES:
|
||||
|
Trade payables
|
$
|
|
||
|
Other current liabilities
|
|
|||
|
Current maturities of operating lease liabilities
|
|
|||
|
|
||||
|
NON-CURRENT LIABILITIES:
|
||||
|
Operating lease liabilities
|
|
|||
|
Deferred tax liability, net
|
|
|||
|
|
||||
|
Total liabilities
|
$
|
|
||
|
Purchase consideration payable
|
$
|
|
||
|
Put option liability
|
$
|
|
||
| NOTE 19 - |
ADDITIONAL INFORMATION TO PROFIT OR LOSS ITEMS (Cont.)
|
| F. |
Other expenses (Cont.):
|
| 2. |
On June 30, 2023, an entity responsible for operating the Israeli medical cannabis distribution licensed center that was acquired within the Panaxia Transaction, ceased its operations at the licensed trading house located in Lod, Israel. Consequently, the Company transitioned the operation that was conducted through IMC Pharma to third-party entities and to its own trading house. During the year ended December 31, 2024, the Company recorded a goodwill impairment of $
|
| 3. |
For more information regarding impairment of intangible assets and goodwill allocated to Israeli CGU and German CGU, see Note 10 above.
|
| G. |
Finance expenses (income) net:
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Finance income:
|
||||||||||||
|
Revaluation of loans receivables
|
$
|
|
$
|
(
|
)
|
$
|
|
|||||
|
Change in fair value of derivative warrants liabilities (Note 16)
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Change in fair value of derivative pre-funded warrants liabilities (Note 18B4)
|
(
|
)
|
|
|
||||||||
|
Exchange rate differences and other finance income
|
(
|
)
|
(
|
)
|
(
|
) | ||||||
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
||||
|
Finance expenses:
|
||||||||||||
|
Compensation expenses in respect to guarantees granted by main shareholder
|
$
|
|
$
|
|
$
|
|
||||||
|
Change in fair value of derivative pre-funded warrants liabilities (Note 18B4)
|
|
|
|
|||||||||
|
Issuance costs allocated to derivative warrants liability (Notes 18B1, 18B3)
|
|
|
|
|||||||||
|
Extension fee and discount amortization expenses in respect to convertible debentures (Note 15)
|
|
|
|
|||||||||
|
Revaluation of investment in affiliate (Note 8)
|
|
|
|
|||||||||
|
Interest and discount amortization expenses on credit from bank institution and others (Note 14)
|
|
|
|
|||||||||
|
Impairment of loans receivables |
||||||||||||
|
Exchange rate differences and other finance income
|
|
|
|
|||||||||
|
$
|
|
$
|
|
$
|
|
|||||||
|
|
$
|
|
$
|
(
|
)
|
|||||||
| NOTE 20 - |
TAXES ON INCOME
|
| A. |
Tax rates applicable to the Group:
|
| 1. |
The Company is subject to tax rates applicable in Canada. The combined federal and provincial rate is
|
| 2. |
The Israeli subsidiaries are subject to Israeli corporate income tax rate of
|
| 3. |
The German subsidiary is subject to weighted tax rate of approximately
|
| B. |
Carryforward net operating losses for tax purposes:
|
| 1. |
As of December 31, 2025, the carryforward net operating tax losses of the Israeli subsidiaries amounted to approximately $
|
| 2. |
As of December 31, 2025, the carryforward net operating tax losses of the German subsidiary amounted to approximately $
|
| C. |
Income tax expense (tax benefit):
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Current taxes
|
$
|
|
$
|
|
$
|
|
||||||
|
Deferred taxes, net
|
(
|
)
|
(
|
)
|
|
|||||||
|
Taxes on income from previous years
|
|
(
|
)
|
|
||||||||
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||
| NOTE 20 - |
TAXES ON INCOME (Cont.)
|
| D. |
Reconciliation of tax expense (benefit) and the accounting loss multiplied by the Company's domestic tax rate for:
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Loss before income tax
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Statutory tax rate in Canada
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Increase (decrease) in income tax due to:
|
||||||||||||
|
Non-deductible expenses (non-taxable income), net for tax purposes
|
|
(
|
)
|
(
|
)
|
|||||||
|
Effect of different tax rates of subsidiaries
|
(
|
)
|
|
|
||||||||
|
Adjustments in respect of current income tax of previous years
|
|
(
|
)
|
|
||||||||
|
Recognition of tax benefits in respect of losses incurred in previous years
|
|
|
|
|||||||||
|
Unrecognized tax benefit in respect of losses incurred for the year
|
|
|
|
|||||||||
|
Other adjustments
|
|
|
|
|||||||||
|
Income tax expense (benefit)
|
$
|
|
$
|
(
|
)
|
$
|
|
|||||
| NOTE 21 - |
OPERATING SEGMENTS
|
| A. |
General information:
Since inception date, the operation of the Group has been conducted through one operating segment, (i.e., sales of medical cannabis products and other products) to customers through certain geographical areas (i.e. Israel and Germany).
|
|
Israel
|
Germany
|
Adjustments
|
Total
|
|||||||||||||
|
Year ended December 31, 2025
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
|||||
|
Unallocated corporate expenses
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||||
|
Total operating loss
|
$
|
(
|
)
|
|||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Year ended December 31, 2024
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss (income)
|
$
|
(
|
)
|
$
|
|
$
|
|
$
|
(
|
)
|
||||||
|
Unallocated corporate expenses
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||||
|
Total operating loss
|
$
|
(
|
)
|
|||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Year ended December 31, 2023
|
||||||||||||||||
|
Revenue
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Segment loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
$
|
(
|
)
|
|||||
|
Unallocated corporate expenses
|
$
|
(
|
)
|
$
|
(
|
)
|
||||||||||
|
Total operating loss
|
$
|
(
|
)
|
|||||||||||||
|
Depreciation, amortization and impairment
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
| B. |
Major customers
During the years ended December 31, 2025 and 2024, the Company had major customers which accounted for approximately
|
| NOTE 22 - |
BALANCES AND TRANSACTIONS WITH INTERESTED AND RELATED PARTIES
|
| A. |
Balances and transactions:
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Other current assets - main shareholders (Note 6)
|
$
|
|
$
|
|
||||
|
Other current liabilities - subleasing income received in advance from an entity controlled by main shareholders (None 13)
|
$
|
|
$
|
|
||||
|
Other current liabilities - management fee payable to an entity controlled by main shareholder (Note 13)
|
$
|
|
$
|
|
||||
|
Loans and accrued expenses from main shareholder (Note 14)
|
$
|
|
$
|
|
||||
|
Convertible debentures issued to main shareholders (Note 15)
|
$
|
|
$
|
|
||||
|
Derivative warrants liabilities and prefunded warrants issued to main shareholders (Note 16)
|
$
|
|
$
|
|
||||
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
General and administrative expenses - management fee (1)
|
$
|
|
$
|
|
$
|
|
||||||
|
Selling and marketing expenses - management fee (2)
|
$
|
|
$
|
|
$
|
|
||||||
|
General and administrative expenses - sublease income (3)
|
$
|
|
$
|
|
$
|
|
||||||
|
Finance income (expense) - Interest in income (expenses) and discount
amortization expenses on loans granted to (received from) main shareholders
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
|
||||
|
Finance income - Revaluation of derivative warrants liabilities granted to main shareholders
|
$
|
|
$
|
|
$
|
|
||||||
|
Finance (income) expenses - Revaluation of prefunded warrants granted to main shareholder
|
$
|
(
|
)
|
$
|
|
$
|
|
|||||
|
Finance expenses - Extension fee and discount amortization expenses in respect of convertible debentures granted to main shareholders
|
$
|
|
$
|
|
$
|
|
||||||
|
Finance expenses - Compensation expenses in respect of guarantees granted by main shareholder
|
$
|
|
$
|
|
$
|
|
||||||
| (1) |
Includes compensation for management services incurred indirectly by an entity controlled by the main shareholder.
|
| (2) |
On November 1, 2023, IMC Holdings signed a service agreement with a relative of one of the Company's main shareholders (the “Relative”) according to which the Relative provides IMC Holdings with strategy and marketing services within a defined hourly cap as set in the agreement, in return for a fixed monthly amount of
|
| NOTE 22 - |
BALANCES AND TRANSACTIONS WITH INTERESTED AND RELATED PARTIES (Cont.)
|
| A. |
Balances and transactions:
|
| (3) |
Includes income from subleasing of portion of premises to an entity controlled by the main shareholder based on sublease agreement executed on August 15, 2024, pursuant to IMC Holdings entitles to receive a monthly lease fee over the Initial Lease Period and the Option Period was determined at a fixed amount of
For more information regarding the first amendment of the sublease agreement, see Note 24D below.
|
| (4) |
For more information regarding execution of Note Purchase Agreement with certain investor, pursuant to which the Company issued a convertible note in aggregate principal amount of approximately US$
|
| B. |
Compensation of key management personnel of the Group:
|
|
Year ended
December 31,
|
||||||||||||
|
2025
|
2024
|
2023
|
||||||||||
|
Payroll and related expenses
|
$
|
|
$
|
|
$
|
|
||||||
|
Share-based compensation
|
$
|
|
$
|
|
$
|
|
||||||
|
Professional fees (*)
|
$
|
|
$
|
|
$
|
|
||||||
| (*) |
Includes management fees charges during the years ended December 31, 2025, 2024 and 2023 of $
|
| A. |
Financial risk management
|
| 1. |
General
The Company's activity exposes it to a variety of financial risks which include market risks, credit risks and liquidity risks. At each period, the Group examines the aforesaid financial risks and makes decisions accordingly.
Risk management is carried out by the Company's management, which identifies, evaluates and defines the risks as much as possible.
For more information regarding balance of financial assets and liabilities that are denominated or linked to foreign currency, see Note 23E below.
|
| 2. |
Financial risk factors
The Group has exposure to the following risks from its use of financial instruments:
|
| A. |
Unquoted equity instruments risk
Investment in unquoted equity instruments is sensitive to market price risk arising from uncertainties about future value of this investment. The Company's Board reviews and approves all decisions related to investment in unquoted equity instruments. As of December 31, 2025 and 2024, exposure to investment in one unquoted equity instrument measured at fair value was $
|
| B. |
Price risks of Company's common share
As of the reported dates, the Company is exposed to risks arising from changes in the price of its warrants and/or prefunded warrants measured at fair value through profit or loss as resulted from issuance of warrants to investors through private placement offering transaction and/or settlement agreements and issuance of the prefunded warrants as compensation to related party due to personal guarantees provided (see also Note 16 above). The fair value of such derivative liabilities is subject the Company to recognize losses in case there will be change in the price of the Company's ordinary share. However, the settlement of these liabilities will be through the exercise of the Company’s common shares.
|
| C. |
Liquidity risk
Since its inception date, the Company has financed its business activities through raising capital, inter alia, through public offering, non-brokered private placement transactions and credits from bank institution and others.
As of December 31, 2025 and 2024, the Company’s negative working capital amounted to $
The Company's policy is to manage its liquidity by examining current forecasts to manage the cash for operational needs in the normal course of business. Depending on current needs, the Company periodically carries out additional raising of capital.
|
| A. |
Financial risk management (Cont.)
|
| 2. |
Financial risk factors (Cont.)
|
| D. |
Credit risks
|
| 1. |
As of the reported dates, the cash is mostly deposited in various bank institutions in Israel and Germany. The management regularly evaluates the financial strength of the financial institutions with which the Company engages. Accordingly, the Company's management believes that the credit risk to these balances is low.
The Group is exposed to credit risks resulting from its operating activities (mainly from outstanding trade receivables). Upon the preparation of forecasted credit losses, the Company uses its previous experience and information accrued from different financial resources. Based on that, the Company segregates its customers to classes according to the different risk levels in a manner in which it can estimate the probability of a credit default.
|
| 2. |
Below is the breakdown of the Company’s financial assets subject to credit risks:
|
|
December 31,
|
||||||||
|
2025
|
2024 | |||||||
|
Cash
|
$
|
|
$
|
|
||||
|
Restricted cash
|
$
|
|
$
|
|
||||
|
Trade receivables
|
$
|
|
$
|
|
||||
| E. |
Currency rate risk:
As of December 31, 2024, a portion of the Group's financial assets and liabilities denominated in EUR, NIS and USD currency consist of cash and restricted cash in the amount of EUR
|
| B. |
Fair value of financial instruments
|
| C. |
A summary of financial instruments broken down by group:
|
|
December 31,
|
||||||||
|
2025
|
2024
|
|||||||
|
Financial assets measured at depreciated cost
|
||||||||
|
Cash
|
$
|
|
$
|
|
||||
|
Restricted cash
|
|
|
||||||
|
Trade receivables
|
|
|
||||||
|
Other current assets
|
|
|
||||||
|
|
|
|||||||
|
Financial liabilities measured at fair value
|
||||||||
|
Investments in affiliate
|
|
|
||||||
|
Financial liabilities measured at depreciated cost
|
||||||||
|
Operating leasing liabilities (including current maturity)
|
|
|
||||||
|
Credit from bank institution and others (including current maturity)
|
|
|
||||||
|
Convertible debentures
|
|
|
||||||
|
Trade payables
|
|
|
||||||
|
Other current liabilities
|
|
|
||||||
|
|
|
|||||||
|
Financial liabilities measured at fair value
|
||||||||
|
Derivative warrants liabilities and prefunded warrants
|
$
|
|
$
|
|
||||
| D. |
Company capital risk management policy
|
| NOTE 24 - |
SUBSEQUENT EVENTS
|
| A. |
Settlement agreements
|
| 1. |
Following to Note 17A3 above, on December 31, 2025, the Board approved the Company entering into settlement agreement pursuant to which the Company shall pay a total amount of $
|
| 2. |
On December 31, 2025, the Board approved the Company entering into settlement agreement under which the Company shall pay a total amount of $
|
| B. |
Convertible promissory notes offering
|
| NOTE 24 - |
SUBSEQUENT EVENTS (Cont.)
|
|
B. |
(Cont.) |
|
|
The Notes include customary limitations on conversion, including a beneficial ownership cap of
The Warrants entitle their holder to purchase one Common Share (each, a “Warrant Share”) at an exercise price of $
The Purchase Agreements include customary representations, warranties and covenants of the Company and the Investor, including the Company’s obligation to reserve sufficient Common Shares for issuance upon conversion of the Notes and to file a resale registration statement on Form F-3 (the “Registration Statement”) with the SEC providing for the resale by the Investor of the Common Shares issuable upon conversion of the Notes no later than 30 trading days within the date of each of the Purchase Agreement and the Additional Purchase Agreement. The Company has also agreed to use commercially reasonable efforts to cause such Registration Statement to become effective as soon as possible, but in no event later than the date which shall be the earlier of: (x) (i) in the event that the Registration Statement is not subject to a full review by the SEC, 60 calendar days after the closing date of the First Transaction and the Second Transaction, as applicable, or (ii) in the event that the Registration Statement is subject to a full review by the SEC, 90 calendar days after the closing date of the First Transaction and the Secon Transaction, as applicable, and (y) the 5th business day after the date on which the Company is notified (orally or in writing, whichever is earlier) by the SEC that such Registration Statement will not be reviewed or will not be subject to further review. On February 11, 2026, a Registration Statement was declared effective by the SEC.
|
| C. |
Loan agreements
|
| 1. |
On January 12, 2026, IMC Holdings entered into a loan agreement with non-financial institution, to borrow a principal amount of NIS
|
| 2. |
On February 3, 2026, Focus entered into a loan agreement with non-financial institution, to borrow a principal amount of NIS
|
| 3. |
On February 26, 2026, IMC Holdings entered into a loan agreement with a non-financial institution in the amount of NIS
|
| 4. |
On March 12, 2026, IMC Holdings entered into a loan agreement with a non-financial institution in the amount of NIS
|
| D. |
Following to Note 22B above, on February 15, 2026, IMC Holdings entered into first amendment to the sublease agreement with the Lessor, under which it was determined that the Lessor will sublease more spaces but the monthly lease fee will be remained on NIS
|
| E. |
Following to Note 24B above, in March 2026, the Company issued
|
|
| F. |
Following to Note 14B above, in March 2026, the Company extended the credit line received from Bank Mizrahi in total amount of NIS |